Академический Документы
Профессиональный Документы
Культура Документы
Gas Laws Packet 2 ANSWERS
Загружено:
Joseph Turner100%(1)100% нашли этот документ полезным (1 голос)
6K просмотров5 страницgas laws answers
Авторское право
© © All Rights Reserved
Доступные форматы
DOC, PDF, TXT или читайте онлайн в Scribd
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документgas laws answers
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате DOC, PDF, TXT или читайте онлайн в Scribd
100%(1)100% нашли этот документ полезным (1 голос)
6K просмотров5 страницGas Laws Packet 2 ANSWERS
Загружено:
Joseph Turnergas laws answers
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате DOC, PDF, TXT или читайте онлайн в Scribd
Вы находитесь на странице: 1из 5
Gas Laws Packet #2
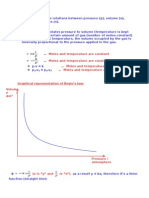
Ideal Gas Law Worksheet PV = nRT
Use the ideal gas law, PerV-nRT, and the universal gas constant R = 0.0821 L*atm
to solve the following problems: K*mol
If pressure is needed in Pa then convert b! multipl!ing b! 101.3kPa / 1atm to get
R =8.31 L*kPa / (K*mole)
"# $f $ have % moles of a gas at a pressure of &'( atm and a volume of ") liters, what
is the temperature*
204.6 K
)# $f $ have an unnown +uantit! of gas at a pressure of "') atm, a volume of ,"
liters, and a temperature of -.
/
0, how man! moles of gas do $ have*
1.26 ol
,# $f $ contain , moles of gas in a container with a volume of (/ liters and at a
temperature of %// 1, what is the pressure inside the container*
1.64 at
%# $f $ have .'. moles of gas at a pressure of /'/2 atm and at a temperature of &(
/
0,
what is the volume of the container that the gas is in*
2!11 L
&# $f $ have ". moles of gas at a temperature of (.
/
0, and a volume of --'-2 liters,
what is the pressure of the gas*
".4 at or "40 kPa
(# $f $ have an unnown +uantit! of gas at a pressure of /'& atm, a volume of )&
liters, and a temperature of ,// 1, how man! moles of gas do $ have*
." ol
For chemistry help, visit www.chemfiesta.com 2000 Cavalcade Publishing - ll !ights !eserved
.# $f $ have )" moles of gas held at a pressure of .- atm and a temperature of
2// 1, what is the volume of the gas*
1#.$ L
-# $f $ have "'2 moles of gas held at a pressure of & atm and in a container with a
volume of &/ liters, what is the temperature of the gas*
1602 K
2# $f $ have )'% moles of gas held at a temperature of 2.
/
0 and in a container with a
volume of %& liters, what is the pressure of the gas*
1.62 at or 164 kPa
"/# $f $ have an unnown +uantit! of gas held at a temperature of ""2& 1 in a
container with a volume of )& liters and a pressure of &(/ atm, how man! moles
of gas do $ have*
142.% or 14! ol
""# $f $ have /').& moles of gas at a temperature of .& 1 and a pressure of "'.&
atmospheres, what is the volume of the gas*
.#% L
")# $f $ have .) liters of gas held at a pressure of ,'% atm and a temperature of ))& 1,
how man! moles of gas do $ have*
1!.! ol
For chemistry help, visit www.chemfiesta.com 2000 Cavalcade Publishing - ll !ights !eserved
The Ideal and &o'(ned Gas Laws PV = nRT or P
1
V
1
= P
2
V
2
T
1
T
2
"se your #nowledge of the ideal and combined gas laws to solve the following
problems. $f it involves moles or grams, it must be P% & n!'
1) If four moles of a gas at a pressure of 5.4 atmospheres have a volume of
120 liters, what is the temperature?
1973 K
2) If I initially have a gas with a pressure of 84 ka an! a temperature of "5
0
# an! I heat it an a!!itional 2"0 !egrees, what will the new pressure $e?
%ssume the volume of the &ontainer is &onstant.
