Академический Документы
Профессиональный Документы
Культура Документы
Superficies Termodinamicas
Загружено:
Max Jhon Arias BaspineiroИсходное описание:
Оригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Superficies Termodinamicas
Загружено:
Max Jhon Arias BaspineiroАвторское право:
Доступные форматы
Superficies Termodinámicas
8-Sep-06 Alicia Ma. Esponda Cascajares 1
Ley de Boyle-Mariotte
En 1662 Robert Boyle formuló la generalización que
relación el volumen de una masa de gas con la
presión ejercida sobre el.
Estos resultados fueron confirmados por Louis
Mariotte.
Determinó que V α 1/P, cuando la temperatura y la
masa permanecen constantes.
Si la presión ejercida sobre una masa de gas
aumenta, el volumen disminuye por lo que el numero
de moléculas por unidad de volumen aumenta junto
con el numero de impactos moleculares sobre las
paredes del recipiente, aunque la fuerza de cada
impacto permanece constante.
8-Sep-06 Alicia Ma. Esponda Cascajares 2
Ley de Boyle-Mariotte
1
V =K⋅ o P ⋅ V = constante
P
Un mismo gas que
cambie de estado: P1 ⋅ V1 = P2 ⋅ V2
8-Sep-06 Alicia Ma. Esponda Cascajares 3
Ley de Charles
En 1801 Jacques Charles estableció la relación entre
la variación del volumen con la temperatura.
Determinó que V α T, cuando la presión y la masa
permanecen constantes.
A medida que el gas se calienta, la velocidad media
de las moléculas aumenta produciéndose mas
impactos por unidad de tiempo sobre las paredes del
recipiente y a una mayor fuerza por impacto. Como
la presión permanece constante, el volumen debe
aumentar para compensar este incremento en el
efecto de presión.
8-Sep-06 Alicia Ma. Esponda Cascajares 4
Ley de Charles
o V
V = K ⋅T = constante
T
Un mismo gas que
V1 V2
cambie de estado: =
T1 T2
8-Sep-06 Alicia Ma. Esponda Cascajares 5
Ley de Gay-Lussac
Joseph Gay-Lussac encontró la relación existente
entre la variación de presión y la temperatura.
Determinó que P α T, cuando el volumen y la masa
permanecen constantes.
Si se aumenta la temperatura de una masa de gas
manteniendo el volumen constante, las moléculas
aumentan de velocidad, resultando en un mayor
numero de impactos por unidad de tiempo sobre las
paredes del recipiente generando un aumento de la
presión.
8-Sep-06 Alicia Ma. Esponda Cascajares 6
Ley de Gay-Lussac
o P
P = K ⋅T = constante
T
Un mismo gas que
P1 P2
cambie de estado: =
T1 T2
8-Sep-06 Alicia Ma. Esponda Cascajares 7
Ley General de los Gases
Con cualesquiera dos de
estas tres leyes, se
puede establecer la Ley
General de los Gases P1 ⋅ V1 P2 ⋅ V2
=
que establece el T1 T2
comportamiento de los
gases de bajas
densidades.
8-Sep-06 Alicia Ma. Esponda Cascajares 8
Ley de Avogadro
Amadeo Avogadro enuncio su ley como
explicación a la ley de Gay-Lussac.
“Volúmenes iguales de gases, en las mismas
condiciones de temperatura y presión,
contienen el mismo número de moléculas”.
A partir de sus experimentos determino el
numero de Avogadro NA, que representa el
numero de átomos que hay en un átomo-
gramo de cualquier elemento.
NA=6.023x1023[mol-1]
8-Sep-06 Alicia Ma. Esponda Cascajares 9
Ecuaciones de Estado
Es cualquier ecuación que exprese la
relación entre la temperatura, la presión
y el volumen de una masa determinada
de una substancia.
Las leyes vistas anteriormente son
ecuaciones de estado.
8-Sep-06 Alicia Ma. Esponda Cascajares 10
Ecuaciones de Estado
De las leyes vistas sabemos que
PVαT
Se puede asignar una igualdad utilizando
una constante de proporcionalidad.
PV=CT
La constante de proporcionalidad es igual a
una constante k por el numero de
moléculas del gas.
