Академический Документы
Профессиональный Документы
Культура Документы
Photoluminescence
Загружено:
Mustaque AliИсходное описание:
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Photoluminescence
Загружено:
Mustaque AliАвторское право:
Доступные форматы
Introduction to Photoluminescence Spectroscopy
Lecturer: Shou-Yi Kuo
Outline
1.
Fundamental physics
Electron in solids Properties of semiconductors
2.
Experimental Setup
Element of PL spectroscopy
3.
Practical examples
III-Nitride materials Quantum structures
1. Fundamental physics
Electron
in solids
-Formation of energy bands -Classification of solids
Properties
of semiconductors
-Intrinsic and extrinsic semiconductors -Size effects
The Band Theory of Solids
Energy level
Energy bands
Formation of Energy Bands
What Is a Semiconductor ?
Conductivity
Definition
Metals: Good Conductors! Semiconductor/Semimetal: Insulators:
No rigid boundaries!
Semimetal Metal
Bandgap
Definition
Semiconductor: ~ Small bandgap insulator
Strictly speaking, must be capable of being doped
Typical semiconductor bandgaps:
0 ~ Eg ~3.5 eV
Metals & semimetals: Eg = 0 eV Insulators: Eg > 3.5 eV
10
Typical Band Structures at T = 0 K
Band gap Band gap
11
Metal, Semiconductors and Insulators
12
Metal, Semiconductors and Insulators
13
Classification of Semiconductors
14
15
16
Semiconductor Bandstructure
band structure
17
Band gap
18
Elemental Semiconductors and Dopants
19
Elemental and Compound Semiconductors
20
Properties of Elemental and Compound Semiconductors
21
Pure Semiconductors
22
Electrical Conduction in Intrinsic Silicon at T = 0 K
23
Electrical Conduction in Intrinsic Silicon at T > 0 K
24
Charge Carriers in Intrinsic Semiconductors
25
Charge Carriers Concentration And Fermi Level
26
Intrinsic Conductivity
27
What is the Density of States ?
The density of states is the number of states per unit volume per unit energy interval that are available for occupation by electrons (or holes). Optical absorption must be proportional to the density of states, because a photon cannot be absorbed if there is no final state available for the electronic transition. For 3-D parabolic bands (bulk), N(E)= (1/2p2)(2m*/2)3/2E, where m* is the effective mass of the electron. For a 2-D quantum well, N(E)= m*/p2, independent of E. For each quantum state in the quantum well, there will be a step in the density of states.
28
Fermi Distribution Function Density of States and filled Electron States
29
Band Gap and Concentration of Charge Carriers Vs. Temperature
30
Semiconductor Doping
Doped semiconductors are called extrinsic semiconductors. One places a small amount of impurity atoms into the pure element melt, e.g. arsenic atoms into silicon crystals. The extra electron of arsenic is not paired, so it is forced up into the gap between the valence and conduction bands. The net result is that photons of lower energy can ionize an electron or hole to produce a carrier.
31
Doped Semiconductor
ED EA
Some combinations of elements may not be viable.
Lattice structure breaks down at interesting concentration levels.
Difficult to do the chemistry to make it.
32
n-type doping
Phosphorous (or other column V element) atom replaces silicon atom in crystal lattice. Since phosphorous has five outer shell electrons, there is now an extra electron in the structure. Material is still charge neutral, but very little energy is required to free the electron for conduction since it is not participating in a bond.
ED
ED (eV)
33
p-type doping
Boron (or other column III element) has been added to silicon. There is now an incomplete bond pair, creating a vacancy for an electron. Little energy is required to move a nearby electron into the vacancy. Like a moving bubble in liquids. As the hole propagates, positive charge is moved across the silicon.