146.7 kPa
") 'y &ar has an internal volume of 2(00 liters. If the sun heats my &ar from
a temperature of 20
0
# to a temperature of 55
0
#, what will the pressure
insi!e my &ar $e? %ssume the pressure was initially )(0 mm *g.
850.8 or 851 mm Hg
4) *ow many moles of gas are in my &ar in pro$lem +"?
108 mol
5) % toy $alloon fille! with air has an internal pressure of 1.25 atm an! a
volume of 2.50 ,. If I take the $alloon to the $ottom of the o&ean where
the pressure is -5 atmospheres, what will the new volume of the $alloon
$e? *ow many moles of gas !oes the $alloon hol!? .%ssume / 0 285 1)
.0329 L
.13 mol
For chemistry help, visit www.chemfiesta.com 2000 Cavalcade Publishing - ll !ights !eserved
)I*+, G-. L-W. W/RK.0++T 1od(2(ed '3 )r. 4asann5
0reated b! Tara 3' 4oore at www'learning'mgccc'cc'ms'us5p5sciencedocs5gaslawwsheet'htm
,(rect(ons : 6nswer each +uestion below' Then wr(te the nae o2 the 6as law 7sed to solve each +uestion
in the left margin ne7t to each +uestion'
"' 6 gas occupies ,'&3 at )'& mm 8g pressure' 9hat is the volume at "/ mm 8g at the same temperature*
.$%" L
)' 6 constant volume of o7!gen is heated from "//0 to "-&0' The initial pressure is %'" atm' 9hat is the final
pressure*
".0! at
,' 6 sample of )&3 of :8, gas at "/0 is heated at constant pressure until it fills a volume of &/3' 9hat is the
new temperature in 0*
2#! &
%' 6 certain +uantit! of argon gas is under "( torr pressure at )&,1 in a ")3 vessel' 8ow man! moles of argon
are present*
.012 ol
&' 6n unnown gas weighs ,%g and occupies ('.3 at ) atm and )%&1' 9hat is its molecular weight*
"1.1 68ol
(' 6n ideal gas occupies %//ml at )./ mm 8g and (&0' $f the pressure is changed to "'% atm and the
temperature is increased to "//0, what is the new volume*
110.4 L or .110 L
.' 9hat is the volume of ),g of neon gas at "0 and a pressure of ) atm*
12.$ L
-' $f "" moles of 80l gas occupies "&3 at ,//0, what is the pressure in torr*
269220 torr
2' The pressure is ('& atm, )', mole of ;r) gas occupies 2', 3 ' 9hat is the temperature in 0*
%. 0
For chemistry help, visit www.chemfiesta.com 2000 Cavalcade Publishing - ll !ights !eserved
"/' 6 (//m3 balloon is filled with helium at .//mm 8g barometric pressure' The balloon is released and climbs
to an altitude where the barometric pressure is %//mm 8g' 9hat will the volume of the balloon be if, during
the ascent, the temperature drops from )% to &0*
#$! L or .#$! L
""' 6n unnown gas has a volume of )//3 at & atm and -"%/0' 9hat is its volume at <TP*
20"2.6 or 20"! L
")' $n an autoclave, a constant amount of steam is generated at a constant volume' Under "'// atm pressure the
steam temperature is "//0' 9hat pressure setting should be used to obtain a "(&0 steam temperature for
the sterili=ation of surgical instruments*
21.22 kPa
",' 6ir contains o7!gen, nitrogen, carbon dio7ide, and trace amounts of other gases' 9hat is the partial pressure
of o7!gen >P?)# at 101.!kPa o2 total :ress7re if it@s nown that the partial pressures of nitrogen, carbon
dio7ide, and other gases are .2'"Pa, /'/%/Pa, and /'2%Pa, respectivel!* 9hat is the name of the gas law
used for this*
21.22 kPa
"%' A7plain wh! the rates of diffusion of nitrogen gas and carbon mono7ide gas are almost identical at the same
temperature*
.(nce the(r olar asses are the sae9 the(r rates o2 d(227s(on
sho7ld 'e the sae accord(n6 the Graha;s law
"&' 9hat distinguishes effusion from diffusion* 8ow are these processes similar*
Biffusion is gas moving spontaneousl! from areas of high concentration to low
concentration
Affusion is diffusion of a gas escaping from a tin! opening
"(' 9hich of the gases effuses faster at the same temperature: molecular chlorine, nitrogen dio7ide, ammonia or
molecular nitrogen gas* 6nd wh!*
<astest .lowest s(nce lower asses e227se 2aster
=0
!
