Академический Документы
Профессиональный Документы
Культура Документы
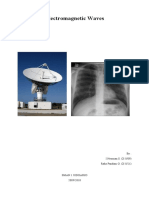
Electromagnetic Radiation (EM Radiation or EMR) Is A Fundamental Phenomenon of
Загружено:
joejoe4321joeОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Electromagnetic Radiation (EM Radiation or EMR) Is A Fundamental Phenomenon of
Загружено:
joejoe4321joeАвторское право:
Доступные форматы
Electromagnetic radiation (EM radiation or EMR) is a fundamental phenomenon of
electromagnetism, behaving as waves and also as particles called photons which travel through
space carrying radiant energy. In a vacuum, it propagates at the speed of light, normally in
straight lines. EMR is emitted and absorbed by charged particles. As an electromagnetic wave, it
has both electric and magnetic field components, which synchronously oscillate perpendicular to
each other and perpendicular to the direction of energy and wave propagation.
In classical physics, EMR is produced when charged particles are accelerated by forces acting on
them. Electrons are responsible for emission of most EMR because they have low mass, and
therefore are easily accelerated by a variety of mechanisms. Quantum processes can also produce
EMR, such as when atomic nuclei undergo gamma decay, and processes such as neutral pion
decay.
EMR carries energysometimes called radiant energythrough space continuously away from
the source (this is not true of the near-field part of the EM field). EMR also carries both
momentum and angular momentum. These properties may all be imparted to matter with which
it interacts. When created, EMR is produced from other types of energy and it is converted to
other types of energy when it is destroyed.
The electromagnetic spectrum, in order of increasing frequency and decreasing wavelength, can
be divided, for practical engineering purposes, into radio waves, microwaves, infrared radiation,
visible light, ultraviolet radiation, X-rays and gamma rays. The eyes of various organisms sense
a relatively small range of frequencies of EMR near and including the visible spectrum or light.
Visible light is that part of the spectrum to which human eyes respond. Higher frequencies
(shorter wavelengths) have more energy in the photons, according to the well-known law E=h,
where E is the energy per photon, is the frequency carried by the photon, and h is Planck's
constant. A single gamma ray photon carries far more energy than a single photon of visible
light.
The photon is the quantum of the electromagnetic interaction, and is the basic constituent of all
forms of EMR. The quantum nature of light becomes more apparent at high frequencies (thus
high photon energy). Such photons behave more like particles than lower-frequency photons do.
Electromagnetic waves in free space must be solutions of Maxwell's electromagnetic wave
equation. Two main classes of solutions are known, namely plane waves and spherical waves.
The plane waves may be viewed as the limiting case of spherical waves at a very large (ideally
infinite) distance from the source. Both types of waves can have a waveform which is an
arbitrary time function (so long as it is sufficiently differentiable to conform to the wave
equation). As with any time function, this can be decomposed by means of Fourier analysis into
its frequency spectrum, or individual sinusoidal components, each of which contains a single
frequency, amplitude, and phase. Such a component wave is said to be monochromatic. A
monochromatic electromagnetic wave can be characterized by its frequency or wavelength, its
peak amplitude, its phase relative to some reference phase, its direction of propagation, and its
polarization.
Electromagnetic radiation is associated with EM fields that are free to propagate themselves
without the continuing influence of the moving charges that produced them, because they have
achieved sufficient distance from those charges. Thus, EMR is sometimes referred to as the far
field. In this language, the near field refers to EM fields near the charges and current that directly
produced them, as for example with simple magnets and static electricity phenomena. In EMR,
the magnetic and electric fields are each induced by changes in the other type of field, thus
propagating itself as a wave. This close relationship assures that both types of fields in EMR
stand in phase and in a fixed ratio of intensity to each other, with maxima and nodes in each
found at the same places in space.
The effects of EMR upon biological systems (and also to many other chemical systems, under
standard conditions) depend both upon the radiation's power and frequency. For lower
frequencies of EMR up to those of visible light (i.e., radio, microwave, infrared), the damage
done to cells and also to many ordinary materials under such conditions is determined mainly by
heating effects, and thus by the radiation power. By contrast, for higher frequency radiations at
ultraviolet frequencies and above (i.e., X-rays and gamma rays) the damage to chemical
materials and living cells by EMR is far larger than that done by simple heating, due to the
ability of single photons in such high frequency EMR to damage individual molecules
chemically.