EA
EA (eV)
34
Donors and Acceptors
Calculation using the hydrogen model HH+ + e-
35
Ionization energies of impurities
36
Intrinsic and Extrinsic (n- and p-Type) Semiconductors
37
Distribution Function, Density of States and filled Electron states
38
Semiconductor Energy Band Model
Recall
39
Energy Band Model for a Doped Semiconductor
40
Energy Band Model for Compensated Semidonductor
41
Electrical Conductivity vs. Temperature
42
Charge Carrier Concentration vs. Reciprocal Temperature
43
Extrinsic, Saturation and Intrinsic Behavior vs. Reciprocal Temperature
44
45
46
47
Energy Quantization :Bulk material
48
2D nanostructure :quantum well
49
1D nanostructure :quantum wire
50
0D nanostructure :quantum dot
dispersion
51
For 3-D parabolic bands (bulk), N(E)= (1/22)(2m*/2)3/2E, where m* is the effective mass of the electron. For a 2-D quantum well, N(E)= m*/2, independent of E. For each quantum state in the quantum well, there will be a step in the density of states.
52
2. Experimental Setup
Element
of PL spectroscopy - Monochromator - Light source - Detector
53
Wavelength range
54
How to measure a spectrum
55
Optical Absorption
56
57
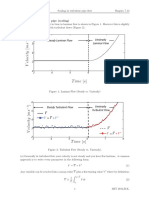
Photoluminescence (PL) basics
Types of luminescence
Cathodoluminescence
Cathode rays (TV and PCs display) Photoluminescence Photon excitation (fluorescent lights) Electroluminescence Electrical injection (LED.s)
58
Luminescence
Classification
Direct Eg, h = Eg energy given off in process.
Fast: 10-8 s or less If after excitation turned off, emission stops in about 10-8 s , called fluorescence. If emission stops in about seconds or even minutes, called phosphorescence. Special materials called: phosphors. thermal re-excitation involved. Color (frequency) depends on impurity since many transitions may involved with (Et).
59
Monochromator
60
Slit
Вам также может понравиться
- 6th Central Pay Commission Salary CalculatorДокумент15 страниц6th Central Pay Commission Salary Calculatorrakhonde100% (436)
- Applied Thermodynamics by Onkar Singh.0001 PDFДокумент330 страницApplied Thermodynamics by Onkar Singh.0001 PDFImran TahirОценок пока нет
- Introduction of Powder Diffraction INDIAДокумент397 страницIntroduction of Powder Diffraction INDIAJuan Pablo Cano Mejia100% (1)
- Mass Spectrometry-Based ProteomicsДокумент10 страницMass Spectrometry-Based ProteomicsChimie OficialОценок пока нет
- Scanning Electron Microscopy (SEM)Документ4 страницыScanning Electron Microscopy (SEM)Er Rachit ShahОценок пока нет
- Graphene From Fundamental-From Fundamental To Future ApplicationsДокумент40 страницGraphene From Fundamental-From Fundamental To Future ApplicationsMustaque Ali100% (2)
- Graphene From Fundamental-From Fundamental To Future ApplicationsДокумент40 страницGraphene From Fundamental-From Fundamental To Future ApplicationsMustaque Ali100% (2)
- XRD TechnicsДокумент90 страницXRD TechnicsAloke VermaОценок пока нет
- Electrochemistry PresentationДокумент36 страницElectrochemistry PresentationMuhammad HaziqОценок пока нет
- Spinel Ferrite Nanostructures for Energy Storage DevicesОт EverandSpinel Ferrite Nanostructures for Energy Storage DevicesRajaram S. ManeРейтинг: 5 из 5 звезд5/5 (1)
- (UV Vis) SpectrosДокумент4 страницы(UV Vis) SpectrosGarion Charles0% (1)
- Raman SpectrosДокумент8 страницRaman SpectrosVarnakavi Naresh100% (1)
