Академический Документы
Профессиональный Документы
Культура Документы
RPH laNUn Amali Week 15 H1 P1
Загружено:
shehdilanunИсходное описание:
Оригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
RPH laNUn Amali Week 15 H1 P1
Загружено:
shehdilanunАвторское право:
Доступные форматы
PERAK MATRICULATION COLLEGE
MINISTRY OF EDUCATION MALAYSIA
CHEMISTRY UNIT
DAILY LESSON PLAN RECORD
PRACTI CAL
MOHAMAD SHEHDI BIN RAGIUN
SEMESTER : I SESSION : 2008 / 2009
DAY : Monday DATE : 8 / 9 / 2008
TIME : 11.00 am – 1.00 pm
8 Ramadhan 1429H
PRACTICAL CLASS : H1 P1 CHEMISTRY LAB : 6
EXPERIMENT : 8 TITLE : Chemical Equilibrium
OBJECTIVES:
1. To study the effect of concentration and temperature on chemical equilibrium.
2. To determine the equilibrium constant, Kc, of a reaction.
REFLECTION :
Today are 8th days fasting for Muslims in Ramadhan al Mubarak. This is the second last
experiment for sem I. Syukur Alhamdulillah lab goes on well. No accident occurs. As
usual, I discuss with the students observation they obtain from the experiment. I also
asked them to calculate the Kc. Summit lab report for this experiment next week.
Insya 4W1.
P/s- Next week no experiment. Just quiz 7 cover chapter 6 Chemical Equilibrium.
NOTES :
The factors that influence chemical equilibrium are concentration, temperature, pressure (for
reactions that involve gases). Le Chatelier’s principle is used to determine the position of the
equilibrium when one of the above factors is changed.
Le Chatelier’s principle states that if a system at equilibrium is disturbed by a change in
temperature, a change in pressure or a change in the concentration of one or more components,
the system will shift its equilibrium position in such a way as to counteract the effects of the
disturbance.
REFERENCES :
1) Chemistry Laboratory Manual Semester I & II, Eight Edition, 2008
2) Chemistry for Matriculation semester I , Second edition, Tan Yin Toon
3) Chemistry for Matriculation semester I, Third edition, Ahmad Kamal Ariffin Abdul Jamil,
Siti Asiah Ahmad Junan, Seng Chye Eng, dan Lim Boon Tik.
4) Kimia Fizikal Asas Matrikulasi, Norbani Abdullah etc. 1998, Kuala Lumpur, Fajar Bakti
5) Kimia Untuk Jurutera dan Ahli teknologi, Halimaton Hamdan etc, 1994, UTM skudai
6) Kimia (Kimia Fizik), Jilid I, STPM (962), Lee Chwee Neo, 1998, Penerbitan Pelangi, JB
7) Chemistry Syllabus Specification, BMKPM
Assessor’s Comment :
……………………………………………………………………………………………………………
……………………………………………………………………………………………………………
Ahlan wa sahlan ya Ramadhan al Mubarak
Happy fasting to all my muslims student May our fasting is accept by
4W1 Insya 4W1 Amin
Вам также может понравиться
- Kinetics and Dynamics of Elementary Gas Reactions: Butterworths Monographs in Chemistry and Chemical EngineeringОт EverandKinetics and Dynamics of Elementary Gas Reactions: Butterworths Monographs in Chemistry and Chemical EngineeringОценок пока нет
- AP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeОт EverandAP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeОценок пока нет
- RPH laNUn Amali Week 15 H3 P27Документ2 страницыRPH laNUn Amali Week 15 H3 P27shehdilanunОценок пока нет
- RPH laNUn Tutor Week 15 H8 T46Документ1 страницаRPH laNUn Tutor Week 15 H8 T46shehdilanunОценок пока нет
- 3 PDFДокумент2 страницы3 PDFMiguel AngelОценок пока нет
