Академический Документы
Профессиональный Документы
Культура Документы
Lapp Angl HPLC Uv
Загружено:
8292550 оценок0% нашли этот документ полезным (0 голосов)
37 просмотров10 страницANALYSIS of FOOD and NATURAL PRODUCTS LABORATORY exercise determination of vitamins, caffeine and preservatives (method: liquid chromatography with UV detection)
Исходное описание:
Оригинальное название
Lapp Angl Hplc Uv
Авторское право
© © All Rights Reserved
Доступные форматы
PDF, TXT или читайте онлайн в Scribd
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документANALYSIS of FOOD and NATURAL PRODUCTS LABORATORY exercise determination of vitamins, caffeine and preservatives (method: liquid chromatography with UV detection)
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PDF, TXT или читайте онлайн в Scribd
0 оценок0% нашли этот документ полезным (0 голосов)
37 просмотров10 страницLapp Angl HPLC Uv
Загружено:
829255ANALYSIS of FOOD and NATURAL PRODUCTS LABORATORY exercise determination of vitamins, caffeine and preservatives (method: liquid chromatography with UV detection)
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PDF, TXT или читайте онлайн в Scribd
Вы находитесь на странице: 1из 10
________________________________________________________________________________
ANALYSIS OF FOOD AND NATURAL PRODUCTS
LABORATORY EXERCISE
Determination of vitamins, caffeine and
preservatives
(method: liquid chromatography with UV detection)
___________________________________________________________
Exercise guarantor: Assoc.Prof. Ing.Vra Schulzov, Ph.D.
CONTENT
______________________________________________________________________________________________________________________
Required knowledge ............................................................................................................................ 3
Evaluation criteria ................................................................................................................................ 3
Laboratory exercise content ................................................................................................................. 3
A. Working procedure for the determination of vitamin C in beverages, fruits and vegetables ......... 4
1. USEFULNESS OF THE METHOD ........................................................................................ 4
2. THE PRINCIPLE OF THE METHOD .................................................................................... 4
3. INSTRUMENTS AND EQUIPMENTS .................................................................................. 4
4. STANDARD AND CALIBRATION SOLUTIONS ............................................................... 4
5. CHEMICALS, SOLUTIONS, AND MATERIALS ................................................................ 4
6. WORK PROCEDURE ............................................................................................................. 4
B. Working procedure for the determination of caffeine, vitamin C and preservatives in beverages
(SN EN 12856)................................................................................................................................... 7
1. USEFULNESS OF THE METHOD ........................................................................................ 7
2. THE PRINCIPLE OF THE METHOD .................................................................................... 7
3. INSTRUMENTS AND EQUIPMENTS .................................................................................. 7
4. STANDARD AND CALIBRATION SOLUTIONS ............................................................... 7
5. CHEMICALS, SOLUTIONS, AND MATERIALS ................................................................ 7
6. WORK PROCEDURE ............................................................................................................. 7
UV spectra of analytes ......................................................................................................................... 9
Required knowledge
1. Principle of sample preparation for unstable compounds determination (isolation and
extraction methods)
2. Principle of liquid chromatography (stationary phases, types of columns, mobile phases,
detectors)
3. Principle of spectrophotometry (Lambert-Beers law) structure and properties of vitamin C
(stability, occurrence in foodstuffs)
Evaluation criteria
- proved theoretical knowledge
- quality of practical work execution
- compliance with laboratory work rules (safety, order)
- protocol integrity of experimental data, results calculation and discussion
Laboratory exercise content
A. Determination of vitamin C in beverages, fruits and vegetables by HPLC/UV method
(accredited SZPI method A/11, SN EN 14130)
- Monitoring of vitamin C stability during sample preparation, storage and
thermal processing
B. Determination of caffeine, vitamin C and preservatives in beverages by HPLC/UV
method (SN EN 12856)
A. Working procedure for the determination of vitamin C in
beverages, fruits and vegetables
(according to accredited method SZPI Praha A/11 and SN EN 14130)
1. USEFULNESS OF THE METHOD
The method is used to determine the content of ascorbic acid in beverages, fruit, vegetables,
vegetable products, and in vitamin supplements.
