Академический Документы
Профессиональный Документы
Культура Документы
Gynaecological Cancer Chemo Protocols
Загружено:
am3l38Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Gynaecological Cancer Chemo Protocols
Загружено:
am3l38Авторское право:
Доступные форматы
Department of Medical Oncology
Chemotherapy Protocols
Protocol: Indications: Schedule: Drug Paclitaxel Carboplatin
Carboplatin/Paclitaxel Ovarian Cancer Adjuvant, Advanced Dose 175mg/m2 AUC 5 Every three weeks iv/infusion/oral 500mls 5% dex/3hrs 500mls 5% dex/1hr q Day 1 Day 1 6
Cycle frequency:
Total number of cycles:
Dose modifications: Discuss with Consultant Administration and safety: Anti-emetic group - Moderately high Delay if neutrophils < 1.5 x 109/L or platelets < 100 x 109/L Paclitaxel given first Pre-medication with dexamethasone (oral or iv), chlorpheniramine and ranitidine Carboplatin dose by EDTA or creatinine clearance. If calculated using formula then AUC 6 Toxicities: Myelosuppression and risk of neutropenic sepsis or haemorrhage, nausea & vomiting, mucositis, alopecia, amenorrhoea, peripheral neuropathy, fluid retention, hypersensitivity reaction, abdominal discomfort, infertility Symptomatic treatment of side effects: Mouth care, diuretics Investigations Pre-treatment: History and Examination Performance score, weight FBC U & Es, LFTs, creatinine, urate, creatinine clearance LDH, CA125 ECG Staging investigations as per protocol Prior to each cycle: Performance score, weight FBC U & Es, LFTs, creatinine LDH, CA125 Mid Treatment: Post Treatment: Reference: Abdominal CT scan prior to fourth cycle if measurable disease present. Ensure CA125 falling. Review in Medical Oncology Clinic 4 weeks after last cycle MRC ICON 3, 2002. Lancet, 360; pages 505-515
3rd Edition
55
Department of Medical Oncology
Chemotherapy Protocols
Protocol: Indications: Schedule: Drug Paclitaxel
Paclitaxel Ovarian Cancer Recurrent Dose 175mg/m2 iv/infusion/oral 500mls 5% dex/3hrs q Day 1 6
Cycle frequency: Every three weeks Dose modifications: Discuss with Consultant
Total number of cycles:
Administration and safety: Anti-emetic group Low Delay if neutrophils < 1.5 x 109/L or platelets < 100 x 109/L Pre-medication with dexamethasone (oral or iv), chlorpheniramine and ranitidine Toxicities: Myelosuppression and risk of neutropenic sepsis or haemorrhage, nausea & vomiting, mucositis, alopecia, amenorrhoea, peripheral neuropathy, fluid retention, hypersensitivity reaction, abdominal discomfort, infertility Symptomatic treatment of side effects: Mouth care, diuretics Investigations Pre-treatment: History and Examination Performance score, weight FBC U & Es, LFTs, creatinine, urate, creatinine clearance LDH, CA125 ECG Staging investigations as per protocol Prior to each cycle: Performance score, weight FBC U & Es, LFTs, creatinine LDH, CA125 Mid Treatment: Post Treatment: Reference: Ensure CA125 falling Review in Medical Oncology Clinic 4 weeks after last cycle Gore et al, 1997. J. Clin. Oncol., 15; pages 2183-2193
3rd Edition
56
Department of Medical Oncology
Chemotherapy Protocols
Protocol: Indications: Schedule: Drug Carboplatin
Carboplatin Ovarian Cancer Advanced, Recurrent Dose AUC 6 Every three weeks iv/infusion/oral 500mls 5% dex/1hr q Day 1 6
Cycle frequency:
Total number of cycles:
Dose modifications: Discuss with Consultant Administration and safety: Anti-emetic group - Moderately high Delay if neutrophils < 1.5 x 109/L or platelets < 100 x 109/L Carboplatin dose by EDTA or creatinine clearance. If calculated using formula then AUC 7 Toxicities: Myelosuppression and risk of neutropenic sepsis or haemorrhage, nausea & vomiting Symptomatic treatment of side effects: Mouth care Investigations Pre-treatment: History and Examination Performance score, weight FBC U & Es, LFTs, creatinine, urate, creatinine clearance LDH, CA125 ECG Staging investigations as per protocol Prior to each cycle: Performance score, weight FBC U & Es, LFTs, creatinine LDH, CA125 Mid Treatment: Post Treatment: Reference: Abdominal CT scan prior to 4th cycle if measurable disease present. Ensure CA125 falling Review in Medical Oncology Clinic 4 weeks after last cycle Taylor et al, 1994. J. Clin. Oncol., 12; pages 2066-2070