> =
2
> =/
2
> &l
2
".' A7plain what each of the following changes would do to the pressure in a closed container >increase or
decrease pressure#' 6# Part of the gas is removed, ;# The container si=e >volume# is decreased, and
0#Temperature is increased'
-. 6as reo?ed decrease (n :ress7re
@. Vol7e decrease (ncrease (n :ress7re
&. Te:erat7re (ncrease (ncrease (n :ress7re
"-' Betermine the total pressure of a gas mi7ture that contains o7!gen, nitrogen and helium in the following
partial pressures of )'/atm for o7!gen, %'.atm for nitrogen and 2"!.2"kPa for helium'
#.2 at or #!1.#6 kPa
For chemistry help, visit www.chemfiesta.com 2000 Cavalcade Publishing - ll !ights !eserved
Вам также может понравиться
- High-Pressure Fluid Phase Equilibria: Phenomenology and ComputationОт EverandHigh-Pressure Fluid Phase Equilibria: Phenomenology and ComputationОценок пока нет
- G484 Module 3 4.3.4 Ideal GasesДокумент10 страницG484 Module 3 4.3.4 Ideal GasesIgnatius AgustaОценок пока нет
- 05 Gases Without AnswersДокумент17 страниц05 Gases Without Answersapi-287405319Оценок пока нет
- Gas Laws:: P V K VДокумент18 страницGas Laws:: P V K VFarah Zu'biОценок пока нет
- Combined Gas Law WorksheetДокумент1 страницаCombined Gas Law Worksheetapi-483662721Оценок пока нет
- Gas Laws Ws PDFДокумент6 страницGas Laws Ws PDFJulia Franchesca BorromeoОценок пока нет
- Gas & Its LawДокумент47 страницGas & Its LawMerahouseОценок пока нет
- Dalton's Law of Partial Pressure Problems and SolutionsДокумент1 страницаDalton's Law of Partial Pressure Problems and SolutionsIrni BeyОценок пока нет
- Chemical EquilibriumДокумент11 страницChemical EquilibriumYuaОценок пока нет
- Redox Titration of Ferri To Ferro Illustrating The Nernst Eq and Beer's LawДокумент3 страницыRedox Titration of Ferri To Ferro Illustrating The Nernst Eq and Beer's LawAlex NavaОценок пока нет
- Chapter: 1 Stoichiometric Relationships: SubtopicsДокумент108 страницChapter: 1 Stoichiometric Relationships: SubtopicsBОценок пока нет
- C1 - Basic Concepts of Chemistry - Solutions (v18) - HD - CLДокумент20 страницC1 - Basic Concepts of Chemistry - Solutions (v18) - HD - CLAashish DubeyОценок пока нет
- Section 3 EnergeticsДокумент47 страницSection 3 Energeticsapi-3734333Оценок пока нет
- Thermochemistry (Important Notes)Документ2 страницыThermochemistry (Important Notes)HenrySeowОценок пока нет
- Empirical Versus Molecular FormulasДокумент5 страницEmpirical Versus Molecular FormulasJaz SantosОценок пока нет
- Chemistry Teacher Book Chapter 5.1Документ6 страницChemistry Teacher Book Chapter 5.1BryceWallsОценок пока нет
- Worksheet 3 (Partial Pressures)Документ2 страницыWorksheet 3 (Partial Pressures)Jose Ruben SortoОценок пока нет
- Chemistry Unit 4 Part 2 ReallyacademicsДокумент45 страницChemistry Unit 4 Part 2 ReallyacademicsWill AndyОценок пока нет
- Dynamic Equilibrium Reactions Reach Constant ConcentrationsДокумент22 страницыDynamic Equilibrium Reactions Reach Constant ConcentrationsAN NGUYENОценок пока нет