2. THE PRINCIPLE OF THE METHOD
Ascorbic acid is extracted from a homogenized sample by the metaphosphoric acid solution.
The extract is filtered and then the concentration of vitamin C is determined by the method of high
performance liquid chromatography with UV detection. Identification and quantification is done by
the method of external standard calibration curve.
3. INSTRUMENTS AND EQUIPMENTS
Liquid chromatograph Hewlett Packard Series 1100 ( UV detector = 254 nm )
Analytical column RP C
18
or RP C
8
(250 x 4,6 mm, 5 m; 125 4 mm, 5 m)
HP Chemstation, Hewlett Packard, USA
Analytical scales
Ultrasonic bath
Laboratory mixer or homogenizer, glass or plastic grater
Cooker, stainless pot, kettle
Vacuum filtration device
4. STANDARD AND CALIBRATION SOLUTIONS
L-ascorbic acid p.a. (PENTA)
Preparation of standard solutions:
Prepare a stock solution of L-ascorbic acid of the concentration of 1 mg/ml in extraction
solvent (preparation of extraction solvent per point 6.1.)
For completion of calibration curve prepare standard solutions of concentration of 10, 30,
60, 90, and 120 mg/l in extraction solution (preparation of extraction solvent per point 6.1)
Note: Maximal time for usefulness of the standard solution is one day.
5. CHEMICALS, SOLUTIONS, AND MATERIALS
- methanol (for HPLC)
- demineralised and distilled water
- metaphosphoric acid
- unless specified otherwise, use chemical of cleanliness class p.a.
- membrane filter of porosity 5 m
- common laboratory glass
6. WORK PROCEDURE
6.1. Preparation of the laboratory sample
Preparation of extraction agent
3% solution of metaphosphoric acid (dissolve 15 g of solid metaphosphoric acid in 500 ml
of distilled water).
Solid Samples
10 - 25 g of solid sample is homogenized with 60 80 ml of extraction agent for the
duration of one minute. The obtained extract is filtered through filtration paper, which is washed
through few times; it is also possible to use vacuum filtration. The filtrate is quantitatively
transferred into a 100 ml volumetric flask and an extraction agent is added up to the 100 ml mark.
All sample solutions are filtered before injection into a liquid chromatograph using a membrane
filter.
Liquid Samples
Liquid materials after throughout mixing are diluted by the extraction agent (the amount is
chosen according to the expected vitamin C content, in such a way that concentration of analyzed
solution is corresponding to the range of calibration of 10 120 mg/l). Before introduction into a
liquid chromatograph, it is necessary to filtrate it through membrane filter.
Tablet
Declaration 100 mg / tablet.
Dissolve the tablet in 100 ml volumetric flask by extraction agent and dilute 20x (0.5 ml to
10 ml) resulting concentration is about 50 mg/l.
Note: In case of need (the result is outside the calibration range), it is possible to dilute the sample
by extraction agent (metaphosphoric acid)
Monitoring of stability of vitamin C during sample preparation, storage and thermal processing
Solid samples
Samples of fruits and vegetables are heated in water and in steam in stainless vessel
conditions will be specified by assistant
Liquid samples
Tea with vitamin content is poured by hot water or stored under defined conditions
6.2. Analysis by liquid chromatography
Analytical column: Analytical column RP C
18
or RP C
8
(250 x 4,6 mm, 5 m; 125
4,6 mm, 5 m;) with precolumn
Mobile phase:
methanol : water (5:95, v/v) pH= 3 (H
3
PO
4
), flow 1 ml/min
Injection volume 20 l
Detection: UV 254 nm
Temperature: Laboratory temperature
6.3. Identification and quantification
Identification is performed by the comparison of retention time of analyte in analyzed
sample with the retention time of the calibration standard. Quantitative analysis is performed using
the external standard method by the calculating of the concentration in analytical sample from the
calibration curve equation (the dependence of the area or height of the peak on the concentration of
standard in mg/l; concentration in the analytical sample must be in interval between the lowest and
highest point of calibration curve) and by recalculation on the original sample by the following
formula:
m
V x
c
a
a
.