Вам также может понравиться
- Electromagnetic Waves: By: I Nyoman S. (X-8/09) Raka Pandam G. (X-8/21)Документ11 страницElectromagnetic Waves: By: I Nyoman S. (X-8/09) Raka Pandam G. (X-8/21)Nyoman SuryadinataОценок пока нет
- Electro Magnetic RadiationДокумент48 страницElectro Magnetic RadiationDeb BasuОценок пока нет
- Unified Field Theory in a Nutshell1: The Quest for the Theory of EverythingОт EverandUnified Field Theory in a Nutshell1: The Quest for the Theory of EverythingОценок пока нет
- Nicolas-Lewis vs. ComelecДокумент3 страницыNicolas-Lewis vs. ComelecJessamine OrioqueОценок пока нет
- Shore Activities and Detachments Under The Command of Secretary of Navy and Chief of Naval OperationsДокумент53 страницыShore Activities and Detachments Under The Command of Secretary of Navy and Chief of Naval OperationskarakogluОценок пока нет
- Maxwell's Equations and the Principles of Electromagnetic PhenomenaОт EverandMaxwell's Equations and the Principles of Electromagnetic PhenomenaОценок пока нет
- ELECTROMAGNETIC FIELDS: Exposure is not optional, no one can avoid itОт EverandELECTROMAGNETIC FIELDS: Exposure is not optional, no one can avoid itОценок пока нет
- Electromagnetic Waves: Description: Electromagnetic Waves Are Formed When An Electric Field ComesДокумент14 страницElectromagnetic Waves: Description: Electromagnetic Waves Are Formed When An Electric Field Comesj_sachin09Оценок пока нет
- Integrating GrammarДокумент8 страницIntegrating GrammarMaría Perez CastañoОценок пока нет
- Richard Herrmann-Fractional Calculus - An Introduction For Physicists-World Scientific (2011)Документ274 страницыRichard Herrmann-Fractional Calculus - An Introduction For Physicists-World Scientific (2011)Juan Manuel ContrerasОценок пока нет
- Budokon - Mma.program 2012 13Документ10 страницBudokon - Mma.program 2012 13Emilio DiazОценок пока нет
- Course Syllabus (NGCM 112)Документ29 страницCourse Syllabus (NGCM 112)Marie Ashley Casia100% (1)
- Electromagnetic SpectrumДокумент17 страницElectromagnetic SpectrumAviation/Space History Library100% (4)
- ML Performance Improvement CheatsheetДокумент11 страницML Performance Improvement Cheatsheetrahulsukhija100% (1)
- Electromagnetic Radiation: Electromagnetic Radiation (EM Radiation or EMR) Is A Form of EnergyДокумент21 страницаElectromagnetic Radiation: Electromagnetic Radiation (EM Radiation or EMR) Is A Form of EnergybekkuОценок пока нет
- Analyzing Evidence of College Readiness: A Tri-Level Empirical & Conceptual FrameworkДокумент66 страницAnalyzing Evidence of College Readiness: A Tri-Level Empirical & Conceptual FrameworkJinky RegonayОценок пока нет
- SpectrosДокумент15 страницSpectrosNarmada BОценок пока нет
- Electromagnetic RadiationДокумент22 страницыElectromagnetic RadiationpriyaОценок пока нет
- Electromagnetic Radiation - Wikipedia, The Free EncyclopediaДокумент19 страницElectromagnetic Radiation - Wikipedia, The Free EncyclopediaDivy AgarwalОценок пока нет
- Electromagnetic RadiationДокумент5 страницElectromagnetic Radiationprateek_goyal2091Оценок пока нет
- Project ReportДокумент30 страницProject ReportShyam Kumar RastogiОценок пока нет
- hν, where E is the energy per ν is the frequency of the photon, and h isДокумент1 страницаhν, where E is the energy per ν is the frequency of the photon, and h isSaad AhsanОценок пока нет
- Electromagnetic RadiationДокумент7 страницElectromagnetic RadiationKevinОценок пока нет
