Академический Документы
Профессиональный Документы
Культура Документы
Chemistry Web-Book (Raymond Chang) Answers To Even-Numbered Problems
Загружено:
RSLИсходное описание:
Оригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Chemistry Web-Book (Raymond Chang) Answers To Even-Numbered Problems
Загружено:
RSLАвторское право:
Доступные форматы
Answers to Even-Numbered Problems
Chapter 1
1.4 (a) Hypothesis, (b) law, (c) theory. 1.12 (a) Physical
change, (b) chemical change, (c) physical change, (d) chemical
change, (e) physical change. 1.14 (a) K, (b) Sn, (c) Cr, (d) B,
(e) Ba, (f) Pu, (g) S, (h) Ar, (i) Hg. 1.16 (a) Homogeneous
mixture, (b) element, (c) compound, (d) homogeneous mixture,
(e) heterogeneous mixture, (f) homogeneous mixture, (g) heterogeneous mixture. 1.22 1.30 3 103 g. 1.24 (a) 41C, (b)
11.3F, (c) 1.1 3 104F. 1.26 (a) 2196C, (b) 2269C, (c)
328C. 1.30 (a) 0.0152, (b) 0.0000000778. 1.32 (a) 1.8 3
1022, (b) 1.14 3 1010, (c) 25 3 104, (d) 1.3 3 103. 1.34 (a)
One, (b) three, (c) three, (d) four, (e) two or three, (f) one, (g)
one or two. 1.36 (a) 1.28, (b) 3.18 3 1023 mg, (c) 8.14 3 107
dm. 1.38 (a) 1.10 3 108 mg, (b) 6.83 3 1025 m3. 1.40 3.1557
3 107 s. 1.42 (a) 81 in/s, (b) 1.2 3 102 m/min, (c) 7.4
km/h. 1.44 88 km/h. 1.46 3.7 3 1023 g Pb. 1.48 (a) 8.56C,
(b) 2459.67F, (c) 7.12 3 1025 m3, (d) 7.2 3 103 L. 1.50 6.25
3 1024 g/cm3. 1.52 (a) Chemical, (b) chemical, (c) physical,
(d) physical, (e) chemical. 1.54 2.6 g/cm3. 1.56 0.882
cm. 1.58 767 mph. 1.60 Liquid must be less dense than ice;
temperature below 0C. 1.62 2.3 3 103 cm3. 1.64 6.3.
1.66 73S. 1.68 (a) 8.6 3 103 L air/day. (b) 0.018 L CO/day.
1.70 5.5 3 1010 L seawater. 1.72 7.0 3 1020 L. 1.74 9.0 3 101
lb Os. 1.76 O: 4.0 3 104 g; C: 1.1 3 104 g; H: 6.2 3 103 g; N:
2 3 103 g; Ca: 9.9 3 102 g; P: 7.4 3 102 g. 1.78 4.6 3 102C;
8.6 3 102F. 1.80 $9.0 3 1011 1.82 5.4 3 1022 Fe atoms.
1.84 36 times. 1.86 1.450 3 1022 mm. 1.88 4 3 103 m.
1.90 1.3 3 103 mL chlorine solution. 1.92 2.5 nm. 1.94 5.5.
Chapter 2
2.8 0.12 mi. 2.14 145. 2.16 N(7,8,7); S(16,17,16); Cu(29,34,29);
Sr(38,46,38); Ba(56,74,56); W(74,112,74); Hg(80,122,80).
201
2.18 (a) 186
74 W. (b) 80Hg. 2.24 (a) Metallic character increases
down a group, (b) metallic character decreases from left to
right. 2.26 F and Cl; Na and K; P and N. 2.32 (a) H2 and F2.
(b) HCl and CO. (c) S8 and P4. 2.34 (protons, electrons):
K1(19,18); Mg21(12,10); Fe31(26,23); Br2(35,36);
Mn21(25,23); C42(6,10); Cu21(29,27). 2.42 (a) AlBr3. (b)
NaSO2. (c) N2O5. (d) K2Cr2O7. 2.44 C2H6O. 2.46 Ionic: NaBr,
BaF2, CsCl. Molecular: CH4, CCl4, ICl, NF3. 2.54 (a) potassium hypochlorite, (b) silver carbonate, (c) nitrous acid, (d)
potassium permanganate, (e) cesium chlorate, (f) potassium
ammonium sulfate, (g) iron(II) oxide, (h) iron(III) oxide, (i) titanium(IV) chloride, (j) sodium hydride, (k) lithium nitride, (l)
sodium oxide, (m) sodium peroxide. 2.56 (a) CuCN, (b)
Sr(ClO2)2, (c) HBrO4, (d) HI, (e) Na2(NH4)PO4, (f) PbCO3,
(g) SnF2, (h) P4S10, (i) HgO, (j) Hg2I2, (k) SeF6. 2.58 C-12 and
C-13. 2.60 I2. 2.62 NaCl is an ionic compound. 2.64 (a) SO2,
(b) S8, (c) Cs, (d) N2O5, (e) O, (f) O2, (g) O3, (h) CH4,
(i) KBr, (j) S, (k) P4, (l) LiF. 2.66 (a) CO2 (solid), (b) NaCl,
(c) N2O, (d) CaCO3, (e) CaO, (f) Ca(OH)2, (g) NaHCO3, (h)
Na2CO3 ? 10H2O, (i) CaSO4 ? 2H2O, (j) Mg(OH)2. 2.68 (a)
Ionic compounds formed between metallic and nonmetallic elements. 2.70 23Na. 2.72 Hg and Br2. 2.74 H2, N2, O2, F2, Cl2,
He, Ne, Ar, Kr, Xe, Rn. 2.76 Unreactive. He, Ne, and Ar are
chemically inert. 2.78 Ra is a radioactive decay product of
U-238. 2.80 Argentina. 2.82 (a) NaH, sodium hydride,
(b) B2O3, diboron trioxide, (c) Na2S, sodium sulfide, (d) AlF3,
aluminum fluoride, (e) OF2, oxygen difluoride, (f) SrCl2, strontium chloride.
Chapter 3
3.6 92.51%. 3.8 5.1 3 1024 amu. 3.12 5.82 3 103 light yr.
3.14 9.96 3 10215 mol Co. 3.16 3.01 3 103 g Au. 3.18 (a)
1.244 3 10222 g/As atom. (b) 9.746 3 10223 g/Ni atom.
3.20 2.98 3 1022 Cu atoms. 3.22 Pb. 3.24 (a) 73.89 g. (b)
76.15 g. (c) 119.37 g. (d) 176.12 g. (e) 101.11 g. (f) 100.95
g. 3.26 6.69 3 1021 C2H6 molecules. 3.28 N: 3.37 3 1026
atoms; C: 1.69 3 1026 atoms; O: 1.69 3 1026 atoms; H: 6.74 3
1026 atoms. 3.30 8.56 3 1022 molecules. 3.34 7. 3.40 C:
10.06%; H: 0.8442%; Cl: 89.07%. 3.42 NH3. 3.44 C2H3NO5.
3.46 39.3 g S. 3.48 5.97 g F. 3.50 (a) CH2O. (b) KCN.
3.52 C5H8O4NNa. 3.60 (a) 2KClO3 88n 2KCl 1 3O2; (b)
2KNO3 88n 2KNO2 1 O2; (c) NH4NO3 88n N2O 1 2H2O;
(d) NH4NO2 88n N2 1 2H2O; (e) 2NaHCO3 88n Na2CO3 1
H2O 1 CO2; (f) P4O10 1 6H2O 88n 4H3PO4; (g) 2HCl 1
CaCO3 88n CaCl2 1 H2O 1 CO2; (h) 2Al 1 3H2SO4 88n
Al2(SO4)3 1 3H2; (i) CO2 1 2KOH 88n K2CO3 1 H2O; (j)
CH4 1 2O2 88n CO2 1 2H2O; (k) Be2C 1 4H2O 88n
2Be(OH)2 1 CH4; (l) 3Cu 1 8HNO3 88n 3Cu(NO3)2 1 2NO 1
4H2O; (m) S 1 6HNO3 88n H2SO4 1 6NO2 1 2H2O; (n) 2NH3
1 3CuO 88n 3Cu 1 N2 1 3H2O. 3.64 1.01 mol Cl2. 3.66 78.3 g
NaHCO3. 3.68 0.324 L. 3.70 0.294 mol KCN. 3.72 20 g
N2O. 3.74 18.0 g O2. 3.78 0.709 g NO2; 6.9 3 1023 mol
NO. 3.80 HCl; 23.4 g. 3.84 (a) 7.05 g. (b) 92.9%. 3.86 3.47 3
103 g. 3.88 Cl2O7. 3.90 (a) 0.212 mol. (b) 0.424 mol.
3.92 18. 3.94 2.4 3 1023 atoms. 3.96 65.4 amu. Zn.
3.98 89.6%. 3.100 C6H12O6. 3.102 Cr. 3.104 1.6 3 104 g/mol.
3.106 NaCl: 32.17%; Na2SO4: 20.09%; NaNO3:
47.75%. 3.108 Ca: 38.76%; P: 19.97%; O: 41.27%. 3.110 Yes.
3.112 2.01 3 1021 molecules. 3.114 16.00 amu. 3.116 (e).
3.118 PtCl2; PtCl4. 3.120 (a) Mn3O4. (b) 3MnO2 88n Mn3O4
A-26
Back
Forward
Main Menu
TOC
Study Guide TOC
Textbook Website
MHHE Website
ANSWERS TO EVEN-NUMBERED PROBLEMS
1 O2. 3.122 6.1 3 105 tons. 3.124 Mg3N2 (magnesium nitride).
3.126 PbC8H20. 3.128 (a) 4.3 3 1022 atoms. (b) 1.6 3 102 pm.
3.130 28.97 g/mol. 3.132 3.1 3 1023 molecules/mol. 3.134 9.09
3 102 kg.
Chapter 4
4.8 (a) Strong electrolyte. (b) Nonelectrolyte. (c) Weak electrolyte.
(d) Strong electrolyte. 4.10 (b) and (c). 4.12 HCl does not ionize in benzene. 4.16 (a) Insoluble, (b) soluble, (c) soluble, (d)
insoluble, (e) soluble. 4.20 (a) Add chloride ions, (b) add sulfate
ions, (c) add carbonate ions, (d) add sulfate ions. 4.28 (a)
Brnsted base. (b) Brnsted base. (c) Brnsted acid.
(d) Brnsted base and Brnsted acid. 4.30 (a) CH3COOH 1 K1
1 OH2 88n K1 1 CH3COO2 1 H2O; CH3COOH 1
OH2 88n CH3COO2 1 H2O. (b) H2CO3 1 2Na1 1
2
2OH2 88n 2Na1 1 CO22
3 1 2H2O; H2CO3 1 2OH 88n
22
1
2
21
2
CO3 1 2H2O. (c) 2H 1 2NO3 1 Ba 1 2OH 88n Ba21 1
1
2
2NO2
3 1 2H2O; 2H 1 2OH 88n 2H2O. 4.40 (a) Fe 88n
Fe31 1 3e2; O2 1 4e2 88n 2O22. Oxidizing agent: O2; reducing agent: Fe. (b) 2Br2 88n Br2 1 2e2; Cl2 1 2e2 88n 2Cl2.
Oxidizing agent: Cl2; reducing agent: Br2. (c) Si 88n
Si41 1 4e2; F2 1 2e2 88n 2F2. Oxidizing agent: F2; reducing
agent: Si. (d) H2 88n 2H1 1 2e2; Cl2 1 2e2 88n 2Cl2.
Oxidizing agent: Cl2; reducing agent: H2. 4.42 (a) 15. (b) 11.