- U3 Oxidation and Reduction PPT WatermarkДокумент45 страницU3 Oxidation and Reduction PPT Watermarkapi-125934329Оценок пока нет
- Practice estimating to find the best answer.: Δt 5 x rate = -Δ (B) 5 x 0.0243 M/s = - Δ (B) -0.12125~ - 0.122 M/sДокумент10 страницPractice estimating to find the best answer.: Δt 5 x rate = -Δ (B) 5 x 0.0243 M/s = - Δ (B) -0.12125~ - 0.122 M/sjeffrey XiaoОценок пока нет
- Johniya Cochran - Ionic and Covalent Bonds ActivityДокумент4 страницыJohniya Cochran - Ionic and Covalent Bonds ActivityJohniya CochranОценок пока нет
- Exothermic and Endothermic ReactionsДокумент5 страницExothermic and Endothermic ReactionsSehyun OhОценок пока нет
- Test Gas LawsДокумент5 страницTest Gas LawsCenando BodanioОценок пока нет
- Chemistry Study SheetДокумент2 страницыChemistry Study SheetSteph PiperОценок пока нет
- Physical Chemistry 1 Prob SetДокумент8 страницPhysical Chemistry 1 Prob SetArrianne Jaye MataОценок пока нет
- UNIT 1 - Assignment 7 - Harder Balancing Problems - Answer KeyДокумент2 страницыUNIT 1 - Assignment 7 - Harder Balancing Problems - Answer KeyAayush ChoudharyОценок пока нет
- Voltaic Cell Design Lab - How Temperature Affects VoltageДокумент2 страницыVoltaic Cell Design Lab - How Temperature Affects VoltageTheVioletFrost83% (6)
- Lab 3-1a, Mass Percent of Carbon in Sodium Bicarbonate (Key)Документ7 страницLab 3-1a, Mass Percent of Carbon in Sodium Bicarbonate (Key)David StainesОценок пока нет
- CH 301 Unit 2 Exam-SolutionsДокумент9 страницCH 301 Unit 2 Exam-SolutionsbrunosipodОценок пока нет
- Photochemical ReactionДокумент16 страницPhotochemical ReactionChandra ReddyОценок пока нет
- Chemistry - Mccord - Exam 1Документ9 страницChemistry - Mccord - Exam 1Miguel MartinezОценок пока нет
- Chem 155 ElectrochemistryДокумент12 страницChem 155 Electrochemistryapi-318921527Оценок пока нет
- AS Chemistry Unit 1 Class Test Jan 2015 QuestionsДокумент26 страницAS Chemistry Unit 1 Class Test Jan 2015 Questionsecs90603Оценок пока нет
- L Oss of E Lectrons Is O Xidation G Ain of E Lectrons Is R EductionДокумент10 страницL Oss of E Lectrons Is O Xidation G Ain of E Lectrons Is R Eductionherman lewisОценок пока нет
- Limiting Reagents LabДокумент7 страницLimiting Reagents Labapi-349567441Оценок пока нет
- 2007 Jun Exam PaperДокумент20 страниц2007 Jun Exam PapertheoggmonsterОценок пока нет
- Organic Chemistry NomenclatureДокумент8 страницOrganic Chemistry NomenclaturetasneemОценок пока нет
- Calorimetry (Formal)Документ17 страницCalorimetry (Formal)Bettinamae Ordiales De Mesa0% (1)
- MCQ Madness 1 PDFДокумент15 страницMCQ Madness 1 PDFnotabc gamerОценок пока нет
- 13 Partial Pressures of GasesДокумент6 страниц13 Partial Pressures of GasesTanisha Marie100% (1)
- Balancing Chemical EquationsДокумент2 страницыBalancing Chemical Equationsirfan_ali_balochОценок пока нет