3rd Edition
57
Department of Medical Oncology
Chemotherapy Protocols
Protocol: Indications: Schedule: Drug Paclitaxel Cisplatin
Cisplatin/Paclitaxel Ovarian Cancer Adjuvant Dose 175mg/m2 75mg/m2 Every three weeks iv/infusion/oral 500mls 5% dex/3hrs 1L N. Saline/2hrs q Day 1 Day 1 6
Cycle frequency:
Total number of cycles:
Dose modifications: Discuss with Consultant Administration and safety: Anti-emetic group - High Delay if neutrophils < 1.5 x 109/L or platelets < 100 x 109/L Paclitaxel given first Pre-medication with dexamethasone (oral or iv), chlorpheniramine and ranitidine Pre & post hydration, mannitol, potassium & magnesium Toxicities: Myelosuppression and risk of neutropenic sepsis or haemorrhage, nausea & vomiting, mucositis, alopecia, amenorrhoea, peripheral neuropathy, hypersensitivity reaction, fluid retention, ototoxicity, constipation, nephrotoxicity, infertility Symptomatic treatment of side effects: Mouth care, encourage oral fluids Investigations Pre-treatment: History and Examination Performance score, weight FBC U & Es, LFTs, Mg2+, Ca2+, creatinine, urate, creatinine clearance LDH, CA125 ECG +/- Audiometry Staging investigations as per protocol Prior to each cycle: Performance score, weight FBC U & Es, LFTs, Mg2+, Ca2+, creatinine LDH, CA125 Mid Treatment: Post Treatment: Reference: Abdominal CT scan prior to fourth cycle if measurable disease present. Ensure CA125 falling Review in Medical Oncology Clinic 4 weeks after last cycle Neijt et al, 2000. J. Clin. Oncol., 18; pages 3084-3092
3rd Edition
58
Department of Medical Oncology
Chemotherapy Protocols
Protocol: Indications:
Liposomal Doxorubicin Ovarian Cancer Recurrent iv/infusion/oral 250mls 5% dex/1hr q Day 1 6-12
Schedule: Drug Dose Liposomal Doxorubicin 50mg/m2 Cycle frequency: Every 4 weeks
Total number of cycles:
Dose modifications: Discuss with consultant Administration and safety: Anti-emetic group - Moderate Delay if neutrophils < 1.5 x 109/L or platelets < 100 x 109/L Do not retreat unless all previous skin lesions resolved Toxicities: Myelosuppression and risk of neutropenic sepsis or haemorrhage, palmarplantar syndrome, nausea & vomiting, mucositis, cardiotoxicity, alopecia, amenorrhoea, peripheral neuropathy, carcinogenesis, infertility Symptomatic treatment of side effects: Mouth care, advice regarding palmar-plantar syndrome Investigations Pre-treatment: History and Examination Performance score, weight FBC U & Es, LFTs, creatinine, urate LDH, CA125 ECG +/- Echocardiogram Staging investigations as per protocol Prior to each cycle: Performance score, weight FBC U & Es, LFTs, creatinine LDH, CA125 Mid Treatment: Post Treatment: Reference: Abdominal CT scan prior to 4th cycle if measurable disease present. Ensure CA125 falling Review in Medical Oncology Clinic 4 weeks after last cycle Gordon et al, 2000. J. Clin. Oncol., 18; pages 3093-3100
3rd Edition
59
Department of Medical Oncology
Chemotherapy Protocols
Protocol: Indications: Schedule: Drug Cisplatin Methotrexate Bleomycin
PMB (Cisplatin/Methotrexate/Bleomycin) Cervix Cancer Advanced, Recurrent Dose 60mg/m2 300mg/m2 30,000iu Every 2 weeks iv/infusion/oral 1L N. Saline/1hr 1L N. Saline/12hrs 200mls N. Saline/30mins q Day 2 Day 1 Day 1 3
Cycle frequency:
Total number of cycles:
Dose modifications: Discuss with Consultant Administration and safety: Anti-emetic group High Delay if neutrophils < 1.5 x 109/L or platelets < 100 x 109/L Ensure alkalinisation of urine Pre & post hydration, mannitol, potassium & magnesium Calcium Folinate rescue 24 hours after methotrexate infusion Toxicities: Myelosuppression and risk of neutropenic sepsis or haemorrhage, nausea & vomiting, mucositis, mild alopecia, amenorrhoea, peripheral neuropathy, nephrotoxicity, ototoxicity, pneumonitis, carcinogenesis, infertility Symptomatic treatment of side effects: Mouth care, encourage oral fluids Investigations Pre-treatment: History and Examination Performance score, weight FBC U & Es, LFTs, Mg2+, Ca2+, creatinine, urate, creatinine clearance LDH ECG+/- Audiometry Staging investigations as per protocol Prior to each cycle: Performance score, weight FBC U & Es, LFTs, Mg2+, Ca2+, creatinine LDH Mid Treatment: Post Treatment: Reference: Re-assess after 3 cycles Review in Medical Oncology Clinic 4 weeks after last cycle Huskin and Blake, 1991. Int. J. Gynaecol. Oncol., 1; pages 75-80