- Fourier Transform Infrared SpectrosДокумент27 страницFourier Transform Infrared Spectrosrmarin_90Оценок пока нет
- Faraday CupsДокумент10 страницFaraday CupsAshish SharmaОценок пока нет
- Essay RamanДокумент3 страницыEssay RamanChu Wai Seng100% (1)
- Current Topics in Amorphous Materials: Physics & TechnologyОт EverandCurrent Topics in Amorphous Materials: Physics & TechnologyY. SakuraiРейтинг: 5 из 5 звезд5/5 (1)
- Optical Properties of Materials: Reflection, Refraction, Absorption & MoreДокумент18 страницOptical Properties of Materials: Reflection, Refraction, Absorption & MoreAshish Manatosh BarikОценок пока нет
- The Raman EffectДокумент12 страницThe Raman Effectmkbh10Оценок пока нет
- Electrochemical Impedance SpectrosДокумент14 страницElectrochemical Impedance SpectrosFelipe Cepeda Silva100% (1)
- Nuclear Magnetic Resonance Spectros PDFДокумент310 страницNuclear Magnetic Resonance Spectros PDFValemtinoОценок пока нет
- Composite RestorationДокумент54 страницыComposite RestorationVincent SerОценок пока нет
- Conductor of Heat and Electricity QuizДокумент5 страницConductor of Heat and Electricity QuizDaryl OribiadaОценок пока нет
- Implementation of A Two-Surface Plasticity Model For Cyclic Loading in Sand Into The Finite Element MethodДокумент92 страницыImplementation of A Two-Surface Plasticity Model For Cyclic Loading in Sand Into The Finite Element MethodGuillaume MELINОценок пока нет
- Heat transfer calculations through pipes and wallsДокумент26 страницHeat transfer calculations through pipes and wallsAntonio MoralesОценок пока нет
- Fluvial Processes and River EngineeringДокумент6 страницFluvial Processes and River Engineeringagussantoso_st0% (3)
- Uvvis SpectrosДокумент35 страницUvvis Spectrosumesh123patilОценок пока нет
- SEM GuideДокумент98 страницSEM GuideMustaque AliОценок пока нет
- Thermal Evaporation NotesДокумент8 страницThermal Evaporation NotesMrunalОценок пока нет
- Scanning Electron MicrosДокумент14 страницScanning Electron Microszhoushengyuanly100% (1)
- Electron MicrosДокумент11 страницElectron MicrosAbir RoyОценок пока нет
- Electrical and Optical Properties of SB SnO2 Thin Films Obtained by TheДокумент6 страницElectrical and Optical Properties of SB SnO2 Thin Films Obtained by TheFarah HananiОценок пока нет
- UV Spectrophotometer ExplainedДокумент14 страницUV Spectrophotometer ExplainedKrishna DabgarОценок пока нет
- Nanosheet 09179023Документ7 страницNanosheet 09179023ck maitiОценок пока нет
- X-Ray DiffractionДокумент30 страницX-Ray DiffractionMerve Ayvaz KöroğluОценок пока нет
- Atomic Force MicrosДокумент9 страницAtomic Force MicrosPranjalОценок пока нет
- SRM University Nano MaterialsДокумент37 страницSRM University Nano MaterialsVikram JainОценок пока нет
- Resonance Raman SpectrosДокумент14 страницResonance Raman SpectrosSarthak Mandal0% (1)
- FTIR Spectros PDFДокумент3 страницыFTIR Spectros PDFRidaSirtaDewiTRОценок пока нет
- Nanotechnology Lesson For MS StudentsДокумент6 страницNanotechnology Lesson For MS StudentssavithatssОценок пока нет
- Intro To Mass Spec-Part 4-Tandem MS-Pao-Chi Liao PDFДокумент46 страницIntro To Mass Spec-Part 4-Tandem MS-Pao-Chi Liao PDFvijaykavatalkar100% (1)
- Unit 3 PolymersДокумент19 страницUnit 3 PolymersengineeringchemistryОценок пока нет
- XRD ReportДокумент13 страницXRD ReportMukulОценок пока нет
- XRD Analysis TechniqueДокумент33 страницыXRD Analysis TechniqueprinceamitОценок пока нет