The student compares the calculated content of the ascorbic acid with the declared content
of vitamin C on the label of the sample (if it is mentioned), or with a common concentration of
vitamin C in the given sample.
B. Working procedure for the determination of caffeine, vitamin
C and preservatives in beverages (SN EN 12856)
1. USEFULNESS OF THE METHOD
The method is used to determine the content of caffeine, ascorbic acid and preservatives in
beverages (tea, coffee, energetic drinks etc.)
2. THE PRINCIPLE OF THE METHOD
Target compounds are extracted from a homogenized beverage sample by water. The
extract is filtered and then the concentration of analytes is determined by the method of high
performance liquid chromatography with UV detection. Identification and quantification is done by
the method of external standard calibration curve.
3. INSTRUMENTS AND EQUIPMENTS
Liquid chromatograph Hewlett Packard Series 1100 ( UV detector)
Analytical column RP C
18
or RP C
8
(250 x 4,6 mm, 5 m; 125 4 mm, 5 m)
HP Chemstation, Hewlett Packard, USA
Analytical scales
Ultrasonic bath
Vacuum filtration device
4. STANDARD AND CALIBRATION SOLUTIONS
L-ascorbic acid p.a. (PENTA)
Preparation of standard solutions as in point A.
Sorbic acid, benzoic acid, caffeine (Sigma Aldrich)
Standards will be provided by assistant as solid substance or as solution
For completion of calibration curve prepare standard solutions of concentration of 50, 100,
200, and 400 g/ml (from stock solution c = 1000 g/ml).
5. CHEMICALS, SOLUTIONS, AND MATERIALS
- methanol (for HPLC)
- demineralised and distilled water
- acetate buffer (preparation of 500 ml: 0.8204 g of sodium acetate trihydrate and 0.575 ml of
acetic acid put into a 500 ml volumetric flask and fill it with demineralised water)
- unless specified otherwise, use chemical of cleanliness class p.a.
- membrane filter of porosity 5 m
- common laboratory glass
6. WORK PROCEDURE
6.1. Preparation of the laboratory sample
Liquid Samples
Liquid materials, after throughout mixing and degassing in an ultrasonic bath, are diluted by
water agent. Before introduction into liquid chromatograph, it is necessary to filtrate it through
membrane filter.
6.2. Analysis by liquid chromatography
Analytical column: Analytical column RP C
18
or RP C
8
(250 x 4,6 mm, 5 m; 125
4,6 mm, 5 m;) with precolumn
Mobile phase:
Injection volume:
MeOH/acetate buffer (3/7, v/v), flow 1 ml/min
(prepared mobile phases is filtered and degassed in ultrasonic bath
for duration of 10 minutes)
10 or 20 l
Detection: UV 273 nm for caffeine, 254 nm for vitamin C, 228 nm for benzoic
acid, 254 nm for sorbic acid
Temperature: Laboratory temperature
6.3. Identification and quantification
The identification of the analyte is performed by the comparison of its retention time in
analyzed sample with the retention time of the calibration standard. Quantitative analysis is
performed using the external standard method by the calculating of the concentration in analytical
sample from the calibration curve equation (the dependence of the area or height of the peak on the
concentration of standard in mg/l; concentration in the analytical sample must be in interval
between the lowest and highest point of calibration curve) and by recalculation on the original
sample by the following formula:
m
V x
c
a
a
.
The calculated content of the ascorbic acid, caffeine and preservatives in beverages express
in mg/l, content in tea and coffee in mg/kg.