(c) 13. (d) 15. (e) 15. (f) 15. 4.44 All are zero. 4.46 (a) 23.
(b) 2}21}. (c) 21. (d) 14. (e) 13. (f) 22. (g) 13. (h) 16. 4.48 Li
and Ca. 4.50 (a) No reaction. (b) No reaction. (c) Mg 1
CuSO4 88n MgSO4 1 Cu. (d) Cl2 1 2KBr 88n Br2 1 2KCl.
4.52 (a) Combination. (b) Decomposition. (c) Displacement.
(d) Disproportionation. 4.56 0.0618 M. 4.58 10.8 g. 4.60
(a) 1.37 M. (b) 0.426 M. (c) 0.716 M. 4.62 (a) 6.50 g. (b) 2.45 g.
(c) 2.65 g. (d) 7.36 g. (e) 3.95 g. 4.66 0.0433 M. 4.68 126 mL.
4.70 1.09 M. 4.74 35.72%. 4.76 2.31 3 1024 M. 4.80 (a) 6.0 mL.
(b) 8.0 mL. 4.84 9.43 3 1023 g. 4.86 0.06020 M. 4.88 5.40%.
4.90 0.231 mg. 4.92 (i) Only oxygen supports combustion. (ii)
Only CO2 reacts with Ca(OH)2 to form CaCO3 (white precipitate). 4.94 1.26 M. 4.96 0.171 M. 4.98 0.115 M. 4.100 Ag:
1.25 g; Zn: 2.12 g. 4.102 0.0722 M NaOH. 4.104 24.0 g/mol;
Mg. 4.106 1.73 M. 4.108 Only Fe(II) is oxidized by KMnO4 solution and can therefore change the purple color to colorless.
4.110 Ions are removed as the BaSO4 precipitate. 4.112 (i)
Conductivity test. (b) Only NaCl reacts with AgNO3 to form
AgCl precipitate. 4.114 The Cl2 ion cannot accept any electrons. 4.116 Reaction is too violent. 4.118 Use sodium bicar1
bonate: HCO2
3 1 H 88n H2O 1 CO2. NaOH is a cautic substance and unsafe to use in this manner. 4.120 (a) Conductivity.
Reaction with AgNO3 to form AgCl. (b) Soluble in water.
Nonelectrolyte. (c) Possesses properties of acids. (d) Soluble.
Reacts with acids to give CO2. (e) Soluble, strong electrolyte.
Reacts with acids to give CO2. (f) Weak electrolyte and weak
acid. (g) Soluble in water. Reacts with NaOH to produce
Mg(OH)2 precipitate. (h) Strong electrolyte and strong base.
Back
Forward
Main Menu
TOC
A-27
(i) Characteristic odor. Weak electrolyte and weak base.
(j) Insoluble. Reacts with acids. (k) Insoluble. Reacts with
acids to produce CO2. 4.122 NaCl: 43.97%; KCl: 56.03%.
4.124 1.33 g. 4.126 55.80%. 4.128 (a) 1.40 M. (b) 4.96 g.
4.130 (a) Precipitation: Mg21 1 2OH2 88n Mg(OH)2;
acid-base: Mg(OH)2 1 2HCl 88n MgCl2 1 2H2O; redox:
MgCl2 88n Mg 1 Cl2. (b) NaOH is more expensive than CaO.
(c) Dolomite provides additional Mg. 4.132 97.99%. 4.134
Zero. 4.136 0.224%. 4.138 (a) Zn 1 H2SO4 88n ZnSO4 + H2.
(b) 2KClO3 88n 2KCl 1 3O2. (c) Na2CO3 1 2HCl 88n
2NaCl 1 CO2 1 H2O. (d) NH4NO2 88n N2 1 2H2O.
4.140 Vertical: Chromium.
Chapter 5
5.14 0.797 atm; 80.8 kPa. 5.18 53 atm. 5.20 (a) 0.69 L. (b) 61
atm. 5.22 1.3 3 102 K. 5.24 ClF3. 5.30 6.2 atm. 5.32 745 K.
5.34 1.9 atm. 5.36 0.82 L. 5.38 33.6 mL. 5.40 6.1 3 1023 atm.
5.42 35.0 g/mol. 5.44 N2: 2.1 3 1022; O2: 5.7 3 1021; Ar: 3 3
1020. 5.46 2.98 g/L. 5.48 SF4. 5.50 M 1 3HCl 88n 1.5H2 1
MCl3. M2O3; M2(SO4)3. 5.52 2.84 3 1022 mol CO2; 94.7%.
5.54 1.71 3 103 L. 5.56 N2: 0.80; O2: 0.20. 5.58 (a) 0.89 atm.
(b) 1.4 L. 5.60 349 mmHg. 5.62 19.8 g. 5.64 H2: 650 mmHg;
N2: 217 mmHg. 5.72 N2: 472 m/s; O2: 441 m/s; O3: 360 m/s.
5.74 2.8 m/s. 5.80 No. 5.82 Ne. 5.84 C6H6. 5.86 445 mL.
5.88 (a) 9.53 atm. (b) Ni(CO)4 decomposes to give CO, which
increases the pressure. 5.90 1.30 3 1022 molecules; CO2, O2,
N2, H2O. 5.92 5.25 3 1018 kg. 5.94 0.0701 M. 5.96 He:
0.16 atm; Ne: 2.0 atm. 5.98 HCl dissolves in the water, creating a partial vacuum. 5.100 7. 5.102 (a) 61.2 m/s. (b) 4.58 3
1024 s. (c) 366.1 m/s. 5.104 1.58 3 104 L. 5.106 Higher partial
pressure inside a paper bag. 5.108 To equalize the pressure as
the amount of ink decreases. 5.110 0.0821 L ? atm/K ? mol.
5.112 C6H6. 5.114 The low atmospheric pressure caused the
harmful gases (CO, CO2, CH4) to flow out of the mine, and the
man suffocated. 5.116 (a) 4.90 L. (b) 6.0 atm. (c) 1 atm.
5.118 (a) 5 3 10222 atm. (b) 5 3 1020 L. 5.120 91%.
5.122 1.7 3 1012 molecules. 5.124 NO2. 5.126 3.5 3 10230 J.
5.128 2.3 3 103 L. 5.130 1.8 3 102 mL. 5.132 (a) 1.09 3 1044
molecules. (b) 1.18 3 1022 molecules/ breath. (c) 2.60 3 1030
molecules. (d) 2.39 3 10214; 3 3 108 molecules. (e) Complete
mixing of air; no molecules escaped to the outer atmosphere;
no molecules used up during metabolism, nitrogen fixation, etc.
Chapter 6
6.18 728 kJ. 6.20 50.7C. 6.22 25.03C. 6.30 O2. 6.32 (a)
Br2(g): . 0, Br2(l): 5 0; (b) I2(g): . 0, I2(s): 5 0.
6.34 Measure DH for the formation of Ag2O from Ag and O2
and of CaCl2 from Ca and Cl2. 6.36 (a) 2167.2 kJ/mol.
(b) 256.2 kJ. 6.38 (a) 21411 kJ. (b) 21124 kJ. 6.40 218.2
kJ/mol. 6.42 24.51 kJ/g. 6.44 2.70 3 102 kJ. 6.46 284.6 kJ.
6.48 2780 kJ. 6.60 48 J. 6.62 23.1 kJ. 6.64 DH2 2 DH1.
Study Guide TOC
Textbook Website
MHHE Website
A-28
ANSWERS TO EVEN-NUMBERED PROBLEMS
6.66 (a) 2336.5 kJ. (b) NH3. 6.68 2553.8 kJ. 6.70 0.
6.72 2366.5 kJ. 6.74 0.492 J/g ? C. 6.76 The first reaction
(exothermic) can be used to promote the second reaction (endothermic). 6.78 1.09 3 104 L. 6.80 4.10 L. 6.82 5.60 kJ/mol.
6.84 (a). 6.86 (a) 0. (b) 29.1 J. (c) 248 J. 6.88 (a) A more
fully packed freezer has a greater mass and hence a large heat
capacity. (b) Tea or coffee has a greater amount of water,
which has a higher specific heat than noodles. 6.90 21.84 3
103 kJ. 6.92 3.0 3 109. 6.94 5.35 3 103 J/C. 6.96 25.2 3
106 kJ. 6.98 (a) 3.4 3 105 g. (b) 22.0 3 105 kJ. 6.100 (a) 1.4
3 102 kJ. (b) 3.9 3 102 kJ. 6.102 (a) 265.2 kJ. (b) 29.0
kJ. 6.104 2110.5 kJ/mol. It will form both CO and CO2.
6.106 (a) 0.50 J. (b) 32 m/s. (c) 0.12C. 6.108 2277.0 kJ/mol.
6.110 104 g. 6.112 304C. 6.114 1.51 3 103 kJ.
Chapter 7
7.8 (a) 6.58 3 1014 /s. (b) 1.22 3 108 nm. 7.10 1.5 3 102 s.
7.12 4.95 3 1014/s. 7.16 (a) 4.0 3 102 nm. (b) 5.0 3 10219 J.
7.18 1.2 3 102 nm (UV). 7.20 (a) 3.70 3 102 nm. (b) UV.
(c) 5.38 3 10219 J. 7.26 Use a prism. 7.28 Compare the emission spectra with those on Earth of known elements.
7.30 3.027 3 10219 J. 7.32 6.17 3 1014/s. 4.86 3 102 nm.
7.34 5. 7.40 1.37 3 1026 nm. 7.42 1.7 3 10223 nm. 7.54 l 5 2:
ml 5 22, 21, 0, 1, 2. l 5 1: ml 5 21, 0, 1. l 5 0: ml 5 0.
7.56 (a) n 5 4, l 5 1, ml 5 21, 0, 1. (b) n 5 3, l 5 2, ml 5 22,
21, 0, 1, 2. (c) n 5 3, l 5 0, ml 5 0. (d) n 5 5, l 5 3, ml 5 23,
22, 21, 0, 1, 2, 3. 7.58 Differ in orientation only. 7.60 6s, 6p,
6d, 6f, 6g, and 6h. 7.62 2n2. 7.64 (a) 3. (b) 6. (c) 0. 7.66 There
is no shielding in a H atom. 7.68 (a) 2s , 2p. (b) 3p , 3d. (c)
3s , 4s. (d) 4d , 5f. 7.80 Al: 1s22s22p63s23p1. B: 1s22s22p1. F:
1s22s22p5. 7.82 B(1), Ne(0), P(3), Sc(1), Mn(5), Se(2), Kr(0),
Fe(4), Cd(0), I(1), Pb(2). 7.84 Ge: [Ar]4s23d104p2. Fe:
[Ar]4s23d6. Zn: [Ar]4s23d10. Ni: [Ar]4s23d8. W: [Xe]6s24f 145d 4.
TI: [Xe]6s24f 145d 106p1. 7.86 S1. 7.92 [Kr]5s24d5. 7.94 (a)
Incorrect. (b) Correct. (c) Incorrect. 7.96 (a) An e in a 2s and an
e in each 2p orbital. (b) 2 e each in a 4p, a 4d, and a 4f orbital.
(c) 2 e in each of the 5 3d orbitals. (d) An e in a 2s orbital. (e) 2
e in a f orbital. 7.98 Wave properties. 7.100 (a) 8.76 3 10226
nm. (b) 7.38 nm. 7.102 (a) 1.20 3 1018 photons. (b) 3.76 3 108
W. 7.104 419 nm. In principle, yes; in practice, no. 7.106 3.0 3
1019 photons. 7.108 He1: 164 nm, 121 nm, 109 nm, 103 nm (all
in the UV region). H: 657 nm, 487 nm, 434 nm, 411 nm (all in
the visible region). 7.110 1.2 3 102 photons. 7.112 Yellow light
will generate more electrons; blue light will generate electrons
with greater kinetic energy.