- Electromagnetic RadiationДокумент21 страницаElectromagnetic RadiationMokesh KОценок пока нет
- Electromagnetic Radiation - Wikipedia PDFДокумент123 страницыElectromagnetic Radiation - Wikipedia PDFJaneОценок пока нет
- Electromagnetic Radiation 2016Документ22 страницыElectromagnetic Radiation 2016MilisavDjerkovicОценок пока нет
- Electromagnetic Radiation Observations: Electromagnetic Radiation (EM Radiation or EMR) Refers To The Waves (OrДокумент10 страницElectromagnetic Radiation Observations: Electromagnetic Radiation (EM Radiation or EMR) Refers To The Waves (OrYancy MiclatОценок пока нет
- Electromagnetic RadiationДокумент2 страницыElectromagnetic RadiationAman JyotiОценок пока нет
- 1.1 Radiation: Interaction of Radiation With MatterДокумент49 страниц1.1 Radiation: Interaction of Radiation With MatterShiva KumarОценок пока нет
- Physics: TheoryДокумент5 страницPhysics: TheoryIrshad KhanОценок пока нет
- Spectroscopy: Electromagnetic RadiationДокумент19 страницSpectroscopy: Electromagnetic RadiationPriyanka SharmaОценок пока нет
- SCY1612Документ59 страницSCY1612rieОценок пока нет
- Light As An Electromagnetic Wave: General Physics IIДокумент16 страницLight As An Electromagnetic Wave: General Physics IIDarious PenillaОценок пока нет
- Term PaperДокумент12 страницTerm PaperJunaid Mushtaq100% (1)
- CYN002 Spectroscopy Introduction Class2 04042022 Send StudentsДокумент24 страницыCYN002 Spectroscopy Introduction Class2 04042022 Send StudentsAYUSH KISHOREОценок пока нет
- Week 1Документ4 страницыWeek 1jia aganaОценок пока нет
- EMFT Assignment 5Документ48 страницEMFT Assignment 5M. HamzaОценок пока нет
- 005 - ManMadeRadiation (2018)Документ7 страниц005 - ManMadeRadiation (2018)Bert AndersonОценок пока нет
- Remote Sensing 02 The Electromagnetic Spectrum 1Документ14 страницRemote Sensing 02 The Electromagnetic Spectrum 1Rea FloresОценок пока нет
- Electromagnetic RadiationДокумент3 страницыElectromagnetic Radiationpeacekalman63Оценок пока нет
- Electromagnetic RadiationДокумент3 страницыElectromagnetic RadiationBiswajit MohantyОценок пока нет
- Science Reviewer QuizДокумент4 страницыScience Reviewer Quiz28. Pinera, Yna Angela S.Оценок пока нет
- Report SpectrosДокумент4 страницыReport SpectrosFff FffОценок пока нет
- Source of ColorДокумент6 страницSource of ColoryemiОценок пока нет
- Physics ProjectДокумент5 страницPhysics ProjectSHREYANSH SHUKLAОценок пока нет
- Applications of EM FieldsДокумент2 страницыApplications of EM Fieldsalokesh1982Оценок пока нет
- Ijiset - V5 - I12 - 01 (Mri)Документ20 страницIjiset - V5 - I12 - 01 (Mri)Ivan CheahОценок пока нет
- Unidad 3 - Espectro Electromagnético (Inglés)Документ32 страницыUnidad 3 - Espectro Electromagnético (Inglés)bemdasОценок пока нет
- Bus BarДокумент11 страницBus BarDawit Adane KebedeОценок пока нет
- Biological Effects of Electromagnetic Fields On Human's HealthДокумент18 страницBiological Effects of Electromagnetic Fields On Human's HealthMarina ZaraОценок пока нет
- Sunlight:: Sunlight Is A Portion of TheДокумент8 страницSunlight:: Sunlight Is A Portion of TheApu NayemОценок пока нет
- Electromagnetic EnergyДокумент8 страницElectromagnetic Energyedsonmamaradlo23Оценок пока нет
- Description: Electromagnetic Radiation Charged ParticlesДокумент8 страницDescription: Electromagnetic Radiation Charged ParticlesfaithОценок пока нет
- Q2 Science Reviewer 1Документ2 страницыQ2 Science Reviewer 1Jiann AguilarОценок пока нет
- Electromagnetic RadiationДокумент6 страницElectromagnetic Radiation09ANKIT MISHRAОценок пока нет