UV spectra of analytes
Ascorbic acid 12 mg/l
Caffeine 250 mg/l
Sorbic acid 50 mg/ml
n m 200 225 250 275 300 325 350 375
m AU
0
250
500
750
1000
1250
1500
1750
*DAD1, 11.036 (1841 mAU, - ) Ref=2.809 & 14.996 of 110907000004.D
n m 200 220 240 260 280 300
m AU
0
5
10
15
20
25
*DAD1, 1.834 (28.6 mAU, - ) Ref=1.594 & 2.041 of 101005000000.D
Benzoic acid 100 mg/ml
nm 200 220 240 260 280 300 320 340 360 380
mAU
0
50
100
150
200
250
300
350
DAD1, 11.507 (389 mAU, - ) of 033-0301.D
n m 220 240 260 280 300
m AU
0
20
40
60
80
100
120
140
*DAD1, 6.078 (151 mAU, - ) Ref=5.538 & 6.645 of 101008000001.D
Вам также может понравиться
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryОт EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryРейтинг: 3.5 из 5 звезд3.5/5 (231)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)От EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Рейтинг: 4.5 из 5 звезд4.5/5 (121)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaОт EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaРейтинг: 4.5 из 5 звезд4.5/5 (266)
- The Little Book of Hygge: Danish Secrets to Happy LivingОт EverandThe Little Book of Hygge: Danish Secrets to Happy LivingРейтинг: 3.5 из 5 звезд3.5/5 (400)
- Never Split the Difference: Negotiating As If Your Life Depended On ItОт EverandNever Split the Difference: Negotiating As If Your Life Depended On ItРейтинг: 4.5 из 5 звезд4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerОт EverandThe Emperor of All Maladies: A Biography of CancerРейтинг: 4.5 из 5 звезд4.5/5 (271)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeОт EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeРейтинг: 4 из 5 звезд4/5 (5794)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyОт EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyРейтинг: 3.5 из 5 звезд3.5/5 (2259)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersОт EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersРейтинг: 4.5 из 5 звезд4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnОт EverandTeam of Rivals: The Political Genius of Abraham LincolnРейтинг: 4.5 из 5 звезд4.5/5 (234)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreОт EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreРейтинг: 4 из 5 звезд4/5 (1090)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceОт EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceРейтинг: 4 из 5 звезд4/5 (895)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureОт EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureРейтинг: 4.5 из 5 звезд4.5/5 (474)
- The Unwinding: An Inner History of the New AmericaОт EverandThe Unwinding: An Inner History of the New AmericaРейтинг: 4 из 5 звезд4/5 (45)
- The Yellow House: A Memoir (2019 National Book Award Winner)От EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Рейтинг: 4 из 5 звезд4/5 (98)
- Plastics Piping Systems For Non-Pressure Underground Drainage and Sewerage Polyethylene (PE)Документ38 страницPlastics Piping Systems For Non-Pressure Underground Drainage and Sewerage Polyethylene (PE)MohamedHanyОценок пока нет
- Pumps API-610 PDFДокумент5 страницPumps API-610 PDFGeorge_Wabag_2014Оценок пока нет
- Rig Power LLCДокумент12 страницRig Power LLCRichard EVОценок пока нет
- H074Документ312 страницH07482925567% (3)
- Pre Commisioning Start UpДокумент33 страницыPre Commisioning Start Upivanrishi100% (3)
- Certificate of AnalysisДокумент1 страницаCertificate of Analysis829255Оценок пока нет
- Safety Rules BrochureДокумент12 страницSafety Rules BrochureSuraj DeodathОценок пока нет
- W20PGДокумент147 страницW20PGAnup MishraОценок пока нет