7.114 (a) He. (b) N. (c) Na. (d) As. (e) Cl. 7.116 They might
have discovered the wave properties of electrons. 7.118 7.39 3
1022 nm. 7.120 (a) False, (b) false, (c) true, (d) false,
(e) true. 7.122 2.0 3 1025 m/s. 7.124 (a) and (f) violate Pauli
exclusion principle; (b), (d), and (e) violate Hunds rule.
7.126 2.8 3 106 K.
Back
Forward
Main Menu
TOC
Chapter 8
8.20 (a) 1s22s22p63s23p5. (b) Representative. (c) Paramagnetic.
8.22 (a) and (d); (b) and (e); (c) and (f). 8.24 (a) Group 1A, (b)
Group 5A, (c) Group 8A, (d) Group 8B. 8.26 Fe31. 8.28 (a)
[Ne], (b) [Ne], (c) [Ar], (d) [Ar], (e) [Ar], (f) [Ar]3d 6, (g)
[Ar]3d 9, (h) [Ar]3d10. 8.30 (a) Cr31, (b) Sc31, (c) Rh31, (d)
Ir31. 8.32 Be21 and He; F2 and N32; Fe21 and Co31; S22 and
Ar. 8.38 Na . Mg . Al . P . Cl. 8.40 F. 8.42 The effective
nuclear charge of the outermost electrons increases across the
period. 8.44 Mg21 , Na1 , F2 , O22 , N32. 8.46 Te22.
8.48 2199.4C. 8.52 The single 3p electron in Al is well
shielded by the 1s, 2s, and 3s electrons. 8.54 1s22s22p6: 2080
kJ/mol. 8.56 8.43 3 106 kJ/mol. 8.60 Cl. 8.62 The ns1 configuration allows them to accept another electron. 8.66 Fr should
be the most reactive toward water and oxygen, forming FrOH
and Fr2O2 and FrO2. 8.68 The Group 1B elements have higher
ionization energies due to the incomplete shielding of the inner
d electrons. 8.70 (a) Li2O 1 H2O 88n 2LiOH. (b) CaO 1
H2O 88n Ca(OH)2. (c) SO3 1 H2O 88n H2SO4. 8.72 BaO.
8.74 (a) Bromine, (b) nitrogen, (c) rubidium, (d) magnesium.
8.76 (a) Mg21 , Na1 , F2 , O22. (b) O22 , F2 , Na1 ,
Mg21. 8.78 M is K; X is Br. 8.80 N and O1; Ne and N32; Ar
and S22; Zn and As31; Cs1 and Xe. 8.82 (a) and (d). 8.84
Yellow-green gas: F2; yellow gas: Cl2; red liquid: Br2; dark
solid: I2. 8.86 Fluorine. 8.88 H2. 8.90 Li2O (basic); BeO (amphoteric); B2O3 (acidic); CO2 (acidic); N2O5 (acidic). 8.92 It
forms both the H1 and H2 ions; H1 is a single proton. 8.94
0.65. 8.96 76.7%. 8.98 418 kJ/mol. 8.100 7.28 3 103 kJ/mol.
8.102 X: Sn or Pb; Y: P; Z: alkali metal. 8.104 495.9 kJ/mol.
8.106 343 nm. 8.108 604.3 kJ. 8.110 K2TiO4. 8.112 2K2MnF6
1 4SbF5 88n 4KSbF6 1 2MnF3 1 F2. 8.114 N2O (11), NO
(12), N2O3 (13), NO2 and N2O4 (14), N2O5 (15). 8.116 The
larger the effective nuclear charge, the more tightly held are the
electrons. The atomic radius will be small and the ionization
energy will be large. 8.118 M.pt.: 6.3C; b.pt.: 74.9C. 8.120
The heat generated from nuclear decay can decompose compounds. 8.122 Ar: 39.95 amu; K: 39.10 amu. 8.124 Z 5 119;
[Rn]7s25f 146d107p68s1. 8.126 Group 2A. 8.128 (a) SiH4, GeH4,
SnH4, PbH4. (b) RbH more ionic. (c) Ra 1 2H2O 88n
Ra(OH)2 1 H2. (d) Be.
Chapter 9
9.16 (a) RbI, (b) Cs2SO4, (c) Sr3N2, (d) Al2S3.
M
M
9.18 (a)NSrN 1 NSeN88n Sr21SSeS22
M
M
(b)
(c)
(d)
.
9.20 (a) BF3, covalent; KBr, ionic. 9.26 2195 kJ/mol.
Study Guide TOC
Textbook Website
MHHE Website
A-29
ANSWERS TO EVEN-NUMBERED PROBLEMS
9.36 COH , BrOH , NaOI , FOH , LiOCl , KOF.
9.38 ClOCl , BrOCl , SiOC , CsOF. 9.40 (a) Covalent,
(b) polar covalent, (c) ionic, (d) polar covalent.
9.44 (a)
(d)
, (b)
, (c)
dinate covalent bonds. 9.118 347 kJ/mol. 9.120 (a)
(c) 21.2 3 106 kJ.
(b)
9.46
Chapter 10
10.8 (a) Trigonal planar, (b) linear, (c) tetrahedral. 10.10 (a)
Tetrahedral, (b) bent, (c) trigonal planar, (d) linear, (e) square
planar, (f) tetrahedral, (g) trigonal bipyramid, (h) trigonal pyra2
mid, (i) tetrahedral. 10.12 SiCl4, CI4, CdCl24 . 10.18 Electronegativity decreases from F to I. 10.20 Higher. 10.22 (b) 5 (d)
, (c) , (a). 10.32 sp3. 10.34 B: sp2 to sp3; N: remains sp3.
10.36 (a) sp3, (b) sp3, sp2, sp2, (c) sp3, sp, sp, sp3, (d) sp3, sp2,
(e) sp3, sp2. 10.38 sp. 10.40 sp3d. 10.42 9 pi bonds and 9
sigma bonds. 10.48 Electron spins must be paired in H2. 10.50
1
1
Li2
2 5 Li2 , Li2. 10.52 B2 . 10.54 MO theory predicts O2 is
22
1
paramagnetic. 10.56 O2 , O2
2 , O2 , O2 . 10.58 B2 contains
a pi bond; C2 contains 2 pi bonds. 10.62 The circle shows elec-
9.50
9.52
9.54
9.60
9.62
The
octet rule is not obeyed. 9.64 Coordinate covalent bond.
9.68 303.0 kJ/mol. 9.70 (a) 22759 kJ. (b) 22855 kJ.
9.74 Ionic: NaF, MgF2, AlF3; Covalent: SiF4, PF5, SF6, ClF3.
9.76 KF: ionic, high melting point, soluble in water, its melt
and solution conduct electricity. C6H6: covalent and discrete
molecule, low melting point, insoluble in water, does not conduct electricity.
9.78
32
9.80 (a) AlCl2
4 , (b) AlF6 , (c) AlCl3. 9.82 CF2: violates the
octet rule; LiO2: lattice energy too low; CsCl2: second ionization too high to produce Cs21; PI5:I atom too bulky to fit
around P. 9.84 (a) False, (b) true, (c) false, (d) false. 9.86 267
kJ/mol. 9.88 N2. 9.90 NH1
4 and CH4; CO and N2; B3N3H6 and
C6H6. 9.92
9.94 F2
3 violates the octet rule.
9.96
9.98 (c) No bond between C and O, (d) large formal charges.
9.102 (a) 29.2 kJ. (b) 29.2 kJ. 9.104 (a)
(b)
(c)
(d)
9.106 Yes. 9.108 (a)
114 kJ. (b) Extra electron increases repulsion between F
atoms. 9.110 Lone pair on C and negative formal charge on C.
9.112 (a)
. (b) No. 9.114 Violates the
octet rule. 9.116
Back
Forward
The arrows indicate coor-
Main Menu
TOC
tron delocalization. 10.64 (a)
(b) sp2. (c) N forms sigma bonds with F and O atoms. There is
a pi molecular orbital delocalized over N and O atoms.
10.66 sp2. 10.68 Linear. 10.70 The large size of Si results in
poor sideways overlap of p orbitals to form pi bonds.
1
2
10.72 XeF1
3 : T-shaped; XeF5 : square pyramid; SbF6 : octahedral. 10.74 (a) 180, (b) 120, (c) 109.5, (d) about 109.5, (e)
180, (f) about 120, (g) about 109.5, (h) 109.5. 10.76 sp3d.
2
10.78 ICl2
2 and CdBr2. 10.80 sp . Molecule on the right.
10.82 The pi bond in cis-dichloroethylene prevents rotation.
10.84 O3, CO, CO2, NO2, N2O, CH4, CFCl3. 10.86 C: all single-bonded C atoms are sp3, the double-bonded C atoms are
sp2; N: single-bonded N atoms are sp3, N atoms that form one
double bond are sp2, N atom that forms two double bonds is
sp. 10.88 Si has 3d orbitals so water can add to Si (valence
shell expansion). 10.90 C: sp2; N: N atom that forms a double
bond is sp2, the others are sp3. 10.92 (a) Use a conventional
oven. (b) No. Polar molecules would absorb microwaves. (c)
Water molecules absorb part of microwaves. 10.94 The small size
of F results in a shorter bond and greater lone pair repulsion.
Chapter 11
11.8 Methane. 11.10 (a) Dispersion forces, (b) dispersion and
dipole-dipole forces, (c) same as (b), (d) dispersion and ion-ion
forces, (e) same as (a). 11.12 (e). 11.14 Only 1-butanol can
form hydrogen bonds. 11.16 (a) Xe. (b) CS2. (c) Cl2. (d) LiF.
(e) NH3. 11.18 (a) Hydrogen bond, dipole-dipole, and dispersion forces, (b) dispersion forces, (c) dispersion forces, (d) covalent bond. 11.20 The compound on the left can form in-
Study Guide TOC
Textbook Website
MHHE Website
A-30
ANSWERS TO EVEN-NUMBERED PROBLEMS
tramolecular hydrogen bond. 11.32 Between ethanol and glycerol. 11.38 scc: 1; bcc: 2; fcc: 4. 11.40 6.17 3 1023/mol.
11.42 458 pm. 11.44 XY3. 11.48 0.220 nm. 11.56 Each C atom
in diamond is covalently bonded to 4 other C atoms. Graphite
has delocalized electrons in extensive two dimensions.
11.78 2.67 3 103 kJ. 11.80 47.03 kJ/mol. 11.82 Freezing, sublimation. 11.84 When steam condenses at 100C, it releases
heat equal to heat of vaporization. 11.86 331 mmHg. 11.88
75.9 kJ/mol. 11.92 Initially ice melts because of the increase in
pressure. As the wire sinks into the ice, the water above it refreezes. In this way, the wire moves through the ice without
cutting it in half. 11.94 (a) Ice melts. (b) Water vapor condenses to ice. (c) Water boils. 11.96 (d) 11.98 Covalent crystal.
11.100 CCl4. 11.102 760 mmHg. 11.104 It is the critical point.
11.106 Crystalline SiO2. 11.108 (a), (b), (d). 11.110 8.3 3
1023 atm. 11.112 (a) K2S. (b) Br2. 11.114 SO2. 11.116 30.7
kJ/mol. 11.118 Smaller ions have larger charge densities and a
greater extent of hydration. 11.120 (a) 30.7 kJ. (b) 192.5 kJ.
11.122 (a) Decreases, (b) no change, (c) no change.