- RPH laNUn Kuliah SelasaДокумент1 страницаRPH laNUn Kuliah SelasashehdilanunОценок пока нет
- Minggu 1 Dan 2-Kimia Dasar TIДокумент18 страницMinggu 1 Dan 2-Kimia Dasar TIMaiasi Agung RahayuОценок пока нет
- Chem 107Документ4 страницыChem 107WaqasОценок пока нет
- Orientation On GEN CHEM 1 SY 2021 2022Документ30 страницOrientation On GEN CHEM 1 SY 2021 2022JohnОценок пока нет
- National University of Engineering: Electronics Engineering Program - ChemistryДокумент2 страницыNational University of Engineering: Electronics Engineering Program - ChemistryghmoralesfОценок пока нет
- HCHE112 - Physical Chemistry Module Course OutlineДокумент3 страницыHCHE112 - Physical Chemistry Module Course OutlinetkubvosОценок пока нет
- Jawahar Navodaya Vidyalaya, Purulia (W. B) : Name-Smriti Mandal Class-Xii Aissce Roll No.Документ19 страницJawahar Navodaya Vidyalaya, Purulia (W. B) : Name-Smriti Mandal Class-Xii Aissce Roll No.smritymandal883Оценок пока нет
- Lesson Plan CHE430/ CHE495 Organic Chemistry/ Hydrocarbon Chemistry Faculty of Chemical Engineering Universiti Teknologi MaraДокумент9 страницLesson Plan CHE430/ CHE495 Organic Chemistry/ Hydrocarbon Chemistry Faculty of Chemical Engineering Universiti Teknologi MaraMalik HamzaОценок пока нет
- Chem Course OutlineДокумент20 страницChem Course Outlineowenyang1992Оценок пока нет
- Chemistry 9-12 - Curriculum - Revised by HST - 2014 Bishoftu BДокумент258 страницChemistry 9-12 - Curriculum - Revised by HST - 2014 Bishoftu Bberitube77Оценок пока нет
- 4.208 M. Sc. Chemistry Part I Sem I II PDFДокумент35 страниц4.208 M. Sc. Chemistry Part I Sem I II PDFShivam MishraОценок пока нет
- Spring 2012 CHEM. 002 SyllabusДокумент5 страницSpring 2012 CHEM. 002 SyllabusRanvirОценок пока нет
- Chemical Analysis (Full Permission)Документ2 страницыChemical Analysis (Full Permission)Riswan RiswanОценок пока нет
- Lab 4 BCHCL 138EДокумент176 страницLab 4 BCHCL 138ELucky YadavОценок пока нет
- Course Information: Middle East Technical UniversityДокумент5 страницCourse Information: Middle East Technical UniversityErgin ÖzdikicioğluОценок пока нет
- Sheridan Courses OutlinesДокумент25 страницSheridan Courses Outlinestai_sao_khong_1307Оценок пока нет
- MSCCH 503Документ241 страницаMSCCH 503Rahul KashyapОценок пока нет
- Turbo MachineryДокумент24 страницыTurbo MachineryAazim FaridyОценок пока нет
- sc32223 m5 Chemistry 2017 Semester 2Документ5 страницsc32223 m5 Chemistry 2017 Semester 2api-219303331Оценок пока нет
- CH 2351Документ7 страницCH 2351Shanmuga PriyaОценок пока нет
- Palm Acid OilДокумент13 страницPalm Acid OilRaziah Ahmad ZaideiОценок пока нет
- Elective ChemistryДокумент86 страницElective ChemistryGenevieve OwusuОценок пока нет
- Chem 103Документ7 страницChem 103dheeraj105Оценок пока нет
- CHEM 351 Organic Chemistry I SP23 SyllabusДокумент5 страницCHEM 351 Organic Chemistry I SP23 SyllabusajamufamОценок пока нет
- General Chemistry: Southern Leyte State University Hinunangan CampusДокумент33 страницыGeneral Chemistry: Southern Leyte State University Hinunangan CampusLlyОценок пока нет
- Chemistry PDFДокумент78 страницChemistry PDFNicholas SaahОценок пока нет
- 1-SCH 101 - Introduction To Gas Laws and Kinetic Theory-1Документ26 страниц1-SCH 101 - Introduction To Gas Laws and Kinetic Theory-1Peter MbuguaОценок пока нет