C=kN
8-Sep-06 Alicia Ma. Esponda Cascajares 11
Ecuaciones de Estado
La constante k es la constante de
Boltzmann
k=1.381x10-23[J/K]
Es una constante de los gases por molécula.
Sustituyendo lo anterior en la ecuación de
estado tenemos:
PV=kNT
El numero de moléculas de un gas se
relaciona con el numero de Avogadro como:
N=n NA
8-Sep-06 Alicia Ma. Esponda Cascajares 12
Ecuaciones de Estado
Siendo n los moles de substancia.
Sustituyendo queda:
P V = n NA k T
8-Sep-06 Alicia Ma. Esponda Cascajares 13
Constante Universal de los Gases
Se encuentra que para todos los gases
a bajas presiones y altas temperaturas
que hay una constante que se define
como:
Ru = NA k
Y ese valor resulta ser:
Ru=8.3143 [J/molK]
8-Sep-06 Alicia Ma. Esponda Cascajares 14
Ecuaciones de Estado
Substituyendo la constante de los gases en
las ecuaciones de estado tenemos:
P V = n Ru T
Para poder expresar las ecuaciones en
términos de la masa, se hace a través de la
relación de n con la masa, es decir mediante
la masa molar M.
m =n M
8-Sep-06 Alicia Ma. Esponda Cascajares 15
Ecuaciones de Estado
Substituyendo el despeje se m
P ⋅V = Ru ⋅ T
tiene que: M
Se define a R, como la Ru
constante de un gas particular R=
M
como:
De manera que sustituyendo P ⋅V = m ⋅ R ⋅ T
nos queda:
R es diferente para cada gas,
pero su valor se obtiene de
tablas.
8-Sep-06 Alicia Ma. Esponda Cascajares 16
8-Sep-06 Alicia Ma. Esponda Cascajares 17
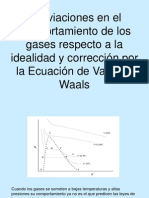
Ecuación de van der Waals
Es una fórmula mas exacta para
expresar el comportamiento de los
gases.
Fue presentada en 1873 como un ⎛ a ⎞
P + 2 ⎟ ⋅ (V − b ) = R ⋅ T
intento de mejorar los resultados ⎜⎝ V ⎠
obtenidos.
La cantidad (a/V2) representa la
corrección por las fuerzas atractivas
intermoleculares y b la corrección
del volumen por el espacio ocupado
por las moléculas.
8-Sep-06 Alicia Ma. Esponda Cascajares 18
Ecuación Benedict-Webb-Rubin
Es la fórmula utilizada para generar las tablas
termodinámicas.
Fue desarrollada en 1940.
Cuenta con 8 constantes empíricas
incluyendo a las constantes a y b de la
ecuación de van der Waals.
C0
R ⋅ T ⋅ B0 − A0 −
P=
R ⋅T
+ T 2 + R ⋅ T ⋅ b − a + a ⋅ α + c ⎛⎜1 + γ ⎞⎟e −γ v2
v v2 v3 v6 v3 ⋅T 2 ⎝ v2 ⎠
8-Sep-06 Alicia Ma. Esponda Cascajares 19
Propiedades del líquido y vapor saturado del
agua (Tabla de Presión)
Presión Tempera Volumen Energía Interna Entalpía Entropía
[MPa] tura Específico [kJ/kg] [kJ/kg] [kJ/kgK]
[°C] [m3/kg]
P T vf vg uf ufg ug hf hfg hg sf sfg sg
0.2 120.23 0.0010 0.88 504.4 2025 2529 504. 2202 2706. 1.530 5.59 7.12
61 57 7 .0 .5 68 .0 6 0 70 71
0.4 143.63 0.0010 0.46 604.2 1949 2553 604. 2133 2738. 1.776 5.11 6.89
84 25 9 .3 .6 73 .8 5 6 93 58
0.6 158.85 0.0011 0.31 669.8 1897 2567 670. 2086 2756. 1.931 4.82 6.76
01 57 8 .5 .4 54 .3 8 1 89 00
0.8 170.43 0.0011 0.24 720.2 1856 2576 721. 2048 2769. 2.046 4.61 6.66
15 04 0 .6 .8 10 .0 1 1 66 27
8-Sep-06 Alicia Ma. Esponda Cascajares 20
Ecuación Lee-Kesler
Es una derivación de la ecuación BWR
desarrollada en 1975, utilizada ahora en las
tablas termodinámicas computarizadas.