11.124 CaCO3(s) 88n CaO(s) 1 CO2(g). Three phases.
11.126 SiO2 is a covalent crystal. CO2 exists as discrete molecules. 11.128 66.8%. 11.130 scc: 52.4%; bcc: 68.0%; fcc: 74.0%.
11.132 1.69 g/cm3. 11.134 (a) Two (diamond/graphite/liquid
and graphite/liquid/vapor). (b) Diamond. (c) Apply high pressure at high temperature.
Chapter 12
12.10 Cyclohexane cannot form hydrogen bonds. 12.12 The
longer chains become more nonpolar. 12.16 (a) 25.9 g. (b) 1.72
3 103 g. 12.18 (a) 2.68 m. (b) 7.82 m. 12.20 0.010 m.
12.22 5.0 3 102 m. 18.3 M. 12.24 (a) 2.41 m. (b) 2.13 M.
(c) 0.0587 L. 12.28 45.9 g. 12.36 CO2 pressure is greater at the
bottom of the mine. 12.38 0.28 L. 12.52 1.3 3 103 g.
12.54 Eathanol: 30.0 mmHg; 1-propanol: 26.3 mmHg.
12.56 128 g. 12.58 0.59 m. 12.60 120 g/mol. C4H8O4.
12.62 28.6C. 12.64 4.3 3 102 g/mol. C24H20P4. 12.66 1.75 3
104 g/mol. 12.68 343 g/mol. 12.74 Boiling point, vapor-pressure, osmotic pressure. 12.76 0.50 m glucose . 0.50 m acetic
acid . 0.50 m HCl. 12.78 0.942 m. 12.80 7.6 atm. 12.82 1.6
atm. 12.86 3.5 atm. 12.88 (a) 104 mmHg. (b) 116 mmHg.
12.90 2.95 3 103 g/mol. 12.92 No. 12.94 AlCl3 dissociates into
Al31 and 3 Cl2 ions. 12.96 NaCl: 143.8 g; MgCl2: 5.14 g;
Na2SO4: 7.25 g; CaCl2: 1.11 g; KCl: 0.67 g; NaHCO3: 0.17
g. 12.98 The molar mass in B is twice as large as that in A. A
dimerization process. 12.100 (a) Last alcohol, (b) methanol, (c)
last alcohol. 12.102 I2-water: weak dipole-induced dipole; I2
3water: favorable ion-dipole interaction. 12.104 (a) Same NaCl
solution on both side, (b) only water would move from left to
right, (c) normal osmosis. 12.106 12.3 M. 12.108 14.2%.
12.110 (a) Decreases with lattice energy, (b) increases with polarity of solvent, (c) increases with enthalpy of hydration.
12.112 1.80 g/mL. 5.0 3 102 m. 12.114 0.815. 12.116 NH3 can
form hydrogen bonds with water. 12.118 2.8%. 12.120 1.2 3
102 g/mol. It forms a dimer in benzene.
Back
Forward
Main Menu
TOC
Chapter 13
1 D[H2]
D[O2]
1 D[H2O]
13.6 (a) rate 5 2}} }}
5 2}}
5 }} }}
(b) rate 5
2 Dt
Dt
2
Dt
1 D[NH3]
1 D[O2]
1 D[NO]
1 D[H2O]
5 2}} }}
5 }} }} 5 }} }}
13.8 (a)
2}} }}
4
Dt
5 Dt
4 Dt
6
Dt
0.049 M/s. (b) 20.025 M/s. 13.16 rate 5 k[F2][ClO2]. 2.4 3 1024
M/s. 13.18 (a) rate 5 k[X]2[Y]. (b) 0.38 M/s. 13.20 (a) 0.046 s21.
(b) 0.13 M21s21. 13.22 First order. 13.28 (a) 0.0198 s21. (b) 151 s.
13.30 3.6 s. 13.38 135 kJ/mol. 13.40 641 K. 13.42 51.0
kJ/mol. 13.50 (a) rate 5 k[X2][Y]. (b) Reaction is zero order in
Z. (c) X2 1 Y 88n XY 1 X (slow). X 1 Z 88n XZ (fast).
k k2
13.62 Temp13.52 Mechanism I. 13.60 rate 5 }1}[E][S].
k21
erature, energy of activation, concentration of reactants, catalyst. 13.64 22.6 cm2; 44.9 cm2. The large surface area of grain
dust can result in a violent explosion. 13.66 (a) rate 5
k[H2][NO]2. (b) 0.38 M22s21. (c) H2 1 2NO 88n N2 1 H2O
1 O (slow). O 1 H2 88n H2O (fast). 13.68 Water is present in
excess so its concentration does not change appreciably.
13.70 10.7 M21s21. 13.72 2.63 atm. 13.74 M22s21. 13.76 56.4
min. 13.78 (b), (d), (e). 13.80 0.098%. 13.82 (a) Increase,
(b) decrease, (c) decrease, (d) increase. 13.84 0.0896 min21.
13.86 1.12 3 103 min. 13.88 (a) I2 absorbs visible light to form
I atoms. (b) UV light is needed to dissociate H2. 13.90 (a) rate
5 k[X][Y]2. (b) 1.9 3 1022 M22s21. 13.92 Second order. 2.4
3 107 M21s21. 13.94 Because the engine is relatively cold so
the exhaust gases will not fully react with the catalytic converter. 13.96 (a) Reaction has a large activation energy,
(b) Activation energy is very small and orientation factor not
important. 13.98 3.9 3 105 yr. 13.100 (a) Mn21. (b) Without
the catalyst, reaction would be termolecular. (c) Homogeneous.
13.102 0.45 atm. 13.104 (a) k1[A] 2 k2[B]. (b) [B] 5
(k1/k2)[A]. 13.106 rate 5 k[surface area of Mg][H1]2.
(b) 0.32%.
Chapter 14
14.14 1.08 3 107. 14.16 3.5 3 1027. 14.18 (a) 0.082. (b) 0.29.
14.20 0.105. 2.05 3 1023. 14.22 7.09 3 1023. 14.24 3.3
14.26 3.56 3 1022. 14.28 4.0 3 1026. 14.30 5.6 3 1023.
14.34 0.64 M22s21. 14.38 [NH3] will increase and [N2] and
[H2] will decrease. 14.40 NO: 0.50 atm; NO2: 0.020 atm.
14.42 [I] 5 8.58 3 1024 M; [I2] 5 0.0194 M. 14.44 (a) 0.52.
(b) [CO2] 5 0.48 M, [H2] 5 0.020 M, [CO] 5 0.075 M, [H2O] 5
0.065 M. 14.46 [H2] 5 [CO2] 5 0.05 M, [H2O] 5 [CO] 5 0.11 M.
14.52 (a) Shift position of equilibrium to the right, (b) no effect, (c) no effect. 14.54 (a) No effect, (b) no effect, (c) shift
the position of equilibrium to the left, (d) no effect, (e) to the
left. 14.56 (a) To the right, (b) to the left, (c) to the right, (d) to
the left, (e) no effect. 14.58 No change. 14.60 (a) More CO2
will form. (b) No change. (c) No change. (d) Some CO2 will
combine with CaO to form CaCO3. (e) Some CO2 will react
with NaOH so equilibrium will shift to the right. (f) HCl reacts
with CaCO3 to produce CO2. Equilibrium will shift to the left.
Study Guide TOC
Textbook Website
MHHE Website
ANSWERS TO EVEN-NUMBERED PROBLEMS
(g) Equilibrium will shift to the right. 14.62 (a) NO: 0.24 atm;
Cl2: 0.12 atm. (b) 0.017. 14.64 (a) No effect, (b) more CO2
and H2O will form. 14.66 8 3 10244. The reaction has a very
large activation energy. 14.68 (a) 1.7. (b) A: 0.69 atm, B: 0.81
atm. 14.70 1.5 3 105. 14.72 H2: 0.28 atm, Cl2: 0.049 atm, HCl:
1.67 atm. 14.74 5.0 3 101 atm. 14.76 3.86 3 1022. 14.78 3.13.
14.80 N2: 0.860 atm, H2: 0.366 atm, NH3: 4.40 3 1023 atm.
14.82 (a) 1.16. (b) 53.7%. 14.84 (a) 0.49 atm. (b) 0.23, (c)
0.037, (d) greater than 0.037 mol. 14.86 [H2] 5 0.070 M, [I2]
5 0.182 M, [HI] 5 0.825 M. 14.88 (c). 14.90 (a) 4.2 3 1024.
(b) 0.83. (c) 1.1. (d) In (b): 2.3 3 103; in (c): 0.021. 14.92 0.0231;
9.6 3 1024. 14.94 NO2: 1.2 atm; N2O4: 0.12 atm. Kp 5 12.
14.96 (a) The equilibrium will shift to the right, (b) to the
right, (c) no change, (d) no change, (e) no change, (f) to the
left. 14.98 NO2: 0.10 atm; N2O4: 0.088 atm. 14.100 (a) 1.03
atm. (b) 0.39 atm. (c) 1.67 atm. (d) 62.0%. 14.102 22 mg/m3.
Chapter 15
2
2
2
2
15.4 (a) NO2
2 , (b) HSO4 , (c) HS , (d) CN , (e) HCOO .
2
15.6 (a) H2S, (b) H2CO3, (c) HCO2
3 , (d) H3PO4, (e) H2PO4 , (f)
22
2
2
HPO4 , (g) H2SO4, (h) HSO4 , (i) HSO3 . 15.8 (a) CH2ClCOO2,
32
2
22
2
22
(b) IO2
4 , (c) H2PO4 , (d) HPO4 , (e) PO 4 , (f) HSO4 , (g) SO4 ,
2
22
2
2
22
(h) IO3 , (i) SO3 , (j) NH3, (k) HS , (l) S , (m) OCl . 15.16 1.6
3 10214 M. 15.18 (a) 10.74. (b) 3.28. 15.20 (a) 6.3 3 1026 M.
(b) 1.0 3 10216 M. (c) 2.7 3 1026 M. 15.22 (a) Acidic, (b) neutral, (c) basic. 15.24 0.444. 15.26 0.118. 15.32 (a) Strong,
(b) weak, (c) weak, (d) weak, (e) strong. 15.34 (b) and (c). 15.36
No. 15.42 [H1] 5 [CH3COO2] 5 5.8 3 1024 M, [CH3COOH] 5
0.0181 M. 15.44 2.3 3 1023 M. 15.46 (a) 3.5%, (b) 33%,
(c) 79%. 15.48 (a) 3.9%. (b) 0.30%. 15.52 7.1 3 1027. 15.54
1.5%. 15.60 HCl: 1.40; H2SO4: 1.31. 15.62 [H1] 5 [HCO2
3 ] 5 1.0
211
3 1024 M, [CO22
M. 15.66 (a) H2SO4 . H2SeO4,
3 ] 5 4.8 3 10
(b) H3PO4 . H3AsO4. 15.68 The conjugate base of phenol can be
stabilized by resonance. 15.76 HZ , HY , HX. 15.78 4.82. 15.80
Basic. 15.84 (a) Al2O3 , BaO , K2O, (b) CrO3 , Cr2O3 , CrO.
15.86 Al(OH)3 1 OH2 88n Al(OH)2
4 . Lewis acid-base reaction.
15.90 AlCl3 is the Lewis acid, Cl2 is the Lewis base. 15.92 CO2
and BF3. 15.94 0.106 L. 15.96 No. 15.98 No. 15.100 CrO is basic
and CrO3 is acidic. 15.102 4.0 3 1022. 15.104 7.00. 15.106 NH3.
