Академический Документы
Профессиональный Документы
Культура Документы
Acids
Загружено:
sayma_akhter5074Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Acids
Загружено:
sayma_akhter5074Авторское право:
Доступные форматы
NOCKHARDY
K NOTES
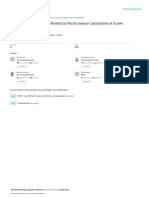
pH = log10 [ H [ H+(aq) ] = antilog ( pH) [ ] = concentration in mol dm-3
+ (aq)
ACIDS & BASES
]
GL AN C E
A LEVEL CHEMISTRY
AT
pH
[H+]
1 10
TWO THEORIES
-1
10 10 10 10 10 10 10 10
-2 - 11
10 10 10 10
-3 - 10 -9 -8 -7 -6
10
-4 -5
10
-5
-6
-7
-8
-9
LEWIS [OH]
10
ACIDS BASES
- 14
10 10 10
LONE PAIR ACCEPTORS LONE PAIR DONORS
- 13 - 12 pH
10
- 10 -4
10
10
- 11 -3
10
10
- 12 -2
10
10
- 13 -1
10
10
- 14
BRNSTED-LOWRY pH
strongly acidic weakly acidic 0 1 2 3 4 5 6
ACIDS BASES
PROTON DONORS PROTON ACCEPTORS
7 neutral
10 weakly alkaline
11
12
13
14 strongly alkaline
TYPES OF ACID IONIC PRODUCT OF WATER
HCl, H2SO4
Kw
STRONG
Completely dissociate into ions
HA(aq)
CH3COOH
>
(aq)
A(aq)
Water dissociates Kw = [H+(aq)] [OH(aq)]
H2O(l) =
H+(aq) + OH(aq) 10-14 mol2 dm-6 (at 25C)
WEAK
Partially dissociate into ions
HA(aq)
(aq)
A(aq)
The value of Kw varies with temperature - it is based on an equilibrium Temperature / C Kw / 10 -14 mol 2 dm-6 0 20 0.11 0.68 25 1.0 30 1.47 60 5.6
J. L. HOPTON 1999
www.XtremePapers.net
+ OH(aq) NH4
(aq) +
MONOPROTIC
HCl, CH3COOH, HNO3
DIPROTIC
H2SO4
TYPES OF BASE
STRONG
NaOH(s) > Na
(aq)
Ka
Ka =
[H
+ (aq)]
[A(aq)] [HA(aq)]
WEAK
NH3 (aq) + H2O(l)
+ OH(aq)
DISSOCIATION CONSTANT FOR A WEAK ACID
mol dm-3
Вам также может понравиться
- Vitamin CДокумент2 страницыVitamin CMiguel MercadoОценок пока нет
- Plant ReproductionДокумент72 страницыPlant Reproductionsayma_akhter5074Оценок пока нет
- Paper 7 Notes - FinalДокумент7 страницPaper 7 Notes - FinalUlee Abdul RaufОценок пока нет
- PH PR Lightexperiments PDFДокумент15 страницPH PR Lightexperiments PDFjameelmohamed1Оценок пока нет
- DNA AnalysisДокумент39 страницDNA AnalysisBio SciencesОценок пока нет
- Genetics FactsДокумент19 страницGenetics Factssayma_akhter5074Оценок пока нет
- Avogadro's ConstantДокумент9 страницAvogadro's Constantsayma_akhter5074Оценок пока нет
- CFTR and The Causes of CFДокумент12 страницCFTR and The Causes of CFsayma_akhter5074Оценок пока нет
- DnaДокумент32 страницыDnaBio SciencesОценок пока нет
- Cardiac CycleДокумент17 страницCardiac Cyclesayma_akhter5074Оценок пока нет
- CF Gene TherapyДокумент47 страницCF Gene Therapysayma_akhter5074Оценок пока нет
- Wave MotionДокумент1 страницаWave Motionsayma_akhter5074Оценок пока нет
- ReversibleДокумент15 страницReversiblekmoiz427Оценок пока нет
- Activation Energy and Reaction ProfileДокумент18 страницActivation Energy and Reaction Profilesayma_akhter5074Оценок пока нет
- Alkanes: Draw Out and Name The Structural Isomers of C H and C HДокумент7 страницAlkanes: Draw Out and Name The Structural Isomers of C H and C Hsayma_akhter5074Оценок пока нет
- Basic Notes On Exothermic and EndothermicДокумент19 страницBasic Notes On Exothermic and Endothermicsayma_akhter5074Оценок пока нет
- Nitrogen and Its CompoundsДокумент4 страницыNitrogen and Its Compoundssayma_akhter5074Оценок пока нет
- Chemical Kinetics (Rates of Reaction)Документ9 страницChemical Kinetics (Rates of Reaction)Syed OmerОценок пока нет
- Dynamics: Newton's Laws of MotionДокумент2 страницыDynamics: Newton's Laws of Motionsayma_akhter5074Оценок пока нет
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeОт EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeРейтинг: 4 из 5 звезд4/5 (5794)
- The Yellow House: A Memoir (2019 National Book Award Winner)От EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Рейтинг: 4 из 5 звезд4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceОт EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceРейтинг: 4 из 5 звезд4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersОт EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersРейтинг: 4.5 из 5 звезд4.5/5 (344)
- The Little Book of Hygge: Danish Secrets to Happy LivingОт EverandThe Little Book of Hygge: Danish Secrets to Happy LivingРейтинг: 3.5 из 5 звезд3.5/5 (399)
- The Emperor of All Maladies: A Biography of CancerОт EverandThe Emperor of All Maladies: A Biography of CancerРейтинг: 4.5 из 5 звезд4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaОт EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaРейтинг: 4.5 из 5 звезд4.5/5 (266)