Tiene 12 constantes empíricas.
⎛ γ ⎞
⎜− ⎟
⎛ ⎞ ⎜ v' 2 ⎟
⎜β + γ 2
Pr ⋅ v' r B C D c4 ⎝ ⎠
⎟
r
= 1+ + 2 + 5 + 3 ⎜ ⎟
Tr ⋅ v' r
2
Tr v' r v' r v' r ⎝ v' r ⎠
8-Sep-06 Alicia Ma. Esponda Cascajares 21
Ecuación Lee-Kesler
Donde:
b b b
B = b 1 − 2
− 3
2
− 4
3
T r T r T r
c c
C = c 1 − 2
− 3
3
T r T r
d
D = d 1 − 2
T r
v
v 'r =
R ⋅ T c
P c
8-Sep-06 Alicia Ma. Esponda Cascajares 22
8-Sep-06 Alicia Ma. Esponda Cascajares 23
Temperatura y Presión Críticas
Las bajas temperaturas y altas presiones
favorecen la conversión de los gases en
líquidos y de los líquidos en sólidos ya que se
tiende a incrementar la atracción molecular y
se disminuye la energía cinética de las
moléculas.
Si la temperatura no se reduce, por medio de
la presión no siempre se convierte un gas en
un liquido.
8-Sep-06 Alicia Ma. Esponda Cascajares 24
Temperatura y Presión Criticas
Temperatura Crítica es la temperatura por
encima de la cual la substancia no puede
licuarse por la sola aplicación de presión.
Presión Crítica es la presión necesaria para
licuar un gas a su temperatura critica.
Cada substancia tiene una temperatura y
presión critica especifica.
Por encima de ese punto (critico) no hay
distinción entre las fases de las substancias.
8-Sep-06 Alicia Ma. Esponda Cascajares 25
Temperatura y Presión Criticas
Cuanto mayor es la temperatura critica,
menor es la presión necesaria para
licuar un gas a temperatura ambiente.
8-Sep-06 Alicia Ma. Esponda Cascajares 26
8-Sep-06 Alicia Ma. Esponda Cascajares 27
8-Sep-06 Alicia Ma. Esponda Cascajares 28
8-Sep-06 Alicia Ma. Esponda Cascajares 29
8-Sep-06 Alicia Ma. Esponda Cascajares 30
8-Sep-06 Alicia Ma. Esponda Cascajares 31
8-Sep-06 Alicia Ma. Esponda Cascajares 32
8-Sep-06 Alicia Ma. Esponda Cascajares 33
8-Sep-06 Alicia Ma. Esponda Cascajares 34
Gas Ideal
Es todo gas que P ⋅V = m ⋅ R ⋅ T
cumple con las
ecuaciones de P ⋅ V = n ⋅ Ru ⋅ T
estado:
Estas ecuaciones
funcionan a bajas
densidades y en un
cierto rango de
temperatura y
presión.
8-Sep-06 Alicia Ma. Esponda Cascajares 35
Factor de Compresibilidad
Es un factor que se introduce
para definir en que rango de Pv
densidad y de presión- Z≡
temperatura funcionan las RT
ecuaciones de estado.
Z es igual a 1 para un gas
ideal.
Si Z<1 la densidad es mucho
mayor que la que predicen
las ecuaciones de estado.
8-Sep-06 Alicia Ma. Esponda Cascajares 36
8-Sep-06 Alicia Ma. Esponda Cascajares 37
Principio de los Estados
Correspondientes
El factor de
compresibilidad para P
todos los gases es PR ≡
aproximadamente el Pc
mismo cuando estos
tienen la misma
T
presión y
temperatura
TR ≡
reducida. Tc
8-Sep-06 Alicia Ma. Esponda Cascajares 38
8-Sep-06 Alicia Ma. Esponda Cascajares 39
Calidad
mvapor mg
Calidad = x = =
mtotal mg + m f
El subíndice g indica el estado de vapor
saturado
El subíndice f se refiere al estado de
líquido saturado.