- LCMS Introduction Part 2Документ34 страницыLCMS Introduction Part 2rostaminasabОценок пока нет
- SEM for materials characterization: a versatile tool for quantitative 3D analysisДокумент4 страницыSEM for materials characterization: a versatile tool for quantitative 3D analysisReyesHerreraSckalОценок пока нет
- Fabry Perot PDFДокумент2 страницыFabry Perot PDFAnitaОценок пока нет
- Photoluminescence Efficiency in Wide-Band-Gap III-nitride Semiconductors and Their Heterostructures (PDFDrive)Документ122 страницыPhotoluminescence Efficiency in Wide-Band-Gap III-nitride Semiconductors and Their Heterostructures (PDFDrive)bayou71usaОценок пока нет
- 13C NMR SpectrosДокумент16 страниц13C NMR Spectrosapi-3723327100% (4)
- Electronic SpectrosДокумент82 страницыElectronic SpectrosEub EuОценок пока нет
- Ftir SpectrosДокумент36 страницFtir SpectrosHafiz Mudaser AhmadОценок пока нет
- Nanoclusters: Properties, Production and ApplicationsДокумент24 страницыNanoclusters: Properties, Production and ApplicationsRaj Zas0% (1)
- Nano LithographyДокумент14 страницNano LithographyMohammad RameezОценок пока нет
- Surface Enhanced Raman SpectrosДокумент23 страницыSurface Enhanced Raman SpectrosRevati MutturОценок пока нет
- Polarized Light Microscopy Principles and Practice PDFДокумент15 страницPolarized Light Microscopy Principles and Practice PDFIgor GabrielОценок пока нет
- Carbon Nanotube CompositesДокумент8 страницCarbon Nanotube CompositesgnanalakshmiОценок пока нет
- MolproДокумент591 страницаMolproKalakkan KollamОценок пока нет
- X-Ray Reflectivity Studies of Thin FilmДокумент9 страницX-Ray Reflectivity Studies of Thin Filmarpon32100% (2)
- Presentation For Polymer ScienceДокумент18 страницPresentation For Polymer ScienceBoier Sesh Pata100% (1)
- NMR Coupling Constants (JДокумент72 страницыNMR Coupling Constants (JShrutiОценок пока нет
- The ancient properties of nanoclusters: Understanding the structure and applications of nanoclustersДокумент32 страницыThe ancient properties of nanoclusters: Understanding the structure and applications of nanoclustersBhawana VermaОценок пока нет
- Organic-Inorganic Hybrid Perovskite Solar CellsДокумент16 страницOrganic-Inorganic Hybrid Perovskite Solar Cellssalome RaymondОценок пока нет
- High Spin and Low Spin ComplexesДокумент13 страницHigh Spin and Low Spin ComplexesMa'arif A. SyafiiОценок пока нет
- UV-Vis Spectroscopy: Understanding Chromophores and SolventsДокумент50 страницUV-Vis Spectroscopy: Understanding Chromophores and Solventssharifah sakinah syed soffianОценок пока нет
- Laser: Department of Applied SciencesДокумент64 страницыLaser: Department of Applied SciencesAnimesh BiswasОценок пока нет
- Contact AngleДокумент7 страницContact AngleOh Ha Ni OthmanОценок пока нет
- Geophysical Techniques Lab Report:: Ferromagnetism, para Magnetism and DiamagnetismДокумент4 страницыGeophysical Techniques Lab Report:: Ferromagnetism, para Magnetism and DiamagnetismDeniz AkoumОценок пока нет
- Jacques Tissue OpticsДокумент60 страницJacques Tissue OpticsrickОценок пока нет
- Transition Metal ToxicityОт EverandTransition Metal ToxicityG. W. RichterОценок пока нет
- JEOLguideДокумент36 страницJEOLguideVânia BuenoОценок пока нет
- Sem 4Документ29 страницSem 4Raluca CristacheОценок пока нет
- Photo-Responsive Properties of Organic Inorganic MatricesДокумент6 страницPhoto-Responsive Properties of Organic Inorganic MatricesMustaque AliОценок пока нет