- Never Split the Difference: Negotiating As If Your Life Depended On ItОт EverandNever Split the Difference: Negotiating As If Your Life Depended On ItРейтинг: 4.5 из 5 звезд4.5/5 (838)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryОт EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryРейтинг: 3.5 из 5 звезд3.5/5 (231)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureОт EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureРейтинг: 4.5 из 5 звезд4.5/5 (474)
- Team of Rivals: The Political Genius of Abraham LincolnОт EverandTeam of Rivals: The Political Genius of Abraham LincolnРейтинг: 4.5 из 5 звезд4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyОт EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyРейтинг: 3.5 из 5 звезд3.5/5 (2259)
- The Unwinding: An Inner History of the New AmericaОт EverandThe Unwinding: An Inner History of the New AmericaРейтинг: 4 из 5 звезд4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreОт EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreРейтинг: 4 из 5 звезд4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)От EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Рейтинг: 4.5 из 5 звезд4.5/5 (120)
- 2 - Low Temperature CorrosionДокумент65 страниц2 - Low Temperature CorrosiondennykvgОценок пока нет
- Drug Testing MethodsДокумент35 страницDrug Testing Methodswilly irawanОценок пока нет
- DLP G8 Q1 WK 3 D1 (11) JaneДокумент7 страницDLP G8 Q1 WK 3 D1 (11) JaneCathy MoronioОценок пока нет
- Physics Syllabus: GRADE 10 - 12Документ79 страницPhysics Syllabus: GRADE 10 - 12Mapalo Chirwa100% (4)
- 17 262 Specimen Blanking Machines For Specimen Preparation of Metals PI enДокумент5 страниц17 262 Specimen Blanking Machines For Specimen Preparation of Metals PI enDanan GentleОценок пока нет
- Polymers 14 03851Документ14 страницPolymers 14 03851Ricardo JVОценок пока нет
- Kemtek Ni - 512: Semi Bright Electroless Nickel ProcessДокумент4 страницыKemtek Ni - 512: Semi Bright Electroless Nickel ProcessAbbas RangoonwalaОценок пока нет
- Revision SPM 2018 Paper 2Документ70 страницRevision SPM 2018 Paper 2Azie Nurul Akhtar75% (4)
- Hesss Law WorksheetДокумент3 страницыHesss Law WorksheetAtulya BharadwajОценок пока нет
- BOSH Lecture 4 - Industrial HygieneДокумент72 страницыBOSH Lecture 4 - Industrial HygieneVitaliana VillaverdeОценок пока нет
- HIS Operation Centum VPДокумент16 страницHIS Operation Centum VPgopalОценок пока нет
- Himma2018-Review N2 - MembraneДокумент35 страницHimma2018-Review N2 - MembranefaezОценок пока нет
- Engineering Project Proposal by SlidesgoДокумент10 страницEngineering Project Proposal by SlidesgoMrtfthОценок пока нет
- Synthesis and Characterization of Nano Banana Fibre Reinforced Polymer Nano CompositesДокумент133 страницыSynthesis and Characterization of Nano Banana Fibre Reinforced Polymer Nano CompositesBoopathi RajaОценок пока нет
- Complex Formation Between Poly (Vinyl Alcohol) and Borate IonДокумент3 страницыComplex Formation Between Poly (Vinyl Alcohol) and Borate IonHaroon RashidОценок пока нет
- (Asce) PS 1949-1204 0000381Документ11 страниц(Asce) PS 1949-1204 0000381Hop Minh NguyenОценок пока нет
- Removing Contaminants From Crude Oil - AthlonДокумент5 страницRemoving Contaminants From Crude Oil - AthlonIrene CarrilloОценок пока нет
- 108 Chapter 3 StoichiometryДокумент29 страниц108 Chapter 3 Stoichiometryzabdullahstud1Оценок пока нет
- KU SPH 101 Electricity and Magnetism 1 NotesДокумент107 страницKU SPH 101 Electricity and Magnetism 1 NotesCallum Fallen100% (1)
- Classification of Economic: DR S e eДокумент2 страницыClassification of Economic: DR S e ekishan kumarОценок пока нет
- Square-Wave VoltammetryДокумент15 страницSquare-Wave VoltammetryNatasa VukicevicОценок пока нет
- Vasco 6000 EsiteДокумент2 страницыVasco 6000 EsiteBruno GomesОценок пока нет
- Pharmasutic (Physical Pharmacy) Assignment 01Документ14 страницPharmasutic (Physical Pharmacy) Assignment 01AAMIR NAWAZОценок пока нет
- Screw Compressors ReviewДокумент19 страницScrew Compressors ReviewCarlos Maldonado AlmeidaОценок пока нет
- Activated Sludge - Kinetic ModelДокумент19 страницActivated Sludge - Kinetic ModelDevendra SharmaОценок пока нет
- Fluid I - Lec 3 and 4 - ProductionДокумент34 страницыFluid I - Lec 3 and 4 - Productionamr mohamedОценок пока нет
- Sample Exit EXAM Perpared by Abel MДокумент6 страницSample Exit EXAM Perpared by Abel MTadesse MegersaОценок пока нет
- Theories On The Origin of The Solar System: By: Cuerpo, L.And Francisco, AДокумент21 страницаTheories On The Origin of The Solar System: By: Cuerpo, L.And Francisco, ARichell G.Оценок пока нет
- lt2 Science 3Документ4 страницыlt2 Science 3katherine corveraОценок пока нет
- Hydraulic Components and Systems (2012)Документ203 страницыHydraulic Components and Systems (2012)bach.leconmeomapОценок пока нет