15.108 (a) 7.43. (b) pD , 7.43. (c) pD 1 pOD 5 14.87. 15.110
1.79. 15.112 F2 reacts with HF to form HF2
2 , thereby shifting
the ionization of HF to the right. 15.114 6.80. 15.116 [H1] 5
22
[H2PO2
4 ] 5 0.0239 M, [H3PO4] 5 0.076 M, [HPO4 ] = 6.2 3
28
32
218
1
10 M, [PO4 ] 5 1.2 3 10
M. 15.118 [Na ] 5 0.200 M,
2
23
[HCO2
M, [H2CO3] 5 2.4 3 1028 M,
3 ] 5 [OH ] 5 4.6 3 10
[H1] 5 2.2 3 10212 M. 15.120 The H1 ions convert CN2 to HCN,
which escapes as a gas. 15.122 0.25 g. 15.124 20.20. 15.126 (a)
Equilibrium will shift to the right, (b) to the left, (c) no effect, (d)
to the right. 15.128 The amines are converted to their salts RNH1
3.
15.130 1.6 3 1024. 15.132 4.40. 15.134 In a basic medium, the
ammonium salt is converted to the pungent-smelling ammonia.
Back
Forward
Main Menu
TOC
A-31
Chapter 16
16.4 (a) 11.28. (b) 9.07. 16.8 (b), (c), (d), (f). 16.10 4.74 for
both. (a) is more effective. 16.12 7.03 16.14 10. More effective
against the acid. 16.16 (a) 4.82. (b) 4.64. 16.18 HC. 16.22 90.1
g/mol. 16.24 0.466 M. 16.26 [H1] 5 3.0 3 10213 M, [OH2] 5
0.0335 M, [Na1] 5 0.0835 M, [CH3COO2] 5 0.0500 M,
[CH3COOH] 5 8.3 3 10210 M. 16.28 8.23. 16.32 CO2 dissolves in water to form H2CO3 which neutralizes NaOH.
16.34 5.70. 16.42 (a) 7.8 3 10210. (b) 1.8 3 10218. 16.44 1.80 3
10210. 16.46 2.2 3 1024 M. 16.48 2.3 3 1029. 16.50 [Na1] 5
21
2
0.045 M, [NO2
3 ] 5 0.076 M, [Sr ] 5 0.016 M, [F ] 5 1.1 3
1024 M. 16.52 pH greater than 2.68 and less than 8.11.
16.56 (a) 0.013 M. (b) 2.2 3 1024 M. (c) 3.3 3 1023 M.
16.58 (a) 1.0 3 1025 M. (b) 1.1 3 10210 M. 16.60 (b), (c), (d),
and (e). 16.62 (a) 0.016 M. (b) 1.6 3 1026 M. 16.64 Yes.
23
16.68 [Cd21] 5 1.1 3 10218 M, [Cd(CN)22
M,
4 ] 5 4.2 3 10
2
25
21
[CN ] 5 0.48 M. 16.70 3.5 3 10 M. 16.72 (a) Cu 1
1
2
2
4NH3 34 Cu(NH3)21
4 . (b) Ag 1 2CN 34 Ag(CN)2 .
(c) Hg21 1 4Cl2 34 HgCl22
.
16.76
0.011
M.
16.78
Use
Cl2
4
ions or flame test. 16.80 From 2.51 to 4.41. 16.82 1.28 M.
16.84 [H1] 5 2.99 3 10213 M, [OH2] 5 0.0335 M,
[CH3COO2] 5 0.0500 M, [CH3COOH] 5 8.3 3 10210 M,
[Na1] 5 0.0835 M. 16.86 9.97 g. pH 5 13.04. 16.88 6.0 3 103.
16.90 0.036 g/L. 16.92 (a) 1.37. (b) 5.97. (c) 10.24.
16.94 Original precipitate was HgI2. In the presence of excess
KI, it redissolves as HgI22
4 . 16.96 7.8210.38. 16.98 (a) 3.60.
(b) 9.69. (c) 6.07. 16.100 137 g/mol. Ba. 16.102. 2. 16.104 (a)
12.6. (b) 8.8 3 1026 M. 16.106 (a) Sulfate, (b) sulfide, (c) iodide. 16.108 They are insoluble. 16.110 The ionized polyphenols have a dark color. The H1 ions from lemon juice shift the
equilibrium to the light color acid. 16.112 Yes. 16.114 (c).
16.116 (a) 1.7 3 1027 M. (b) MgCO3 is more soluble than
CaCO3. (c) 12.40. (d) 1.9 3 1028 M. (e) Ca21.
Chapter 17
17.6 3.3 3 1024 atm. 17.8 N2: 3.96 3 1018 kg; O2: 1.22 3 1018
kg; CO2: 2.63 3 1015 kg. 17.12 3.57 3 10219 J. 17.22 5.6 3
1014 kJ. 17.24 The wavelength is not short enough. 17.26 434
nm. 17.40 1.3 3 1010 kg. 17.42 Ethane and propane are greenhouse gases. 17.50 4.34. 17.58 1.2 3 10211 M/s. 17.60 (b).
17.66 0.12%. 17.68 Endothermic. 17.70 O2. 17.72 5.72.
17.74 394 nm. 17.76 It has a high activation energy. 17.78 Size
of tree rings are related to CO2 content. Age of CO2 in ice can
be determined by radiocarbon dating. 17.80 165 kJ/mol.
Chapter 18
18.6 (a) 0.02. (b) 9 3 10219. (c) 2 3 102181. 18.10 (c) , (d) ,
(a) , (e) , (b). 18.12 (a) 47.5 J/K. (b) 212.5 J/K. (c) 2242.8
J/K. 18.14 (a) S , 0, (b) S . 0, (c) S . 0, (d) S , 0.
Study Guide TOC
Textbook Website
MHHE Website
A-32
ANSWERS TO EVEN-NUMBERED PROBLEMS
18.18 (a) 21139 kJ. (b) 2140.0 kJ. (c) 22935.0 kJ. 18.20 (a)
At all temperatures. (b) Below 111 K. 18.24 8.0 3 104 J. 18.26
4.5 3 10281. 18.28 (a) 224.6 kJ. (b) 21.10 kJ. 18.30 2341
kJ/mol. 18.32 22.87 kJ. 18.36 1 3 103. 18.38 (a) 8.0 3 104 J.
(b) 4.0 3 104 J. (c) 23.2 3 104 J. (d) 6.4 3 104 J. 18.40 (i) No
reaction is possible because G . 0. (ii) The reaction has a
very large activation energy. (iii) Reactants already at their
equilibrium concentrations. 18.42 In all cases H . 0 and S
. 0. G , 0 for (a), 5 0 for (b), and . 0 for (c). 18.44 S .
0. 18.46 (b) Svap are larger for ethanol and water because of
hydrogen bonding. 18.48 (a) 2CO 1 2NO 88n 2CO2 1 N2. (b)
Oxidizing agent: NO; reducing agent: CO. (c) 1 3 10121. (d)
1.2 3 1018. From left to right. (e) No. 18.50 2.6 3 1029. 18.52
976 K. 18.54 S , 0; , 0. 18.56 55 J/K. 18.58 Increase in
entropy of the surroundings offsets the decrease in entropy of
the system. 18.60 56 J/K. 18.62 4.5 3 105. 18.64 4.5 3 10275
atm. 18.66 (a) True, (b) true, (c) false. 18.68 C 1 CuO 34
CO 1 Cu. 6.1 18.70 Crystal structure has disorder or has impurity. 18.72 (a) 7.6 3 1014. (b) 4.1 3 10212. 18.74 (a) A reverse
disproportionation reaction. (b) 8.2 3 1015. (c) Less effective.
18.76 1.8 3 1070. Reaction has a large activation energy. 18.78
2214.3 kJ. 18.80 KP 5 36. 981 K. No. 18.82 Yes. 18.84 XCO
5 0.45; XCO2 5 0.55.
Chapter 19
19.2 (a) Mn21 1 H2O2 1 2OH2 88n MnO2 1 2H2O. (b)
22
22
2Bi(OH)3 1 3SnO22
2 88n 2Bi 1 3H2O 1 3SnO3 . (c) Cr2O7
1
22
31
2
1 14H 1 3C2O4 88n 2Cr 1 6CO2 1 7H2O. (d) 2Cl 1
1
2ClO2
3 1 4H 88n Cl2 1 2ClO2 1 2H2O. 19.12 2.46 V.
19.14 Cl2(g) and MnO2
4 (aq). 19.16 Only (a) and (d) are spontaneous. 19.18 (a) Li, (b) H2, (c) Fe21, (d) Br2. 19.22 0.368 V.
19.24 (a) 2432 kJ, 5 3 1075, (b) 2104 kJ, 2 3 1018, (c)
2178 kJ, 1 3 1031, (d) 21.27 3 103 kJ, 8 3 10211. 19.26 0.37
V, 236 kJ, 2 3 106. 19.30 (a) 2.23 V, 2.23 V, 2430 kJ. (b)
0.02 V, 0.04 V, 223 kJ. 19.32 0.083 V. 19.34 0.010 V.
19.38 1.09 V. 19.46 (b) 0.64 g. 19.48 (a) $2.09 3 103. (b)
$2.46 3 103. (c) $4.70 3 103. 19.50 (a) 0.14 F. (b) 0.121 F.
(c) 0.10 F. 19.52 (a) Ag1 1 e2 88n Ag. (b) 2H2O 88n O2 1
4H1 1 4e2. (c) 6.0 3 102 C. 19.54 (a) 0.589 Cu. (b) 0.133 A.
19.56 2.3 h. 19.58 9.66 3 104 C. 19.60 0.0710 F. 19.62 0.156
M. 19.64 45.1%. 19.66 (b) 5.40%. 19.68 0.231 mg Ca21/mL
blood. 19.70 (a) 0.80 V. (b) 2Ag1 1 H2 88n 2Ag 1 2H1. (c)
(i) 0.92 V. (ii) 1.10 V. (d) The cell operates as a pH meter.
19.72 Fluorine gas reacts with water. 19.74 2.5 3 102 h.
21
1
255
19.76 Hg21
M.
2 . 19.78 [Mg ] 5 0.0500 M, [Ag ] 5 7 3 10
1.44 g. 19.80 (a) 0.206 L H2. (b) 6.09 3 1023/mol. 19.82 (a)
21356.8 kJ. (b) 1.17 V. 19.84 13. 19.86 6.8 kJ, 0.064.
19.88 1.4 A. 19.90 14. 19.92 1.60 3 10219 C/e2. 19.94 A cell
made of Li/Li1 and F2/F2 gives the maximum voltage of 5.92
V. 19.96 2 3 1020. 19.98 0.59 V. 19.100 (a) The reduction potential of O2 is insufficient to oxidize gold. (b) Yes. (c) 2Au 1
3F2 88n 2AuF3. 19.102 [Fe21] 5 0.0920 M, [Fe31] 5 0.0680 M.
Back
Forward
Main Menu
TOC
19.104 (b) 104 A ? h. (c) 2.01 V; 23.88 3 105 J. 19.106 (a)
Unchanged, (b) unchanged, (c) squared, (d) doubled, (e) doubled. 19.108 Stronger. 19.110 4.4 3 102 atm. 19.112 (a)
Zn 88n Zn21 1 2e2; }12} O2 1 2e2 88n O22. 1.65 V. (b) 1.63 V.
(c) 4.87 3 103 kJ/kg. (d) 62 L.