- Electron Affinity and Ionization EnergiesДокумент251 страницаElectron Affinity and Ionization Energiesgkawsar22Оценок пока нет
- AP Chemistry - Hess's Law LabДокумент3 страницыAP Chemistry - Hess's Law LabJonathan Chen83% (12)
- 2011wksht Heating Curve - CalcsДокумент3 страницы2011wksht Heating Curve - Calcsapi-2982478730% (2)
- CHM 101 Lecture Note-Gas LawsДокумент11 страницCHM 101 Lecture Note-Gas LawsMichael DanielsОценок пока нет
- PHET Gases - Kinetic MOLECULAR THEORY-dikonversiДокумент6 страницPHET Gases - Kinetic MOLECULAR THEORY-dikonversiLidya utari100% (1)
- Chemistry Unit 4 Part 1 ReallyacademicsДокумент41 страницаChemistry Unit 4 Part 1 ReallyacademicsWill AndyОценок пока нет
- Gas Laws Worksheetsand SolutionsДокумент9 страницGas Laws Worksheetsand SolutionskjgfsogkpsОценок пока нет
- Dang Nguyen Exp 1 Measurements-1Документ5 страницDang Nguyen Exp 1 Measurements-1Nguyễn Hoàng ĐăngОценок пока нет
- StoichiometryДокумент4 страницыStoichiometryCourtney JenningsОценок пока нет
- Chemistry Unit 4 Part 3 ReallyacademicsДокумент35 страницChemistry Unit 4 Part 3 ReallyacademicsWill AndyОценок пока нет
- Gas Laws worksheet solutionsДокумент3 страницыGas Laws worksheet solutionslyn janeОценок пока нет
- AP Ch. 12-13 Kinetics & Equilibrium Review AnswersДокумент35 страницAP Ch. 12-13 Kinetics & Equilibrium Review AnswersRucar Rad0% (1)
- CH U6 A4 Stoichiometry WorksheetДокумент3 страницыCH U6 A4 Stoichiometry WorksheetUltrapure Analytichem100% (1)
- 6 Chem PackДокумент5 страниц6 Chem PackCody YangОценок пока нет
- Chemical Equilibrium .PresentationДокумент17 страницChemical Equilibrium .PresentationtalhawasimОценок пока нет
- Enzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisОт EverandEnzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisРейтинг: 4 из 5 звезд4/5 (2)
- NAS1832 Thru NAS1836, Inserts: Genuine Aircraft Hardware CoДокумент3 страницыNAS1832 Thru NAS1836, Inserts: Genuine Aircraft Hardware CoSon NguyenОценок пока нет
- 2.8 Commissioner of Lnternal Revenue vs. Algue, Inc., 158 SCRA 9 (1988)Документ10 страниц2.8 Commissioner of Lnternal Revenue vs. Algue, Inc., 158 SCRA 9 (1988)Joseph WallaceОценок пока нет
- Offer LetterДокумент2 страницыOffer LetterIpe ClosaОценок пока нет
- ResultGCUGAT 2018II - 19 08 2018Документ95 страницResultGCUGAT 2018II - 19 08 2018Aman KhokharОценок пока нет
- Money Purse Telugu BookДокумент3 страницыMoney Purse Telugu BookAsheervadam Suraboina0% (3)
- Fdas Quotation SampleДокумент1 страницаFdas Quotation SampleOliver SabadoОценок пока нет
- Marraige and CompatabilityДокумент3 страницыMarraige and CompatabilityRamesh Menon100% (1)
- Loadstar Shipping Vs MesanoДокумент2 страницыLoadstar Shipping Vs MesanoCuddlyОценок пока нет
- 3000 Watt Power InverterДокумент1 страница3000 Watt Power InvertersimonОценок пока нет
- Home - ECourt India Services Harpreet Singh by JN PatelДокумент2 страницыHome - ECourt India Services Harpreet Singh by JN PatelIasam Groups'sОценок пока нет