- Physical Chemistry - Kinetic T3kДокумент9 страницPhysical Chemistry - Kinetic T3ksolhhОценок пока нет
- Co CHM 117 Summer 2018Документ11 страницCo CHM 117 Summer 2018Kamruzzaman ShiponОценок пока нет
- Basic Petroleum ChemistryДокумент162 страницыBasic Petroleum Chemistryenviroash100% (1)
- Course Syllabus Physical CheДокумент7 страницCourse Syllabus Physical CheOsama MohsinОценок пока нет
- National University of Engineering College of Civil Engineering Civil Engineering ProgramДокумент3 страницыNational University of Engineering College of Civil Engineering Civil Engineering ProgramAnonimus290599Оценок пока нет
- CHEM 0100 Course OutlineДокумент8 страницCHEM 0100 Course OutlineAkisha Ali-KhanОценок пока нет
- Chemistry For EngineersДокумент4 страницыChemistry For EngineersRainОценок пока нет
- Std12 Chem 2Документ405 страницStd12 Chem 2RajОценок пока нет
- Chem110 (5484)Документ7 страницChem110 (5484)chanddeveshОценок пока нет
- COURSE GUIDE IN SCIED 225 (Chemistry For Teachers) First Semester SY 2020-2021Документ3 страницыCOURSE GUIDE IN SCIED 225 (Chemistry For Teachers) First Semester SY 2020-2021Ybur Clieve Olsen DahilogОценок пока нет
- General Chemistry Laboratory Manual: Yildiz Technical University Faculty of Art & Science Chemistry DepartmentДокумент36 страницGeneral Chemistry Laboratory Manual: Yildiz Technical University Faculty of Art & Science Chemistry DepartmentLOLOОценок пока нет
- ENGINEERING 3014 Engineering ChemistrySummerДокумент2 страницыENGINEERING 3014 Engineering ChemistrySummerKevin HuangОценок пока нет
- Pilani Campus: Birla Institute of Techonology and Science, PilaniДокумент3 страницыPilani Campus: Birla Institute of Techonology and Science, PilaniBala KrishnaОценок пока нет
- National University of EngineeringДокумент2 страницыNational University of EngineeringMarco A. Castillo LudeñaОценок пока нет
- 02cy0451 Physical Chemistry IIДокумент5 страниц02cy0451 Physical Chemistry IIDwivelia AftikaОценок пока нет
- Chemical EquilibriumДокумент67 страницChemical Equilibriummukesh kumar_0709Оценок пока нет
- Typical Questions That CRE May AnswerДокумент6 страницTypical Questions That CRE May Answercarleston thurgoodОценок пока нет
- M.Sc. Part 1 Sem 1 (Wef 2021-22)Документ22 страницыM.Sc. Part 1 Sem 1 (Wef 2021-22)Shifa ChaudhariОценок пока нет
- Course Syllabus AND Instructor Plan: Waco, TexasДокумент6 страницCourse Syllabus AND Instructor Plan: Waco, TexasAnthony EmerencianaОценок пока нет
- Kinetics of Chemical Processes: Butterworth-Heinemann Series in Chemical EngineeringОт EverandKinetics of Chemical Processes: Butterworth-Heinemann Series in Chemical EngineeringОценок пока нет
- Microwave Assisted Chemistry Experiments: (Organic Synthesis, Chemical analysis and Extraction)От EverandMicrowave Assisted Chemistry Experiments: (Organic Synthesis, Chemical analysis and Extraction)Оценок пока нет
- Novel Nanomaterials for Biomedical, Environmental and Energy ApplicationsОт EverandNovel Nanomaterials for Biomedical, Environmental and Energy ApplicationsXiaoru WangОценок пока нет