8-Sep-06 Alicia Ma. Esponda Cascajares 40
Calidad (prop. Intensivas)
y total − y f y total − y f
Calidad = x = =
y fg yg − y f
Donde y es la propiedad intensiva de
que se trate.
8-Sep-06 Alicia Ma. Esponda Cascajares 41
Ejercicio
Una vasija cuyo volumen es 0.4 [m3]
contiene 2 [kg] de agua en mezcla de
líquido y vapor en equilibrio a una
presión de 600 [kPa]. Calcular:
La calidad de la mezcla
La masa de líquido
La masa de vapor
El volumen de líquido y vapor.
8-Sep-06 Alicia Ma. Esponda Cascajares 42
Propiedades del líquido y vapor saturado del
agua (Tabla de Presión)
Presión Temperatura Volumen Específico Energía Interna Entalpía
[MPa] [°C] [m3/kg] [kJ/kg] [kJ/kg]
P T vf vg uf ufg ug hf hfg hg sf
0.2 120.23 0.001061 0.8857 504.47 2025.0 2529.5 504.68 2202.0 2706.6 1.530
0.4 143.63 0.001084 0.4625 604.29 1949.3 2553.6 604.73 2133.8 2738.5 1.776
0.6 158.85 0.001101 0.3157 669.88 1897.5 2567.4 670.54 2086.3 2756.8 1.931
0.8 170.43 0.001115 0.2404 720.20 1856.6 2576.8 721.10 2048.0 2769.1 2.046
8-Sep-06 Alicia Ma. Esponda Cascajares 43
Ejercicio
La calidad de la
mezcla
Se calcula el volumen V 0.4
v= = = 0.20 ⎡ m 3
⎤
específico: m 2. 0 ⎢⎣ kg ⎥⎦
v fg = v g − v f
De las tablas se
obtiene vfg v fg = 0.3157 − 0.001101
v fg = 0.3146
8-Sep-06 Alicia Ma. Esponda Cascajares 44
Ejercicio
La calidad de la mezcla
v −vf 0.2 − 0.001101
x= = = 0.6322
v fg 0.3146
Es decir 63.22%
8-Sep-06 Alicia Ma. Esponda Cascajares 45
Ejercicio
La masa de líquido
m f = m(1 − x ) = (2.0 )(1 − 0.6322) = 0.7356[kg ]
La masa de vapor
m g = m( x ) = (2.0 )(0.6322) = 1.2644[kg ]
8-Sep-06 Alicia Ma. Esponda Cascajares 46
Ejercicio
El volumen de líquido
[ ]
V f = v f m f = (0.001101)(0.7356) = 0.0008 m 3
El volumen de vapor
[ ]
V g = v g m g = (0.3157 )(1.2644) = 0.3992 m 3
8-Sep-06 Alicia Ma. Esponda Cascajares 47
Вам также может понравиться
- Teoría Del Gas Real: La Relación Exacta Para Gases RealesОт EverandTeoría Del Gas Real: La Relación Exacta Para Gases RealesОценок пока нет
- Libro PDFДокумент226 страницLibro PDFVladimir Morales C100% (4)
- Prob TermodinámicaДокумент87 страницProb TermodinámicaiecarlosОценок пока нет
- Gases 2020-1Документ10 страницGases 2020-1Edward GilОценок пока нет
- Calor y GasesДокумент16 страницCalor y GasesANTOINE HAFID CHUCHON DE LA CRUZОценок пока нет
- TEMA VIII Estado GaseosoДокумент32 страницыTEMA VIII Estado GaseosoGREDY JOSEPH SULCA MARTINEZОценок пока нет
- Quimica 2Документ12 страницQuimica 2Maria Isabel Ángeles Quevedo100% (1)
- Practica 2Документ8 страницPractica 2AyrtonОценок пока нет
- Practica 1 Quimica AplicadaДокумент10 страницPractica 1 Quimica AplicadaAlfredoОценок пока нет
- Ecuación de Estado para Gases RealesДокумент13 страницEcuación de Estado para Gases RealesCristian Ccaso MamaniОценок пока нет
- Cartilla Problema FQ2016Документ100 страницCartilla Problema FQ2016Francesca Toledo100% (1)
- PracticaДокумент34 страницыPracticaDaniel ChávezОценок пока нет
- Estado Gaseoso Quimica GeneralДокумент70 страницEstado Gaseoso Quimica GeneralRiverDavid Rodriguez0% (1)