- CASE BRIEF Switzerland v. Nigeria San Pedro Pio Case PDFДокумент3 страницыCASE BRIEF Switzerland v. Nigeria San Pedro Pio Case PDFDILG STA MARIAОценок пока нет
- Brief History of PhilippinesДокумент6 страницBrief History of PhilippinesIts Me MGОценок пока нет
- Our Native Hero: The Rizal Retraction and Other CasesДокумент7 страницOur Native Hero: The Rizal Retraction and Other CasesKrichelle Anne Escarilla IIОценок пока нет
- Drug Free Workplace Act PolicyДокумент8 страницDrug Free Workplace Act PolicyglitchygachapandaОценок пока нет
- Sagar Kailas Salunke Primary Account Holder Name: Your A/C StatusДокумент3 страницыSagar Kailas Salunke Primary Account Holder Name: Your A/C Statussagar_salunkeОценок пока нет
- Identity Theft WebquestДокумент2 страницыIdentity Theft Webquestapi-256439506Оценок пока нет
- McCurdy FR 6-15-18Документ12 страницMcCurdy FR 6-15-18David FritzОценок пока нет
- Abraham 01Документ29 страницAbraham 01cornchadwickОценок пока нет
- Romans Catholic DefendersДокумент176 страницRomans Catholic DefendersКирие ЭлейсонОценок пока нет
- Puss in Boots Esl Printable Reading Comprehension Questions Worksheet For KidsДокумент2 страницыPuss in Boots Esl Printable Reading Comprehension Questions Worksheet For KidsauraОценок пока нет
- Annual ReportДокумент110 страницAnnual ReportSarwan AliОценок пока нет
- A Project Report ON Practice SchoolДокумент31 страницаA Project Report ON Practice SchoolSiddharth DevnaniОценок пока нет
- Administrative DisciplineДокумент27 страницAdministrative DisciplineSeven De Los ReyesОценок пока нет
- Denmark Trans Fats LawДокумент2 страницыDenmark Trans Fats LawPerwaiz KhanОценок пока нет
- Assurance Services: Definition, Types and LimitationsДокумент146 страницAssurance Services: Definition, Types and LimitationsMudassar Iqbal0% (1)
- War in The Tibet of Old On A Number of Occasions Meant The Military Intervention of Various Mongolian Tribes Into The Internal Affairs of The CountryДокумент44 страницыWar in The Tibet of Old On A Number of Occasions Meant The Military Intervention of Various Mongolian Tribes Into The Internal Affairs of The CountryTikkun OlamОценок пока нет
- Question Bank of Sem - 1 To Sem-9 of Faculty of Law PDFДокумент203 страницыQuestion Bank of Sem - 1 To Sem-9 of Faculty of Law PDFHasnain Qaiyumi0% (1)
- Hacienda Fatima, Et Al. v. National Federation of Sugarcane Workers-Food and General Trade, G.R. No. 149440, Jan. 28, 2003Документ8 страницHacienda Fatima, Et Al. v. National Federation of Sugarcane Workers-Food and General Trade, G.R. No. 149440, Jan. 28, 2003Martin SОценок пока нет
- Napoleon Edwards Amended Complaint - RedactedДокумент13 страницNapoleon Edwards Amended Complaint - Redactedthe kingfishОценок пока нет
- Chapter 14 Exercises - Investments - BodieДокумент2 страницыChapter 14 Exercises - Investments - BodieNguyệtt HươnggОценок пока нет