- Handout 02Документ10 страницHandout 02JAMESОценок пока нет
- History: Spectrum IsДокумент23 страницыHistory: Spectrum IsMuhamad NurhudayahОценок пока нет
- EM Radiation PropertiesДокумент3 страницыEM Radiation PropertiesSundayОценок пока нет
- Module 2 Week 3 4Документ9 страницModule 2 Week 3 4LeiRne Jo KBahingawan TanGuidОценок пока нет
- LightДокумент21 страницаLightSri RizkiОценок пока нет
- 2nd Quarter Reviewer in ScienceДокумент3 страницы2nd Quarter Reviewer in Sciencecali annaОценок пока нет
- Science Reviewer 1Документ15 страницScience Reviewer 1Symon MarianoОценок пока нет
- Electromagnetic Waves (EM Waves)Документ17 страницElectromagnetic Waves (EM Waves)Joshdave cuambotОценок пока нет
- Chapter 8Документ2 страницыChapter 8LoL CluBОценок пока нет
- Properties of LasersДокумент15 страницProperties of LasersTeja NandamuriОценок пока нет
- What is ElectromagnetismДокумент16 страницWhat is ElectromagnetismFelyzia NiamhОценок пока нет
- Grade 10 ReviewerДокумент6 страницGrade 10 ReviewerDino M PangilinanОценок пока нет
- Electromagnetic RadiationДокумент35 страницElectromagnetic RadiationJeff GicharuОценок пока нет
- Mpeg-1 11172-1Документ46 страницMpeg-1 11172-1Hana HoubaОценок пока нет
- Earth-Song WorksheetДокумент2 страницыEarth-Song WorksheetMuhammad FarizОценок пока нет
- Module 3 in Oral Com 1Документ20 страницModule 3 in Oral Com 1Trisha DiohenОценок пока нет
- Identifying States of Matter LessonДокумент2 страницыIdentifying States of Matter LessonRaul OrcigaОценок пока нет
- Unit 11 LeadershipДокумент4 страницыUnit 11 LeadershipMarijana DragašОценок пока нет
- Jobgpt 9d48h0joДокумент6 страницJobgpt 9d48h0jomaijel CancinesОценок пока нет
- James A. Mcnamara JR.: An Interview WithДокумент22 страницыJames A. Mcnamara JR.: An Interview WithMiguel candelaОценок пока нет
- Pyrolysis: Mathematical Modeling of Hydrocarbon Pyrolysis ReactionsДокумент8 страницPyrolysis: Mathematical Modeling of Hydrocarbon Pyrolysis ReactionsBahar MeschiОценок пока нет
- Modbus Quick StartДокумент3 страницыModbus Quick StartNash JungОценок пока нет
- Rak Single DentureДокумент48 страницRak Single Denturerakes0Оценок пока нет
- Absolute TowersДокумент11 страницAbsolute TowersSandi Harlan100% (1)
- DU - BSC (H) CS BookletДокумент121 страницаDU - BSC (H) CS BookletNagendra DuhanОценок пока нет
- CV Jan 2015 SДокумент4 страницыCV Jan 2015 Sapi-276142935Оценок пока нет
- Detect Organic Elements with Sodium FusionДокумент10 страницDetect Organic Elements with Sodium FusionMukundОценок пока нет
- Us Aers Roadmap Noncontrolling Interest 2019 PDFДокумент194 страницыUs Aers Roadmap Noncontrolling Interest 2019 PDFUlii PntОценок пока нет
- Veerabhadra Swamy MantrasДокумент6 страницVeerabhadra Swamy Mantrasगणेश पराजुलीОценок пока нет
- Republic v. EncelanДокумент2 страницыRepublic v. EncelanKyla ReyesОценок пока нет
- New GK PDFДокумент3 страницыNew GK PDFkbkwebsОценок пока нет
- The Great Idea of Brook TaylorДокумент7 страницThe Great Idea of Brook TaylorGeorge Mpantes mathematics teacherОценок пока нет
- SOW Form 4 2017Документ8 страницSOW Form 4 2017ismarizalОценок пока нет
- United States v. Christopher King, 724 F.2d 253, 1st Cir. (1984)Документ9 страницUnited States v. Christopher King, 724 F.2d 253, 1st Cir. (1984)Scribd Government DocsОценок пока нет
- Bunga Refira - 1830104008 - Allophonic RulesДокумент6 страницBunga Refira - 1830104008 - Allophonic RulesBunga RefiraОценок пока нет