- V. Teoria Cinetica de Los GasesДокумент28 страницV. Teoria Cinetica de Los Gasesanon_580798559Оценок пока нет
- Fisico Qumica 1Документ17 страницFisico Qumica 1yhuillcamОценок пока нет
- Informe de Gases IdealesДокумент8 страницInforme de Gases IdealesJulio Cesar CancioОценок пока нет
- Clase 2 Gases RealesДокумент74 страницыClase 2 Gases RealesPETER ANDRES JACOBO ABURTOОценок пока нет
- Capitulo 2Документ9 страницCapitulo 2Gastón SarapuraОценок пока нет
- Estudio de Los GasesДокумент24 страницыEstudio de Los GasesRaysa LlacsahuangaОценок пока нет
- Clase+2 TD 2022Документ34 страницыClase+2 TD 2022patriarca28Оценок пока нет
- GasesДокумент9 страницGasesAndres CasanovaОценок пока нет
- Taller de Gases IdealesДокумент10 страницTaller de Gases IdealesMARIA JOSE QUIROGAS CUELLOОценок пока нет
- Sem 8 SenatiДокумент17 страницSem 8 SenatiAngie EspinozaОценок пока нет
- Ley de CharlesДокумент4 страницыLey de Charlesjoaquin arredondoОценок пока нет
- GAS IDEAL NrasДокумент19 страницGAS IDEAL NrasAngelo Bazán LadinesОценок пока нет
- Clase 2 GasesДокумент52 страницыClase 2 GasesPaula MoralesОценок пока нет
- Laboratorio #4 TermoДокумент11 страницLaboratorio #4 TermoRaelee IsabelОценок пока нет
- Estado GaseosoДокумент12 страницEstado GaseosoandreaОценок пока нет
- Apend CДокумент16 страницApend CJunior PachecoОценок пока нет
- Practica 1 QuimicaДокумент17 страницPractica 1 QuimicaLuis Miguel Juarez MunguiaОценок пока нет
- Semana 2 FQ Gases RealesДокумент24 страницыSemana 2 FQ Gases RealesJacqueline Machón CamposОценок пока нет
- Gases RealesДокумент35 страницGases RealesMarco Cisneros,Yheferson Maza,Diego Díaz,Jairo Giohayro,David Samamé,Gino Cuadros,Ricardo Trinidad100% (1)
- Laboratorio Fisico Quimica AmbientalДокумент32 страницыLaboratorio Fisico Quimica AmbientalmariaОценок пока нет
- UNIDAD II - TermodinamicaДокумент10 страницUNIDAD II - TermodinamicaIlse AhujaОценок пока нет
- Unidad 2 Gas IdealДокумент35 страницUnidad 2 Gas IdealIndiraОценок пока нет
- Gases Ideales y RealesДокумент13 страницGases Ideales y RealesLee HansonОценок пока нет
- Gases-Primer PrincipioДокумент33 страницыGases-Primer PrincipioFer SalamancaОценок пока нет
- Sem 9 Ley de GasesДокумент28 страницSem 9 Ley de GasesHady Quispe LeónОценок пока нет
- El Estado GaseosoДокумент18 страницEl Estado GaseosoMaurian SolisОценок пока нет
- Clase - 1Документ50 страницClase - 1Leonela Ghissel Terrones RochaОценок пока нет
- TermodinámicaДокумент4 страницыTermodinámicaElaisaVarasОценок пока нет
- GASESДокумент16 страницGASEScandereyna99Оценок пока нет
- Sustancia PurasДокумент51 страницаSustancia PurasALEX DE LA CRUZ CCARHUAYPIÑAОценок пока нет
- Laboratorio #4 TermoДокумент11 страницLaboratorio #4 TermoRaelee Isabel0% (1)
- Termo 3Документ36 страницTermo 3José Emilio GuardiaОценок пока нет
- Introduccion Al Estado GaseosoДокумент4 страницыIntroduccion Al Estado GaseosoRDОценок пока нет
- Termodinamica Parte 2Документ11 страницTermodinamica Parte 2Verónica Elina RubelОценок пока нет
- Primera Ley de TermodinamicaДокумент70 страницPrimera Ley de TermodinamicaMANUEL ANGEL ROMERO HERNANDEZОценок пока нет