- Chemical Thermodynamics: Plenary Lectures Presented at the Fourth International Conference on Chemical Thermodynamics Université des Sciences et Techniques de Languedoc, Montpellier, France 26–30 August 1975От EverandChemical Thermodynamics: Plenary Lectures Presented at the Fourth International Conference on Chemical Thermodynamics Université des Sciences et Techniques de Languedoc, Montpellier, France 26–30 August 1975J. RouquerolОценок пока нет
- Student's Guide to Fundamentals of Chemistry: Brescia, Arents, Meislich, TurkОт EverandStudent's Guide to Fundamentals of Chemistry: Brescia, Arents, Meislich, TurkРейтинг: 3 из 5 звезд3/5 (1)
- Pictures of EarthДокумент21 страницаPictures of EarthKosla Vepa99% (113)
- Students H8 Buffer SolutionДокумент1 страницаStudents H8 Buffer SolutionshehdilanunОценок пока нет
- SK017 Paper2 Sem1 0708Документ3 страницыSK017 Paper2 Sem1 0708shehdilanunОценок пока нет
- Objektif Q SKO17 Set 2Документ4 страницыObjektif Q SKO17 Set 2shehdilanunОценок пока нет
- Past Year Analysis SK017Документ2 страницыPast Year Analysis SK017shehdilanunОценок пока нет
- Flash Flood in BoholДокумент32 страницыFlash Flood in BoholshehdilanunОценок пока нет
- Objektif Q SKO17 Set 1Документ4 страницыObjektif Q SKO17 Set 1shehdilanunОценок пока нет
- Objektif Q SKO17 Set 3Документ4 страницыObjektif Q SKO17 Set 3shehdilanunОценок пока нет
- Sem1 Unit6 Chemical EquilibriumДокумент3 страницыSem1 Unit6 Chemical Equilibriumshehdilanun100% (1)
- Sem1 Unit7 Ionic EquilibriaДокумент5 страницSem1 Unit7 Ionic Equilibriashehdilanun50% (2)
- Sem1 Unit8 Phase EquilibriaДокумент5 страницSem1 Unit8 Phase EquilibriashehdilanunОценок пока нет
- Kalendar LanunДокумент1 страницаKalendar LanunshehdilanunОценок пока нет
- Sem1 Unit4 Periodic TableДокумент10 страницSem1 Unit4 Periodic Tableshehdilanun0% (1)
- Sem1 Unit5 Chemical BondingДокумент6 страницSem1 Unit5 Chemical Bondingshehdilanun100% (1)
- The Art of IllnessДокумент9 страницThe Art of IllnessKhayaal100% (4)
- Forecast FiNaL SK 027 0809Документ2 страницыForecast FiNaL SK 027 0809shehdilanunОценок пока нет
- Sem1 Unit1 MatterДокумент9 страницSem1 Unit1 Mattershehdilanun100% (2)
- Sem1 Unit3 Atomic StructureДокумент8 страницSem1 Unit3 Atomic Structureshehdilanun100% (1)
- Sem1 Unit2 States of MatterДокумент7 страницSem1 Unit2 States of MattershehdilanunОценок пока нет
- Redox Chart Ion Equation ChemistryДокумент2 страницыRedox Chart Ion Equation ChemistryshehdilanunОценок пока нет
- Bond Polarity and Inductive EffectsДокумент1 страницаBond Polarity and Inductive EffectsshehdilanunОценок пока нет
- Greatness of All Ah S (1) .W.T ..Документ11 страницGreatness of All Ah S (1) .W.T ..api-3730124Оценок пока нет
- Set IV Objective Sem IIДокумент6 страницSet IV Objective Sem IIshehdilanunОценок пока нет
- Why Do Grignard Reagents React With Carbonyl Compounds?Документ2 страницыWhy Do Grignard Reagents React With Carbonyl Compounds?shehdilanunОценок пока нет
- Chapter 16 OH LaNunДокумент7 страницChapter 16 OH LaNunshehdilanun100% (1)
- The Boiling Point of Comparable Molar MassДокумент2 страницыThe Boiling Point of Comparable Molar MassshehdilanunОценок пока нет
- Memorise Those BrandsДокумент14 страницMemorise Those BrandszaihasrizaОценок пока нет
- How Study Chem ChartДокумент1 страницаHow Study Chem ChartshehdilanunОценок пока нет
- Past Year Analysis Sk017Документ2 страницыPast Year Analysis Sk017shehdilanunОценок пока нет