- Optical property and crystallinities of Si doped β-Ga2O3 thin filmsДокумент4 страницыOptical property and crystallinities of Si doped β-Ga2O3 thin filmsMustaque AliОценок пока нет
- Basics of Molecular Beam EpitaxyДокумент8 страницBasics of Molecular Beam EpitaxyMustaque AliОценок пока нет
- Manual Load CalculationДокумент6 страницManual Load Calculationganesh gundОценок пока нет
- ASTM5Документ1 227 страницASTM5Guatavo91Оценок пока нет
- Buildings Ductile For Good Seismic PerformanceДокумент2 страницыBuildings Ductile For Good Seismic PerformanceWaqas AhmedОценок пока нет
- Vapor-Liquid Equilibria of Nonideal Solutions: Harrison C. Carlson, and Allan P. ColburnДокумент10 страницVapor-Liquid Equilibria of Nonideal Solutions: Harrison C. Carlson, and Allan P. ColburnAlfonso Dominguez GonzalezОценок пока нет
- Emf and Internal Resistance QДокумент32 страницыEmf and Internal Resistance QJellyfishОценок пока нет
- Bernoulli Venturi NozzleДокумент4 страницыBernoulli Venturi NozzleFrank PingolОценок пока нет
- Lecture 13 - Welding DefectsДокумент4 страницыLecture 13 - Welding DefectsMichael TayactacОценок пока нет
- Degrees of Freedom Analysis for Process UnitsДокумент7 страницDegrees of Freedom Analysis for Process UnitsAhmed AliОценок пока нет
- 10 1016@j Matchemphys 2019 05 033Документ13 страниц10 1016@j Matchemphys 2019 05 033Deghboudj SamirОценок пока нет
- TSGENCO Syllabus For ECE, Mechanical, Civil, EEE 2022 - Jobriya - InfoДокумент12 страницTSGENCO Syllabus For ECE, Mechanical, Civil, EEE 2022 - Jobriya - InfoMuhammad Rizwan ur RehmanОценок пока нет
- Designing Structural Components Against Failure Under Static LoadsДокумент8 страницDesigning Structural Components Against Failure Under Static LoadsArpit sharma0% (1)
- CB 352 - Construction Materials Questions and Problems (1) : Arab Academy For Science Technology Faculty of EngineeringДокумент1 страницаCB 352 - Construction Materials Questions and Problems (1) : Arab Academy For Science Technology Faculty of EngineeringAhmed Nageeb Elbelacy0% (1)
- Productflyer - 978 0 387 74364 6Документ1 страницаProductflyer - 978 0 387 74364 6Dhruv PatelОценок пока нет
- Dislocations and Strengthening: Chapter OutlineДокумент19 страницDislocations and Strengthening: Chapter OutlineAshok KumarОценок пока нет
- General Description: SeriesДокумент18 страницGeneral Description: Series123Оценок пока нет
- 2 - 25F13 - Shap7.14 - SHAPIROДокумент3 страницы2 - 25F13 - Shap7.14 - SHAPIROBruno Henrique HoinschyОценок пока нет
- NORSOK M-710 2014 Qualification of Non-Metallic Materials and Manufacturers - PolymersДокумент24 страницыNORSOK M-710 2014 Qualification of Non-Metallic Materials and Manufacturers - PolymersWHWENОценок пока нет
- TDS Zircoseal 300 PPGДокумент2 страницыTDS Zircoseal 300 PPGsimphiweОценок пока нет
- Classnote 1 PDFДокумент78 страницClassnote 1 PDFCJ SpencerОценок пока нет
- Destructive Testing Fusion Welded Joints Weld Metal in SteelДокумент16 страницDestructive Testing Fusion Welded Joints Weld Metal in SteelAarenLiОценок пока нет
- PPTCHAEN21 - Lecture Unit 2. Material Property ChartsДокумент20 страницPPTCHAEN21 - Lecture Unit 2. Material Property ChartsLady PenalozaОценок пока нет
- Certificado Calidad BobinaДокумент1 страницаCertificado Calidad BobinaRoxyОценок пока нет
- Unit 1 Mom (12 Me 33)Документ85 страницUnit 1 Mom (12 Me 33)mona5181Оценок пока нет