- Cuestionario Gases, Termoquímica y ElectroquímicaДокумент24 страницыCuestionario Gases, Termoquímica y ElectroquímicaEmily GonzalesОценок пока нет
- Propiedades Empíricas de Los Gase1Документ8 страницPropiedades Empíricas de Los Gase1juan alberto gareca guerreroОценок пока нет
- Sesiòn Nº14 Teorìa Cinètica de Los GasesДокумент38 страницSesiòn Nº14 Teorìa Cinètica de Los GasesArturo MartinezОценок пока нет
- Unidad 07-GasesДокумент19 страницUnidad 07-Gaseslucas baeОценок пока нет
- Problemario - Unidad - 5 Quimica AplicadaДокумент16 страницProblemario - Unidad - 5 Quimica AplicadauzieОценок пока нет
- Estado GaseosoДокумент13 страницEstado Gaseosocarlos torresОценок пока нет
- Teoria de GasesДокумент6 страницTeoria de GasesClaudia Mabel FloresОценок пока нет
- Seminario N°1 Gases Ideales y RealesДокумент6 страницSeminario N°1 Gases Ideales y Realesnikolle loayzaОценок пока нет
- Gases IdealesДокумент15 страницGases IdealesleslieОценок пока нет
- Estado gaseoso-RESUMIDOДокумент9 страницEstado gaseoso-RESUMIDOESplay YTОценок пока нет
- Instituto Tecnológico Nacional de México Campus Villahermosa Alumno: Catedratica: Asignatura: Carrera: Semestre: FechaДокумент23 страницыInstituto Tecnológico Nacional de México Campus Villahermosa Alumno: Catedratica: Asignatura: Carrera: Semestre: FechaEduardo NiñoОценок пока нет
- Tema 12Документ25 страницTema 12Mortal ResiduoОценок пока нет
- Ingeniería química. Soluciones a los problemas del tomo IОт EverandIngeniería química. Soluciones a los problemas del tomo IОценок пока нет
- Implementacion de Norma Iso 30301 en La UdecДокумент187 страницImplementacion de Norma Iso 30301 en La UdecAndres MohoОценок пока нет
- Carga y Descarga Del CondensadorДокумент10 страницCarga y Descarga Del CondensadorMax Jhon Arias BaspineiroОценок пока нет
- UMSS - WebSISS Sistema de Información San SimónДокумент3 страницыUMSS - WebSISS Sistema de Información San SimónMax Jhon Arias Baspineiro100% (1)
- Corolla PDFДокумент2 страницыCorolla PDFAgustin ColqueОценок пока нет
- Metodología de Planificación de La InvestigaciónДокумент88 страницMetodología de Planificación de La InvestigaciónMax Jhon Arias BaspineiroОценок пока нет
- Tejido Empresarial 1Документ28 страницTejido Empresarial 1Max Jhon Arias BaspineiroОценок пока нет
- Memoria Estado Nación 2012Документ508 страницMemoria Estado Nación 2012Max Jhon Arias BaspineiroОценок пока нет
- Formacion 2011 12 Cursos On LineДокумент112 страницFormacion 2011 12 Cursos On Linerodrigo_7_8100% (2)
- Montalk - Gnosis 2 - La Piedra FilosofalДокумент6 страницMontalk - Gnosis 2 - La Piedra FilosofalEdward LeviОценок пока нет
- Pirobloc Caldera de Fluido Termico PDFДокумент4 страницыPirobloc Caldera de Fluido Termico PDFPablo CoroОценок пока нет
- Q 5 Nivel Pamer UNIДокумент554 страницыQ 5 Nivel Pamer UNIAngelaОценок пока нет
- Transporte y Medición de Hidrocarburos-IPN PDFДокумент140 страницTransporte y Medición de Hidrocarburos-IPN PDFDamian100% (1)