- Chp.7 EnergyConversionДокумент80 страницChp.7 EnergyConversionVisuОценок пока нет
- Thermo QualsДокумент26 страницThermo QualsLuc LeОценок пока нет
- KSA Requirements For FOA CFOT CertificationДокумент4 страницыKSA Requirements For FOA CFOT Certificationmohamed ghazyОценок пока нет
- Microsoft Word MecvinaДокумент37 страницMicrosoft Word MecvinaRam RamirezОценок пока нет
- T2 Skript CH 4Документ26 страницT2 Skript CH 4Muhammad SaleemОценок пока нет
- Glass Handbook 2010 English For Eastern Europe PDFДокумент300 страницGlass Handbook 2010 English For Eastern Europe PDFAnthony Thaddeus AntonioОценок пока нет
- Four Types of Climate in The PhilippinesДокумент2 страницыFour Types of Climate in The PhilippinesAnonymous P1iMibОценок пока нет
- Experiment 2: Free and Force Vortex Table of ContentДокумент33 страницыExperiment 2: Free and Force Vortex Table of Contentmarz95Оценок пока нет
- NGV 2 2007Документ78 страницNGV 2 2007eko handoyoОценок пока нет
- Fluid Mechanics Answers PDFДокумент141 страницаFluid Mechanics Answers PDFazamatОценок пока нет
- Gmaw FcawДокумент80 страницGmaw FcawmarboledtОценок пока нет
- Moving LoadsДокумент21 страницаMoving LoadsJimboy S. CastuloОценок пока нет
- General Comparison Between Aisc LRFD and Asd: Hamid ZandДокумент107 страницGeneral Comparison Between Aisc LRFD and Asd: Hamid Zandadnan85% (13)
- Review of Literature On Steam AccumulatorДокумент10 страницReview of Literature On Steam Accumulatortamil vaananОценок пока нет
- Solutions Manual For College Physics 11e (Volume 2) Raymond Serway Chris VuilleДокумент70 страницSolutions Manual For College Physics 11e (Volume 2) Raymond Serway Chris Vuillesobiakhan52292Оценок пока нет
- 7 MultiphaseFreeSurfaceFilmДокумент25 страниц7 MultiphaseFreeSurfaceFilmJesthyn VC100% (1)
- Case Study - Single-Plane Rotor Balancing On A Disk RotorДокумент11 страницCase Study - Single-Plane Rotor Balancing On A Disk RotorMotionicsОценок пока нет
- Final Report - NTPCДокумент31 страницаFinal Report - NTPCVibhor MishraОценок пока нет
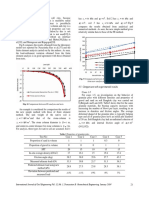
- Kpa C Kpa C Kpa C: Fig. 9 Comparison Between Bearing Capacity Values DeterminedДокумент5 страницKpa C Kpa C Kpa C: Fig. 9 Comparison Between Bearing Capacity Values DeterminedfacedoneОценок пока нет
- EE ProblemsДокумент33 страницыEE ProblemsSaied Aly SalamahОценок пока нет
- 2211 TCP Mechanical Firing Head 130618Документ1 страница2211 TCP Mechanical Firing Head 130618mahsaОценок пока нет
- LPT SopДокумент7 страницLPT SopQUALITYОценок пока нет
- 2023 July Nov ME TA RevДокумент4 страницы2023 July Nov ME TA RevMd IsmayeelОценок пока нет
- E300 List DatasetsДокумент5 страницE300 List Datasetsrezagholami870% (1)
- API 610 10th Edition (Iso 13709) Pump DatasheetДокумент3 страницыAPI 610 10th Edition (Iso 13709) Pump DatasheetMuhammad Azka50% (4)
- Spe 17927 PaДокумент6 страницSpe 17927 PaAlejandro MonroyОценок пока нет
- Dynamic Balancing Machines, Soft-Bearing vs. Hard-BearingДокумент2 страницыDynamic Balancing Machines, Soft-Bearing vs. Hard-BearingS Kumar Pa VeluОценок пока нет
- Piping PresentationДокумент144 страницыPiping PresentationSUNIL TVОценок пока нет
- Atomic Absorption SpectrometryДокумент64 страницыAtomic Absorption Spectrometryanilrockzzz786Оценок пока нет
- Sewa AlatДокумент12 страницSewa Alatcitra puspita sari100% (1)