Chapter 20
20.12 111 h. 20.14 Roast the sulfide followed by reduction of
the oxide with coke or carbon monoxide. 20.16 (a) 8.9 3 1012
cm3. (b) 4.0 3 108 kg. 20.18 Iron does not need to be produced electrolytically. 20.28 (a) 2Na 1 2H2O 88n 2NaOH 1
H2. (b) 2NaOH 1 CO2 88n Na2CO3 1 H2O. (c) Na2CO3 1
2HCl 88n 2NaCl 1 CO2 1 H2O. (d) NaHCO3 1 HCl 88n
NaCl 1 CO2 1 H2O. (e) 2NaHCO3 88n Na2CO3 1 CO2 1
H2O. (f) No reaction. 20.30 5.59 L. 20.34 First react Mg with
HNO3 to form Mg(NO3)2. On heating, 2Mg(NO3)2 88n 2MgO
1 4NO2 1 O2. 20.36 The third electron is removed from the
neon core. 20.38 Helium has a closed-shell noble gas configuration. 20.40 (a) CaO, (b) Ca(OH)2, (c) an aqueous suspension
of Ca(OH)2. 20.44 60.7 h. 20.46 (a) 1.03 V. (b) 3.32 3 104 kJ.
20.48 4Al(NO3)3 88n 2Al2O3 1 12NO2 1 3O2. 20.50 Because
Al2Cl6 dissociates to form AlCl3. 20.52 From sp3 to sp2.
20.54 65.4 g/mol. 20.56 No. 20.58 (a) 1482 kJ. (b) 3152.8 kJ.
20.60 Magnesium reacts with nitrogen to form magnesium nitride. 20.62 (a) Al31 hydrolyzes in water to produce H1 ions.
(b) Al(OH)3 dissolves in a strong base to form Al(OH)2
4.
20.64 CaO 1 2HCl 88n CaCl2 1 H2O. 20.66 Al31 hydrolyzes
in water to produce H1 ions. 20.68 NaF: toothpaste additive;
Li2CO3: to treat mental illness; Mg(OH)2: antacid; CaCO3:
antacid; BaSO4: for X-ray diagnostic of digestive system;
Al(OH)2NaCO3: antacid. 20.70 (i) Both Li and Mg form oxides, (ii) like Mg, Li forms nitride, (iii) the carbonates, fluorides, and phosphates of Li and Mg have low solubilities.
Chapter 21
21.12 (a) Hydrogen reacts with alkali metals to form hydrides,
(b) hydrogen reacts with oxygen to form water. 21.14 Use palladium metal to separate hydrogen from other gases. 21.16 11 kg.
21.18 (a) H2 1 Cl2 88n 2HCl, (b) N2 1 3H2 88n 2NH3. (c)
2Li 1 H2 88n 2LiH. LiH 1 H2O 88n LiOH 1 H2. 21.26
:CqCS22. 21.28 (a) 2NaHCO3 88n Na2CO3 1 H2O 1 CO2.
(b) CO2 reacts with Ca(OH)2 solution to form a white precipitate (CaCO3). 21.30 On heating, the bicarbonate ion decom21
22
poses: 2HCO2
ions com3 88n CO3 1 H2O 1 CO2. Mg
22
bines with CO3 ions to form MgCO3. 21.32 First: 2NaOH 1
CO2 88n Na2CO3 1 H2O. Then: Na2CO3 1 CO2 1 H2O 88n
2NaHCO3. 21.34 Yes. 21.40 (a) 2NaNO3 88n 2NaNO2 1 O2.
(b) NaNO3 1 C 88n NaNO2 1 CO. 21.42 2NH3 1 CO2 88n
(NH2)2CO 1 H2O. At high pressures. 21.44 NH4Cl decomposes to form NH3 and HCl. 21.46 N is in its highest oxidation
Study Guide TOC
Textbook Website
MHHE Website
ANSWERS TO EVEN-NUMBERED PROBLEMS
state (15) in HNO3. 21.48 Favored reaction: 4Zn 1 NO23 1
10H1 88n 4Zn21 1 NH1
4 1 3H2O. 21.50 Linear. 21.52 21168
kJ. 21.54 P4. 21.56 60.4 g. 21.58 sp3. 21.66 2198.3 kJ, 6 3
1034, 6 3 1034. 21.68 0. 21.70 4.4 3 1011 mol; 1.4 3 1013 g.
21.72 79.1 g. 21.74 Cl, Br, and I atoms are too bulky around
the S atom. 21.76 35 g. 21.78 9H2SO4 1 8NaI 88n H2S 1 4I2
1 4H2O 1 8NaHSO4. 21.82 H2SO4 1 NaCl 88n HCl 1
NaHSO4. The HCl gas escapes, driving the equilibrium to the
right. 21.84 25.3 L. 21.86 Sulfuric acid oxidizes sodium bromide to molecular bromine. 21.88 2.81 L. 21.90 I2O5 1
5CO 88n I2 1 5CO2. 21.92 (a) SiCl4, (b) F2, (c) F, (d) CO2.
21.94 No change. 21.96 (a) 2Na 1 D2O 88n 2NaOD 1 D2.
(b) D2 1 Cl2 88n 2DCl. (c) Mg3N2 1 6D2O 88n 3Mg(OD)2
1 2ND3. (d) CaC2 1 2D2O 88n C2D2 1 Ca(OD)2. (e) Be2C
1 4D2O 88n 2Be(OD)2 1 CD4. (f) SO3 1 D2O 88n D2SO4.
21.98 (a) At elevated pressure, water boils above 100C, (b) so
the water is able to melt a larger area of sulfur deposit, (c) sulfur deposits are structurally weak. Conventional mining would
be dangerous. 21.100 The COD bond breaks at a slower rate.
A-33
dioactivity from the hemoglobin molecules. 23.52 An analogous Pauli exclusion principle for nucleons. 23.54 (a) 0.343
4
233
212
millicurie. (b) 237
93Np 88n 2a 1 91Pa. 23.56 (a) 1.040 3 10
J/nucleon. (b) 1.111 3 10212 J/nucleon. (c) 1.199 3 10212
J/nucleon. (d) 1.410 3 10212 J/nucleon. 23.58 187N 88n 188O 1
0
209
4
211
21b. 23.60 Radioactive dating. 23.62 83Bi 1 2a 88n 85At 1
1
209
211
20n. (b) 83Bi(a,2n) 85At. 23.64 The sun exerts a much greater
40
gravity on the particles. 23.66 2.77 3 103 yr. 23.68 (a) 19
K 88n
40
0
9
215
Ar
1
b
.
(b)
3.0
3
10
yr.
23.70
(a)
5.59
3
10
J,
2.84
3
18
11
10213 J. (b) 0.024 mole. (c) 4.06 3 106 kJ. 23.72 2.7 3 1014
I-131 atoms. 23.74 5.9 3 1023/mol. 23.76 All except gravitational. 23.78 U-238 and Th-232. 23.80 8.3 3 1024 nm.
23.82 31H.
Chapter 24
24.12 CH3CH2CH2CH2CH2Cl. CH3CH2CH2CH2CHClCH3.
CH3CH2CHClCH2CH3.
24.14
Chapter 22
22.12 (a) 13, (b) 6, (c) oxalate. 22.14 (a) Na: 11, Mo: 16.
(b) Mg: 12, W: 16. (c) Fe: 0. 22.16 (a) cis-dichlorobis(ethylenediammine)cobalt(III), (b) pentaamminechloroplatinum(IV)
chloride, (c) pentaamminechlorocobalt(III) chloride. 22.18 (a)
[Cr(en)2Cl2]1, (b) Fe(CO)5, (c) K2[Cu(CN)4], (d) [Co(NH3)4
(H2O)Cl]Cl2. 22.24 (a) 2, (b) 2. 22.26 (a) Two geometric isomers, (b) two optical isomers. 22.34 CN2 is a strong-field ligand. Absorbs near UV (blue) so appears yellow. 22.36 255
kJ/mol. 22.38 [Co(NH3)4Cl2]Cl. 22.42 Use 14CN2 label (in
NaCN). 22.44 First Cu(CN)2 (white) is formed. It redissolves
2
31
as Cu(CN)22
4 . 22.46 1.4 3 10 . 22.48 Mn . 22.50 Ti: 13; Fe:
13. 22.52 4 Fe atoms per hemoglobin molecule. 22.54 (a)
[Cr(H2O)6] Cl3. (b) [Cr(H2O)5Cl]Cl2 ? H2O. (c)
[Cr(H2O)4Cl2]Cl ? 2H2O. 22.56 21.8 3 102 kJ; 4 3 1031.
22.58 Iron is more abundant. 22.60 Oxyhemoglobin absorbs
higher energy light. 22.62 All except Fe21, Cu21, and Co21.
22.64 Dipole moment measurement. 22.66 EDTA sequesters essential metal ions (Ca21, Mg21).
24.16 (a) Alkene or cycloalkane, (b) alkyne, (c) alkane, (d) like
(a), (e) alkyne. 24.18 No. 24.20 (a) is alkane and (b) is alkene.
Only an alkene reacts with a hydrogen halide. 24.22 2630.8
kJ. 24.24 (a) cis-1,2-dichlorocylopropane, (b) trans-1,2dichlorocylopropane. 24.26 (a) 2-methylpentane, (b) 2,3,4trimethylhexane, (c) 3-ethylhexane, (d) 3-methyl-1,4-pentadiene, (e) 2-pentyne, (f) 3-phenyl-1-pentene.
24.28 (a)
(b)
(c)
(d)
Chapter 23
4
1
9
25
23.6 (a) 210b. (b) 40
20Ca. (c) 2a. (d) 0n. 23.14 (a) 3Li. (b) 11Na.
17
45
92
195
242
Sc.
23.16
Ne.
(b)
Ca.
(c)
Tc.
(d)
Hg.
(e)
(c) 48
80
96Cm.
21
10
20
43
23.18 6 3 109 kg/s. 23.20 (a) 4.55 3 10212 J; 1.14 3 10212
J/nucleon. (b) 2.36 3 10210 J; 1.28 3 10212 J/nucleon.
23.24 2.77 d. 23.26 2.7 d. 23.28 208
82Pb. 23.30 A: 0; B: 0.25
2
1
81
mole; C: 0; D: 0.75 mole. 23.34 (a) 80
34Se 1 1H 88n 1p 1 34Se.
9
2
1
9
10
1
4
(b) 4Be 1 1H 88n 21p 1 3Li. (c) 5B 1 0n 88n 2a 1
7
198
1
198
1
2
3Li. 23.36 80Hg 1 0n 88n 79Au 1 1p. 23.48 IO3 is only
formed from IO2
.
23.50
Incorporate
Fe-59
into
a
persons
4
body. After a few days isolate red blood cells and monitor ra-
Back
Forward
Main Menu
TOC
24.32 (a) 1,3-dichloro-4-methylbenzene, (b) 2-ethyl-1,4-dinitrobenzene, (c) 1,2,4,5-tetramethylbenzene. 24.36 (a) Ether,
(b) amine, (c) aldehyde, (d) ketone, (e) carboxylic acid, (f) alcohol, (g) amino acid. 24.38 HCOOH 1 CH3OH 88n
HCOOCH3 1 H2O. 24.40 (CH3)2CHOOOCH3. 24.42 (a)
Ketone, (b) ester, (c) ether. 24.44 2174 kJ. 24.46 (a), (c), (d),
(f). 24.48 (a) Rubbing alcohol, (b) vinegar, (c) moth balls, (d)
organic synthesis, (e) organic synthesis, (f) antifreeze, (g) natural gas, (h) synthetic polymer. 24.50 3, (b) 16, (c) 6. 24.52 (a)
C: 15.81 mg, H: 1.33 mg, O: 3.49 mg. (b) C6H6O.
Study Guide TOC
Textbook Website
MHHE Website
A-34
ANSWERS TO EVEN-NUMBERED PROBLEMS
Chapter 25
(c) Phenol.
24.54 Empirical and molecular
formula: C5H10O.
CH2PCHOCH2OOOCH2OCH3 24.56 (a) The C atoms
bonded to the methyl group and the amino group and the H
atom. (b) The C atoms bonded to Br. 24.58 CH3CH2CHO.