- Most Up-To-Date Table Is Available From: Internet E-Mail Last Update 20-Feb-03Документ7 страницMost Up-To-Date Table Is Available From: Internet E-Mail Last Update 20-Feb-03829255Оценок пока нет
- Aw 6-968 Epa 300 Ab PDFДокумент15 страницAw 6-968 Epa 300 Ab PDF829255Оценок пока нет
- Col CareДокумент5 страницCol Care829255Оценок пока нет
- CRLFV Multiresidue MethodsДокумент20 страницCRLFV Multiresidue Methods829255Оценок пока нет
- Aoac970 33Документ1 страницаAoac970 33829255Оценок пока нет
- Ab-098 3 enДокумент8 страницAb-098 3 enVõ Kim UyênОценок пока нет
- S-61 IC Application Note No.: Title: Anions in Ink Using Dialysis For Sample PreparationДокумент1 страницаS-61 IC Application Note No.: Title: Anions in Ink Using Dialysis For Sample Preparation829255Оценок пока нет
- Method TrifluralinДокумент1 страницаMethod Trifluralin829255Оценок пока нет
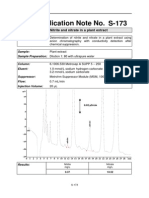
- S-173 IC Application Note No.: Title: Nitrite and Nitrate in A Plant ExtractДокумент1 страницаS-173 IC Application Note No.: Title: Nitrite and Nitrate in A Plant Extract829255Оценок пока нет
- Sấy Cà Phê Nhân 2000kg Khôh - Thiết Bị Sấy Thùng Quay (Kèm Bản Vẽ)Документ26 страницSấy Cà Phê Nhân 2000kg Khôh - Thiết Bị Sấy Thùng Quay (Kèm Bản Vẽ)NamNamОценок пока нет
- CLI VmotionДокумент90 страницCLI VmotionLuan DtОценок пока нет
- Orientation On The Manual For The Evaluation (Autosaved)Документ31 страницаOrientation On The Manual For The Evaluation (Autosaved)Em Boquiren CarreonОценок пока нет
- PC Case ModДокумент42 страницыPC Case ModMihai IavorschiОценок пока нет
- 2018 Sundray Junior Certification Lesson - One - 05 - Fat AP - v3.6.7Документ26 страниц2018 Sundray Junior Certification Lesson - One - 05 - Fat AP - v3.6.7wendy yohanesОценок пока нет
- Koc StandardДокумент40 страницKoc Standardanon_867776278Оценок пока нет
- TurningДокумент9 страницTurningAhmed RexОценок пока нет
- Tricycles Van RaamДокумент29 страницTricycles Van Raampaul20000Оценок пока нет
- Dbi Sala Sealed Blok SRL 34009 34008 DatasheetДокумент17 страницDbi Sala Sealed Blok SRL 34009 34008 DatasheettristenkellyarbОценок пока нет
- L2-ELN-SPE-012.pdf SCADA CablingДокумент7 страницL2-ELN-SPE-012.pdf SCADA CablinghardikОценок пока нет
- SkuДокумент20 страницSkuJLee CarpioОценок пока нет
- Verilog CodesДокумент26 страницVerilog CodesmdlogicsolutionsОценок пока нет
- P4M800 Pro m7Документ31 страницаP4M800 Pro m7rarahimОценок пока нет
- 648 Commander Wrapper UserGuideДокумент146 страниц648 Commander Wrapper UserGuideMelissa TomeОценок пока нет
- SDF Core2Документ251 страницаSDF Core2csontakkeyОценок пока нет
- AVC6000 Product Sheet - 0519Документ6 страницAVC6000 Product Sheet - 0519Gerardo AvendañoОценок пока нет
- Elm 327 DSHДокумент76 страницElm 327 DSHekysperОценок пока нет
- Module 6 STATISTICAL CONCEPTS OF QUALITY MANAGEMENTДокумент5 страницModule 6 STATISTICAL CONCEPTS OF QUALITY MANAGEMENTjennie kimОценок пока нет
- Cics and XML: Geoff SharmanДокумент28 страницCics and XML: Geoff SharmangatrivedОценок пока нет
- Calculation - Method - ULF Unbalanced Load Flow ETAP PDFДокумент8 страницCalculation - Method - ULF Unbalanced Load Flow ETAP PDFZulqibalОценок пока нет
- Yuken Valves Valvulas Direccionales PDFДокумент184 страницыYuken Valves Valvulas Direccionales PDFSimon NgigiОценок пока нет
- UsabqtrДокумент4 страницыUsabqtrVian ZiroОценок пока нет
- HASR09A HASC09A HASR12A HASC12A HASR24A HASC24A HASR30C HASC30C Service ManualДокумент71 страницаHASR09A HASC09A HASR12A HASC12A HASR24A HASC24A HASR30C HASC30C Service ManualUEE100% (1)
- JasperReports Server REST API Reference PDFДокумент174 страницыJasperReports Server REST API Reference PDFJosé Antonio Gómez ViracaОценок пока нет
- Product 11 LayherДокумент40 страницProduct 11 LayherMariela Fernández Morey100% (1)