3rd Edition
60
Department of Medical Oncology
Chemotherapy Protocols
Protocol: Indications: Schedule: Drug Cisplatin 5-Fluorouracil
CF (Cisplatin/5-Fluorouracil) Cervix Cancer Neoadjuvant, Advanced Dose 75mg/m2 1g/m2/24 hrs Every three weeks iv/infusion/oral 1L N. Saline/2hrs continuous infusion q Day 1 Days 2-5 3
Cycle frequency:
Total number of cycles:
Dose modifications: Discuss with Consultant Administration and safety: Anti-emetic group High Delay if neutrophils < 1.5 x 109/L or platelets < 100 x 109/L Ensure adequate renal function Pre & post hydration, mannitol, potassium & magnesium Concurrent radiotherapy may be necessary Doses may change according to Radiotherapy schedule Toxicities: Myelosuppression and risk of neutropenic sepsis or haemorrhage, nausea & vomiting, mucositis, alopecia, cardiotoxicity, peripheral neuropathy, palmar-plantar syndrome, nephrotoxicity, ototoxicity, diarrhoea, carcinogenesis, infertility Symptomatic treatment of side effects: Mouth care, anti-diarrheals, pyridoxine Investigations Pre-treatment: History and Examination Performance score, weight FBC U & Es, LFTs, Mg2+, Ca2+, creatinine, urate, creatinine clearance LDH ECG Staging investigations as per protocol Prior to each cycle: Performance score, weight FBC U & Es, LFTs, Mg2+, Ca2+, creatinine LDH Post Treatment: Reference: Review in Medical Oncology Clinic 3 weeks after last cycle Morris et al, 1999. N. Engl. J. Med., 340; pages 1137-1143
3rd Edition
61
Вам также может понравиться
- The Yellow House: A Memoir (2019 National Book Award Winner)От EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Рейтинг: 4 из 5 звезд4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeОт EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeРейтинг: 4 из 5 звезд4/5 (5795)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureОт EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureРейтинг: 4.5 из 5 звезд4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryОт EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryРейтинг: 3.5 из 5 звезд3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceОт EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceРейтинг: 4 из 5 звезд4/5 (895)
- Never Split the Difference: Negotiating As If Your Life Depended On ItОт EverandNever Split the Difference: Negotiating As If Your Life Depended On ItРейтинг: 4.5 из 5 звезд4.5/5 (838)
- The Little Book of Hygge: Danish Secrets to Happy LivingОт EverandThe Little Book of Hygge: Danish Secrets to Happy LivingРейтинг: 3.5 из 5 звезд3.5/5 (400)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersОт EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersРейтинг: 4.5 из 5 звезд4.5/5 (345)
- The Unwinding: An Inner History of the New AmericaОт EverandThe Unwinding: An Inner History of the New AmericaРейтинг: 4 из 5 звезд4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnОт EverandTeam of Rivals: The Political Genius of Abraham LincolnРейтинг: 4.5 из 5 звезд4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyОт EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyРейтинг: 3.5 из 5 звезд3.5/5 (2259)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaОт EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaРейтинг: 4.5 из 5 звезд4.5/5 (266)
- The Emperor of All Maladies: A Biography of CancerОт EverandThe Emperor of All Maladies: A Biography of CancerРейтинг: 4.5 из 5 звезд4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreОт EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreРейтинг: 4 из 5 звезд4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)От EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Рейтинг: 4.5 из 5 звезд4.5/5 (121)