24.60 (a) Alcohol, (b) ether, (c) aldehyde, (d) carboxylic acid,
(e) amine. 24.62 The acids in lemon juice convert the amines
to the ammonium salts which have very low vapor pressures.
24.64 Methane (CH4), ethanol (C2H5OH), methanol (CH3OH),
isopropanol (C3H7OH), ethylene glycol (CH2OHCH2OH),
naphthalene (C10H8), acetic acid (CH3COOH). 24.66 (a) 1,
(b) 2, (c) 5. 24.68 Br2 dissociates into Br atoms which react
with CH4 to form CH3Br and HBr.
Back
Forward
Main Menu
TOC
25.8 O(CH2OCHClOCH2OCCl2O
) 25.10 By an addition
reaction involving styrene monomers. 25.12 (a)
CH2PCHOCHPCH2 (b) HO2C(CH2)6NH2 25.22 At 35C the
enzyme begins to denature. 25.28 Proteins are made of 20
amino acids. Nucleic acids are made of 4 building blocks
(purines, pyrimidines, sugar, phosphate group) only. 25.30 C-G
base pairs have 3 hydrogen bonds; A-T base pairs have two hydrogen bonds. 25.32 Leg muscles are active, have a high metabolic rate and hence a high concentration of myoglobin. The
iron content in Mb makes the meat look dark. 25.34 Insects
have blood that contains no hemoglobin. It is unlikely that a
human-sized insect could obtain sufficient oxygen for metabolism by diffusion. 25.36 There are 4 Fe atoms per hemoglobin
molecule. 25.38 Mostly dispersion forces. 25.40 Gly-Ala-PheGlu-His-Gly-Ala-Leu-Val. 25.42 No. Enzymes only act on one
of the two optical isomers of a compound.
Study Guide TOC
Textbook Website
MHHE Website
Вам также может понравиться
- Kjeldahl Computation, % Protein Determination PDFДокумент1 страницаKjeldahl Computation, % Protein Determination PDFHyumi DarthОценок пока нет
- Analytical Chemistry 20132Документ1 страницаAnalytical Chemistry 20132zzrot1Оценок пока нет
- 레이먼드 창의 대학기초화학 7판 선택된 문제의 정답Документ4 страницы레이먼드 창의 대학기초화학 7판 선택된 문제의 정답hjw5835209Оценок пока нет
- ÀÌ Õ Å Ã ÀÇ ÀÏ ÝÈ ÇÐ 14ÆÇ ¿ À ®Á Ä (Â Ö Øè Æ÷ÇÔ)Документ10 страницÀÌ Õ Å Ã ÀÇ ÀÏ ÝÈ ÇÐ 14ÆÇ ¿ À ®Á Ä (Â Ö Øè Æ÷ÇÔ)7bjg72xwh8Оценок пока нет
- Chemistry SolutionДокумент11 страницChemistry Solution:JОценок пока нет
- Answers To Selected Problems: A NB DДокумент5 страницAnswers To Selected Problems: A NB DpdfshareОценок пока нет
- StoichiometryДокумент5 страницStoichiometryHoney SilorioОценок пока нет
- Chapter 3 ConceptsДокумент4 страницыChapter 3 ConceptsEmiliaFigueroaAizpurúaОценок пока нет
- Chemistry Exercises EduДокумент1 страницаChemistry Exercises EduNacho SilvestreОценок пока нет
- Exercise 2 Answer KeyДокумент2 страницыExercise 2 Answer Keyyuen lok hinОценок пока нет
- CHEM 1411 - General Chemistry I Practice Problems, Chapters 1-3Документ8 страницCHEM 1411 - General Chemistry I Practice Problems, Chapters 1-3Lheidyy36Оценок пока нет
- Chapter 4 Answers Practice ExamplesДокумент7 страницChapter 4 Answers Practice ExamplesEmre Enes EdizОценок пока нет
- Uniten Chemistry FoundationДокумент13 страницUniten Chemistry FoundationAidil AizadОценок пока нет
- 레이먼드 창의 기본일반화학 14판 연습문제 답 (짝수 번호만 포함)Документ4 страницы레이먼드 창의 기본일반화학 14판 연습문제 답 (짝수 번호만 포함)키Оценок пока нет
- CHM 420 Questions (Chapter 2) Stoichiometry Naming: HAAH/2013Документ2 страницыCHM 420 Questions (Chapter 2) Stoichiometry Naming: HAAH/2013FAtma HAnysОценок пока нет
- TP2 QuimicaДокумент3 страницыTP2 Quimicafacundo mendez girardiОценок пока нет
- Stoichiometry WorksheetДокумент4 страницыStoichiometry WorksheetLily PanОценок пока нет
- Chapter 3Документ13 страницChapter 3Siti Afiqah TajuddinОценок пока нет
- Chm092 Tutorial 1 ChemistryДокумент2 страницыChm092 Tutorial 1 ChemistryALorenso SAmОценок пока нет
- Neet Kaka Test 02Документ4 страницыNeet Kaka Test 02Uvaish KHANОценок пока нет
- McMurry Chemistry Answers. Ch3Документ2 страницыMcMurry Chemistry Answers. Ch3Skruzdelyte MielaОценок пока нет
- Chapter 5: Chemical AccountingДокумент9 страницChapter 5: Chemical AccountingNahed YacoubОценок пока нет
- Solucionario Mortimer 5 Ed PDFДокумент96 страницSolucionario Mortimer 5 Ed PDFmvhernanОценок пока нет
- Assignment F22 1Документ15 страницAssignment F22 1linkeyue330Оценок пока нет
- Stoichiometry RevisionДокумент4 страницыStoichiometry RevisionAshwin Balaji0% (1)
- Moles and Stoichiometry Practice ProblemsДокумент5 страницMoles and Stoichiometry Practice ProblemsGiselle R Ranchez0% (1)
- Grade 11 Chemistry Review With AnswersДокумент15 страницGrade 11 Chemistry Review With AnswersRiham ElhabyanОценок пока нет
- Question Bank Chemistry 1Документ34 страницыQuestion Bank Chemistry 1Karanam.Ramakumar100% (3)
- Mole ConceptДокумент6 страницMole ConceptIndrasen KumarОценок пока нет
- Mole Concept Booklet SolutionДокумент33 страницыMole Concept Booklet SolutionAkshay PatelОценок пока нет
- ChemistryДокумент40 страницChemistryts8166100% (3)
- Salt Analysis (Answer) (12th)Документ16 страницSalt Analysis (Answer) (12th)Raju SinghОценок пока нет
- Gt-13 Jee Mains (Chem) Held On 31-March-15Документ2 страницыGt-13 Jee Mains (Chem) Held On 31-March-15blue_l1Оценок пока нет
- EntropyДокумент1 страницаEntropymark_smith_169Оценок пока нет
- Review For Test 2 ch3 and ch4Документ5 страницReview For Test 2 ch3 and ch4Alison VelázquezОценок пока нет
- CH 07Документ33 страницыCH 07Fabio OliveiraОценок пока нет
- Standard Entropies: Substance Substance SubstanceДокумент2 страницыStandard Entropies: Substance Substance SubstanceAliati Risma YantiОценок пока нет
- SBC 3Документ4 страницыSBC 3Erehh JeagerОценок пока нет
- November 2017 Chemistry SL Exam Paper 1Документ27 страницNovember 2017 Chemistry SL Exam Paper 1Arti ChamoliОценок пока нет
- Apex Tutorial: 12) Chemistry PathshalaДокумент2 страницыApex Tutorial: 12) Chemistry PathshalaratanОценок пока нет
- Chapter 3 Answers Practice Examples: 4.0 10 G MGCL 2.44×10 O 3.69 10 Au AtomsДокумент8 страницChapter 3 Answers Practice Examples: 4.0 10 G MGCL 2.44×10 O 3.69 10 Au AtomsEmre Enes EdizОценок пока нет
- Summative Exams For General and Analytical ChemistryFOR STUDENTДокумент5 страницSummative Exams For General and Analytical ChemistryFOR STUDENTsantos earlОценок пока нет
- Ch. 1,2,3,4Документ29 страницCh. 1,2,3,4Kankana ChoudhuryОценок пока нет
- Group2 and 7 RevisedДокумент46 страницGroup2 and 7 Revised123456Оценок пока нет
- Stoichiometry ProblemsДокумент6 страницStoichiometry ProblemsMieka LacisteОценок пока нет
- Practice Exam 2Документ10 страницPractice Exam 2Niomi ButtermilkОценок пока нет
- Mole Calculations Unit TestДокумент8 страницMole Calculations Unit Testapi-340717257Оценок пока нет
- Iitian Sushil Kumar: B.Tech Iit Madras M.Tech Iit MadrasДокумент3 страницыIitian Sushil Kumar: B.Tech Iit Madras M.Tech Iit MadrasprernashaluОценок пока нет
- A. BRFДокумент14 страницA. BRFIrish MercadoОценок пока нет
- Single Answer Type Questions: (+4, - 1)Документ5 страницSingle Answer Type Questions: (+4, - 1)Aadish JainОценок пока нет
- Chem 171-2-3: Final Exam Review Multiple Choice ProblemsДокумент9 страницChem 171-2-3: Final Exam Review Multiple Choice ProblemsSatram DasОценок пока нет
- Nelson Grade 11 Chemistry ReviewДокумент10 страницNelson Grade 11 Chemistry Reviewexhalait67% (3)
- Exercise HygieneДокумент4 страницыExercise HygieneputerikhaleedaaisyaОценок пока нет
- Quice Review Center: C) The Total Mass of The Atom A) RBДокумент5 страницQuice Review Center: C) The Total Mass of The Atom A) RBMary Francia RicoОценок пока нет
- CH 9 Packet KEYДокумент5 страницCH 9 Packet KEYEvoli NatasОценок пока нет
- 2022 2023 General Chemistry I Study Question Set 2Документ1 страница2022 2023 General Chemistry I Study Question Set 2Ömer Burak YükselОценок пока нет
- JEE Main Entrance Test Chemistry Model Paper 2Документ5 страницJEE Main Entrance Test Chemistry Model Paper 2PremKumarKalikiriОценок пока нет
- Stoichiometry 1Документ4 страницыStoichiometry 1hey mama don’t stress your mindОценок пока нет
- Mole Concept 11 PDFДокумент26 страницMole Concept 11 PDFSamyak Jha100% (1)
- Jee 2014 Booklet1 HWT StoichiometryДокумент10 страницJee 2014 Booklet1 HWT StoichiometryvarunkohliinОценок пока нет
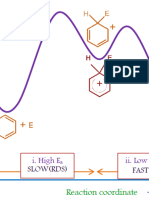
- Carbon Monoxide or Carbonyl: MO DescriptionДокумент3 страницыCarbon Monoxide or Carbonyl: MO DescriptionRSLОценок пока нет
- Coordination Isomers ListДокумент1 страницаCoordination Isomers ListRSLОценок пока нет
- S-Block Elments: Inorganic ChemistryДокумент8 страницS-Block Elments: Inorganic ChemistryRSLОценок пока нет
- Mechanism of Organic ReactionДокумент4 страницыMechanism of Organic ReactionRSLОценок пока нет
- NH4BH4Документ1 страницаNH4BH4RSLОценок пока нет
- Basics of ElectrochemistryДокумент22 страницыBasics of ElectrochemistryRSLОценок пока нет
- Epoxides Ring-Opening - Chemistry LibreTextsДокумент3 страницыEpoxides Ring-Opening - Chemistry LibreTextsRSLОценок пока нет
- Priority List IUPACДокумент1 страницаPriority List IUPACRSLОценок пока нет
- DebereinerДокумент4 страницыDebereinerRSLОценок пока нет
- Turkevich1985 Article ColloidalGoldPartII PDFДокумент7 страницTurkevich1985 Article ColloidalGoldPartII PDFRSLОценок пока нет
- Colloidal Gold. Part I: Historical and Preparative Aspects, Morphology and StructureДокумент6 страницColloidal Gold. Part I: Historical and Preparative Aspects, Morphology and StructureRSL100% (1)
- Piezoelectric Materials: Crystal Orientation and Poling Direction - COMSOL Blog PDFДокумент4 страницыPiezoelectric Materials: Crystal Orientation and Poling Direction - COMSOL Blog PDFRSLОценок пока нет