- INFORME TÉCNICO #070 C FITSA Pontón Tucumana-Pomacocha - Con FirmaДокумент17 страницINFORME TÉCNICO #070 C FITSA Pontón Tucumana-Pomacocha - Con FirmaMary QuiñonezОценок пока нет
- Practica 1 - Secador de BandejasДокумент18 страницPractica 1 - Secador de BandejasMaria Judit Martinez Puerto100% (1)
- Aguan ContaminadaДокумент7 страницAguan ContaminadaJose Ramirez ramirezОценок пока нет
- CALDERASДокумент6 страницCALDERASJean Carlos QvisPeОценок пока нет
- Anexo 1. Descripción Estratigráfica Colegio Boston College Laguna Del Sol, Comuna de Padre HurtadoДокумент18 страницAnexo 1. Descripción Estratigráfica Colegio Boston College Laguna Del Sol, Comuna de Padre HurtadoJosé Ignacio J. Sarria MartínezОценок пока нет
- Quimica de Las ArcillasДокумент63 страницыQuimica de Las Arcillasrdos14100% (2)
- Plan de ContingenciasДокумент56 страницPlan de ContingenciasCinthya Araoz TejadaОценок пока нет
- Cómo Funciona Una Salinera y Cuál Es Su Imparto en El Medio AmbienteДокумент5 страницCómo Funciona Una Salinera y Cuál Es Su Imparto en El Medio AmbienteIm RauruОценок пока нет
- Proyecto Final Informe FinalДокумент9 страницProyecto Final Informe FinalPablo GragedaОценок пока нет
- Ensaye Al Fuego Analisis de OroДокумент39 страницEnsaye Al Fuego Analisis de OroVictor Alvarez100% (3)
- Analisis Uso de GLP Vs DieselДокумент68 страницAnalisis Uso de GLP Vs DieselEnrique MoralesОценок пока нет
- Indice de Desarrollo en Guatemala y CentroamericaДокумент18 страницIndice de Desarrollo en Guatemala y CentroamericaErika Tecú100% (1)
- Guia CelulaДокумент3 страницыGuia CelulaMiriam spulerОценок пока нет
- Dimensionamiento de Banco de Baterías-1Документ5 страницDimensionamiento de Banco de Baterías-1gilbertomjcОценок пока нет
- Laboratorio Termodinámica 2Документ9 страницLaboratorio Termodinámica 2Dagoberto Ramos0% (1)
- Trabajos en CalienteДокумент37 страницTrabajos en CalienteRafael Leguizamon RafaelОценок пока нет
- Estudio de Factibilidad para La Implementacion de Panales Solares en Los Montes de MariaДокумент125 страницEstudio de Factibilidad para La Implementacion de Panales Solares en Los Montes de MariaJUAN DE DIOS OBREGON - CEO EN LISIN EMPRESARIAL SASОценок пока нет
- El Andar Como Practica Estetica 1Документ5 страницEl Andar Como Practica Estetica 1Esther RamirezОценок пока нет
- Acacerbo - 5-Modelos de Depositos MineralesДокумент35 страницAcacerbo - 5-Modelos de Depositos MineralesKevin JaimesОценок пока нет
- Laboratorio Transición - JUNIOДокумент2 страницыLaboratorio Transición - JUNIOTransicion SebastianoОценок пока нет
- Estudio Geologico y GeotecnicoДокумент17 страницEstudio Geologico y GeotecnicoGonzalo Ylich Villagra GarciaОценок пока нет
- BMP LomparteДокумент124 страницыBMP LomparteDaniel SantosОценок пока нет
- Prontuario Del AceroДокумент21 страницаProntuario Del AceroRolando Flores IngalaОценок пока нет
- RRHH-P-05 Ver 01 Procedimiento de Prevención y Sanción Del Hostigamiento Sexual LaboralДокумент9 страницRRHH-P-05 Ver 01 Procedimiento de Prevención y Sanción Del Hostigamiento Sexual Laboralnelida ramos floresОценок пока нет