- M Sandamela Salivary Gland SandamelaДокумент45 страницM Sandamela Salivary Gland SandamelaRockson SamuelОценок пока нет
- Granulomatous Mastitis: The Histological Differentials: Click To Edit Master Subtitle StyleДокумент13 страницGranulomatous Mastitis: The Histological Differentials: Click To Edit Master Subtitle StyleAsmara SyedОценок пока нет
- Diagnostic and Therapeutic Nuclear Medicine For Neuroendocrine TumorsДокумент483 страницыDiagnostic and Therapeutic Nuclear Medicine For Neuroendocrine TumorsIulia Alexandra Strugari100% (1)
- Surgical Management For Lung CancerДокумент45 страницSurgical Management For Lung CancerarifgteguhОценок пока нет
- Brain TumorДокумент7 страницBrain TumorPintu Kumar100% (1)
- Practice Bulletin: Management of Adnexal MassesДокумент14 страницPractice Bulletin: Management of Adnexal MassesDasha VeeОценок пока нет
- Practical 7 Dysplasia Carcinogen Es IsДокумент19 страницPractical 7 Dysplasia Carcinogen Es IsRujha Haniena Ahmad RidzuanОценок пока нет
- Globocan 2020Документ2 страницыGlobocan 2020alfred benedict bayanОценок пока нет
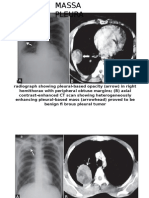
- Massa PleuraДокумент4 страницыMassa PleuraPrayogi KramyОценок пока нет
- Cancer PDFДокумент18 страницCancer PDFAhmed goldenОценок пока нет
- Adnexal Tumors of The SkinДокумент6 страницAdnexal Tumors of The SkinFaduahSalazarОценок пока нет
- Cervical CAДокумент28 страницCervical CABeb Raquel GamiaoОценок пока нет
- Association Subungual Abscess and Pyogenic Granuloma Secondary To Docetaxel TherapyДокумент2 страницыAssociation Subungual Abscess and Pyogenic Granuloma Secondary To Docetaxel TherapyasclepiuspdfsОценок пока нет
- Autophagy and Intermittent Fasting The Connection For Cancer TherapyДокумент7 страницAutophagy and Intermittent Fasting The Connection For Cancer TherapyYoBjjОценок пока нет
- Malignant Pleural Effusion PrevalenceДокумент72 страницыMalignant Pleural Effusion PrevalenceAkbar SyarialОценок пока нет
- NonMelanocytic Cutaneous Neoplasm 2021Документ101 страницаNonMelanocytic Cutaneous Neoplasm 2021karimahihdaОценок пока нет
- Urology Quiz 9 MCQ AnswerДокумент2 страницыUrology Quiz 9 MCQ Answeruroquiz100% (2)
- (Current Cancer Research) Erle S. Robertson - Microbiome and Cancer-Springe PDFДокумент406 страниц(Current Cancer Research) Erle S. Robertson - Microbiome and Cancer-Springe PDFmariomgОценок пока нет
- NLR To Ovarian CancerДокумент8 страницNLR To Ovarian CancersririniaОценок пока нет
- Tamoxifen DrugstudyДокумент2 страницыTamoxifen Drugstudyjessica queenОценок пока нет
- Association of Human Papilloma Virus in Oral Squamous Cell CarcinomaДокумент7 страницAssociation of Human Papilloma Virus in Oral Squamous Cell CarcinomaFabi SamadОценок пока нет
- Pathology Course Audit-3Документ26 страницPathology Course Audit-3Joana Marie PalatanОценок пока нет
- Uterus 4.3.0.0.rel CapcpДокумент24 страницыUterus 4.3.0.0.rel CapcpMARINA SILVA DE ARAUJOОценок пока нет
- Musculoskeletal Cancer Surgery - Malawer PDFДокумент592 страницыMusculoskeletal Cancer Surgery - Malawer PDFanggita100% (1)
- LP Brain TumorДокумент20 страницLP Brain TumorMuhammad PanduОценок пока нет
- Cancer Country Profile Indonesia-2020Документ2 страницыCancer Country Profile Indonesia-2020Dwi AndayaniОценок пока нет
- Cellular Aberration Course Outline2013Документ4 страницыCellular Aberration Course Outline2013Kaycee TolingОценок пока нет
- Triple Negative Breast Cancer Case PresentationДокумент3 страницыTriple Negative Breast Cancer Case PresentationNiloy ChatterjeeОценок пока нет
- Primary Malignant Bone Tumor Chondrosarcoma of The Sternum I Ketut Suyasa, Gede Ketut Alit Satria Nugraha, I Gede Eka WiratnayaДокумент5 страницPrimary Malignant Bone Tumor Chondrosarcoma of The Sternum I Ketut Suyasa, Gede Ketut Alit Satria Nugraha, I Gede Eka WiratnayaIkram BachtiarОценок пока нет
- Brain Cancer ThesisДокумент7 страницBrain Cancer ThesisKristen Flores100% (2)