- Colloids: Thomas Graham (1861) Studied The Ability of Dissolved Substances ToДокумент28 страницColloids: Thomas Graham (1861) Studied The Ability of Dissolved Substances ToRSLОценок пока нет
- © 1934 Nature Publishing GroupДокумент2 страницы© 1934 Nature Publishing GroupRSLОценок пока нет
- 2019dec-03 - Ionic Equilibrium - PracticeSheetДокумент2 страницы2019dec-03 - Ionic Equilibrium - PracticeSheetRSLОценок пока нет
- List of Straight-Chain AlkanesДокумент6 страницList of Straight-Chain AlkanesRSLОценок пока нет
- Analysis Report of Criminal and Financial Background Details of Winners in Lok Sabha 2019 ElectionsДокумент397 страницAnalysis Report of Criminal and Financial Background Details of Winners in Lok Sabha 2019 ElectionsRSLОценок пока нет
- Ionic Equilibrium - Salt Hydrolysis Practice Questions (Level-1)Документ3 страницыIonic Equilibrium - Salt Hydrolysis Practice Questions (Level-1)RSLОценок пока нет
- EAS Worksheet 1 SubjДокумент1 страницаEAS Worksheet 1 SubjRSLОценок пока нет
- Redox Reactions and Balancing Using Oxidation Number & NfactorДокумент1 страницаRedox Reactions and Balancing Using Oxidation Number & NfactorRSLОценок пока нет
- Chemistry Organic GOC Reaction Mechanism Elimination E1 E2 E1cBДокумент4 страницыChemistry Organic GOC Reaction Mechanism Elimination E1 E2 E1cBRSL100% (1)
- The World of Chemistry Episode 5 - A Matter of StateДокумент2 страницыThe World of Chemistry Episode 5 - A Matter of StateRSL0% (1)
- Koopmans TheoremДокумент14 страницKoopmans TheoremRSLОценок пока нет
- Colligative Properties: Cryoscopy & EbulliosДокумент30 страницColligative Properties: Cryoscopy & EbulliosRSL100% (1)
- Isoelectric Point, PiДокумент2 страницыIsoelectric Point, PiRSLОценок пока нет
- Crossword Coordination CompoundsДокумент2 страницыCrossword Coordination CompoundsRSLОценок пока нет
- Factors Affecting Relative Strengths of Acids and BasesДокумент1 страницаFactors Affecting Relative Strengths of Acids and BasesRSL80% (5)
- Tutorial 3 - Questions Only - PVT Behavior - Ideal Gas - Virial EOSДокумент5 страницTutorial 3 - Questions Only - PVT Behavior - Ideal Gas - Virial EOSMihir Kumar Mech100% (1)
- Calculate The Ultimate Moment of Resistance of The BeamДокумент6 страницCalculate The Ultimate Moment of Resistance of The BeamMatiur Rahman Basum100% (1)
- VideoДокумент7 страницVideoIvy KayoОценок пока нет
- 21 - Suitability of Shredded Tires For Use in Landfill Leachate Collection SystemsДокумент13 страниц21 - Suitability of Shredded Tires For Use in Landfill Leachate Collection SystemsbelavelosoОценок пока нет
- Light Rails: National Standard of The People's Republic of ChinaДокумент13 страницLight Rails: National Standard of The People's Republic of ChinaBaoОценок пока нет
- Kinetic Theory of MatterДокумент5 страницKinetic Theory of Matterapi-3839633Оценок пока нет
- 10em Chem 1 Foundation Chap-1Документ46 страниц10em Chem 1 Foundation Chap-1Legendary MathematicianОценок пока нет
- Theoretical Framework: Compressive Strength TestДокумент11 страницTheoretical Framework: Compressive Strength TestqwertasdfgОценок пока нет
- Normal ResistivityДокумент1 страницаNormal ResistivityShahzad KhanОценок пока нет
- Lopez PrefiДокумент5 страницLopez PrefiCarl Adrienne LopezОценок пока нет
- Study On The Second Stage of Chalcocite Leaching in Column With RedoxДокумент12 страницStudy On The Second Stage of Chalcocite Leaching in Column With RedoxFrancisco CampbellОценок пока нет
- Physical Behavior of Gases: Kinetic TheoryДокумент12 страницPhysical Behavior of Gases: Kinetic TheoryPAUL KOLEREОценок пока нет
- Reinforcement Worksheet of Chemical BondingДокумент7 страницReinforcement Worksheet of Chemical BondingAreeba IftikharОценок пока нет
- FinalExam B AnsKeyДокумент12 страницFinalExam B AnsKeyDVRaoОценок пока нет
- Heat, The Mechanical UniverseДокумент2 страницыHeat, The Mechanical UniversealadinsaneОценок пока нет
- Cement Class 52.5R (Given) Aggregate Type: Coarse Crushed (Given) Graph From (Lecture 8, Slide 22) Compressive Strength 55mpa (Found)Документ11 страницCement Class 52.5R (Given) Aggregate Type: Coarse Crushed (Given) Graph From (Lecture 8, Slide 22) Compressive Strength 55mpa (Found)Shael BridgelalОценок пока нет
- C Mastic: Main ApplicationДокумент1 страницаC Mastic: Main ApplicationHung Mai Van100% (2)
- Current Contractor List Contractor Type PhoneДокумент18 страницCurrent Contractor List Contractor Type PhoneJawhar FarhatОценок пока нет
- Ashirvad UndergroundДокумент27 страницAshirvad Undergroundjinalshah1012713Оценок пока нет
- Wps Ausa Gtaw Smaw 002 2018 Ok RДокумент2 страницыWps Ausa Gtaw Smaw 002 2018 Ok RIvan Dario Carrillo Rubio0% (1)
- Experimental and CFD Analysis of Aluminium Heat Sinks For Avionics ApplicationsДокумент7 страницExperimental and CFD Analysis of Aluminium Heat Sinks For Avionics ApplicationsIJIRSTОценок пока нет
- 2013 Alkane Tutorial (Solutions)Документ7 страниц2013 Alkane Tutorial (Solutions)Pinzhen ChenОценок пока нет
- Lanco Antifloat D-14, TDSДокумент2 страницыLanco Antifloat D-14, TDSZein HayekОценок пока нет
- PT Buana Laksa Abadi: 1. Design CriteriaДокумент3 страницыPT Buana Laksa Abadi: 1. Design CriteriaKris SiregarОценок пока нет
- 175 026510Документ2 страницы175 026510Abu Anas M.SalaheldinОценок пока нет
- Training Report of Different Block of Haldia RefineryДокумент63 страницыTraining Report of Different Block of Haldia Refineryneo_86868675% (4)
- International Earth Science Olympiad (IESO) : RocksДокумент14 страницInternational Earth Science Olympiad (IESO) : RocksansariОценок пока нет
- Techno-Economic Assessment About Propylene GlycolДокумент4 страницыTechno-Economic Assessment About Propylene GlycolIntratec SolutionsОценок пока нет
- Structural Design of A Water Intake Tower Located Inside A ReservoirДокумент10 страницStructural Design of A Water Intake Tower Located Inside A ReservoirArifullah WazirОценок пока нет
- Ja504539w Si 001Документ20 страницJa504539w Si 001Andrea Melissa CabreraОценок пока нет
- An Applied Guide to Water and Effluent Treatment Plant DesignОт EverandAn Applied Guide to Water and Effluent Treatment Plant DesignРейтинг: 5 из 5 звезд5/5 (4)
- Well Control for Completions and InterventionsОт EverandWell Control for Completions and InterventionsРейтинг: 4 из 5 звезд4/5 (10)
- Process Plant Equipment: Operation, Control, and ReliabilityОт EverandProcess Plant Equipment: Operation, Control, and ReliabilityРейтинг: 5 из 5 звезд5/5 (1)
- Process Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersОт EverandProcess Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersОценок пока нет
- Piping Engineering Leadership for Process Plant ProjectsОт EverandPiping Engineering Leadership for Process Plant ProjectsРейтинг: 5 из 5 звезд5/5 (1)
- Distillation Design and Control Using Aspen SimulationОт EverandDistillation Design and Control Using Aspen SimulationРейтинг: 5 из 5 звезд5/5 (2)
- Sodium Bicarbonate: Nature's Unique First Aid RemedyОт EverandSodium Bicarbonate: Nature's Unique First Aid RemedyРейтинг: 5 из 5 звезд5/5 (21)
- Guidelines for Chemical Process Quantitative Risk AnalysisОт EverandGuidelines for Chemical Process Quantitative Risk AnalysisРейтинг: 5 из 5 звезд5/5 (1)
- Lees' Process Safety Essentials: Hazard Identification, Assessment and ControlОт EverandLees' Process Safety Essentials: Hazard Identification, Assessment and ControlРейтинг: 4 из 5 звезд4/5 (4)
- Troubleshooting Vacuum Systems: Steam Turbine Surface Condensers and Refinery Vacuum TowersОт EverandTroubleshooting Vacuum Systems: Steam Turbine Surface Condensers and Refinery Vacuum TowersРейтинг: 4.5 из 5 звезд4.5/5 (2)
- Functional Safety from Scratch: A Practical Guide to Process Industry ApplicationsОт EverandFunctional Safety from Scratch: A Practical Guide to Process Industry ApplicationsОценок пока нет
- Coupled CFD-DEM Modeling: Formulation, Implementation and Application to Multiphase FlowsОт EverandCoupled CFD-DEM Modeling: Formulation, Implementation and Application to Multiphase FlowsОценок пока нет
- The Perfumed Pages of History: A Textbook on Fragrance CreationОт EverandThe Perfumed Pages of History: A Textbook on Fragrance CreationРейтинг: 4 из 5 звезд4/5 (1)
- Fundamentals of Risk Management for Process Industry EngineersОт EverandFundamentals of Risk Management for Process Industry EngineersОценок пока нет
- A New Approach to HAZOP of Complex Chemical ProcessesОт EverandA New Approach to HAZOP of Complex Chemical ProcessesОценок пока нет
- Fun Facts about Hydrogen : Chemistry for Kids The Element Series | Children's Chemistry BooksОт EverandFun Facts about Hydrogen : Chemistry for Kids The Element Series | Children's Chemistry BooksОценок пока нет
- Fun Facts about Carbon : Chemistry for Kids The Element Series | Children's Chemistry BooksОт EverandFun Facts about Carbon : Chemistry for Kids The Element Series | Children's Chemistry BooksОценок пока нет
- Handbook of Cosmetic Science: An Introduction to Principles and ApplicationsОт EverandHandbook of Cosmetic Science: An Introduction to Principles and ApplicationsH. W. HibbottРейтинг: 4 из 5 звезд4/5 (6)
- Cathodic Protection: Industrial Solutions for Protecting Against CorrosionОт EverandCathodic Protection: Industrial Solutions for Protecting Against CorrosionОценок пока нет
- Bioinspired Materials Science and EngineeringОт EverandBioinspired Materials Science and EngineeringGuang YangОценок пока нет