Академический Документы
Профессиональный Документы
Культура Документы
Chromium (VI) Adsorption and Its Kinetic Studies Using Industrial Spent Biomass
Загружено:
Innovative Research Publications0 оценок0% нашли этот документ полезным (0 голосов)
17 просмотров5 страницChromium (VI) Adsorption and its Kinetic Studies Using Industrial Spent Biomass
Оригинальное название
Chromium (VI) Adsorption and its Kinetic Studies Using Industrial Spent Biomass
Авторское право
© © All Rights Reserved
Доступные форматы
PDF, TXT или читайте онлайн в Scribd
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документChromium (VI) Adsorption and its Kinetic Studies Using Industrial Spent Biomass
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PDF, TXT или читайте онлайн в Scribd
0 оценок0% нашли этот документ полезным (0 голосов)
17 просмотров5 страницChromium (VI) Adsorption and Its Kinetic Studies Using Industrial Spent Biomass
Загружено:
Innovative Research PublicationsChromium (VI) Adsorption and its Kinetic Studies Using Industrial Spent Biomass
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PDF, TXT или читайте онлайн в Scribd
Вы находитесь на странице: 1из 5
International Journal of Scientific Engineering and Technology (ISSN : 2277-1581)
Volume No.3 Issue No.9, pp : 1198-1202 1 Sep 2014
IJSET@2014 Page 1198
Chromium (VI) Adsorption and its Kinetic Studies Using Industrial Spent
Biomass
A Niveditha
1,2
, Pradima J
1
, Dr. Archna
2
1
Department of Biotechnology, Sir M Visvesvaraya Institute of Technology, Bangalore -562157, India,
2
Department of Chemical Engineering, M S Ramaiah Institute of Technology, Bangalore-560054, India,
niv231974@gmail.com
Abstract Biosorption is mass transfer phenomenon by
which a substance can be transferred from liquid phase to
the surface of a solid. The substance becomes bound, by
physical and/or chemical interactions. Availability of large
surface area, surface reactivity, high sorption capacity and
low cost makes it feasible over other traditional methods of
treatment. I n this study important sorption parameters were
investigated as a function of time, pH, initial chromium (VI )
concentration, temperature and biomass dosage. Kinetics
and equilibrium data were obtained from batch experiments.
The biosorption kinetics were checked for both first order
and second order models and it was found that the sorption
process followed the pseudo - second order kinetics.
Adsorption equilibria was checked for Langmuir,
Freundlich, Redlich- Peterson and Tempkin models and the
process was well described by Freundlich isotherm
(R
2
=0.9992), RedlichPetersen isotherm (R
2
=0.961) and
Tempkin I sotherm (R
2
=0.9592) models as compared to
Langmuir I sotherm (R
2
=0.5702) model. The maximum
adsorption capacity of the biomass was 14.2 mg/g at a
temperature of 298K and pH of 2.0. The scanning electron
micrograph revealed the surface texture and morphology of
multilayer adsorption. Surface and pore size analysis
through BET revealed that industrial spent has a specific
surface area of 4.8477 m
2
/g and pore diameter 56.865
o
A.
The present work reveals that spent biomass can be
effectively used for Chromium (VI ) removal at low
concentrations.
Keywords: Chromium (VI), biosorption, kinetics,
isotherm, industrial spent
1. Introduction
Removing and recovering heavy metals from industrial
effluents using biological materials has emerged as a potential
alternative to conventional treatment methods. Economy of
environmental remediation necessitates the biological material
to be naturally available, involve less processing, is abundant
in nature or is a by- product or waste from other industry.
Microorganisms, algae, fungi, yeast, agricultural wastes
among other biomaterials have been tested for metal sorption
and have performed well. These biosorbents contain variety of
functional sites like carboxyl, imidazole, sulphydryl, amino,
phosphate, sulfate, thioether, phenol, carbonyl, amide and
hydroxyl groups [i] which have experimentally proved to
adsorb heavy metals in effluents, even at very low
concentrations. This applicability of biosorbents has been
utilized in removal of Chromium (VI) metal ions in this study.
The biosorbent used is herbal spent, devoid of its active
components (extracted for medicinal purposes) and discarded
as waste. Chromium finds wide application in the making of
steel and other alloys, manufacture of dyes, pigments, leather,
textile, wood - preservation and in treatment of cooling tower
water. Hexavalent chromium is one of the most toxic heavy
metals and may cause a series of health effects, ranging from
shortness of breath to lung cancer [ii]. The removal and
recovery of these heavy metals from industrial effluents is very
important for the environmental protection and by large, human
health. Different alternatives are available in literature,
including chemical precipitation, carbon adsorption, ion
exchange and membrane separation process among others.
However there are limitations in these processes, such as high
cost, low efficiency, labor intensive operation, and lack of
selectivity of the precipitation process [iii]. In the present work,
the influence of several parameters, such as time, pH, initial
chromium (VI) ion concentration, temperature and biomass
dosage were investigated. The kinetics and adsorption
equilibria were obtained from batch experiments. The
biosorbent was analyzed for its porosity and surface area
characteristics through BET analysis and also scanned through
the scanning electron microscope.
II. Material and Methodology
Preparation of biomass
Spent biomass of plant origin, whose active components were
extracted under super critical conditions, was discarded as an
industrial waste in the form of dry powder. This was used as
biosorbent and was obtained from Ms. Sami Labs Pvt Ltd,
Bangalore. The spent biomass was sieved through a 100 m
mesh, dried at a temperature of 60C and used directly without
any pre-treatment.
Chemicals
All chemicals used in this study were of analytical grade and
the solutions were prepared using distilled water. A stock
solution of Cr (VI) (100ppm) was prepared by dissolving
potassium di chromate. Cr (VI) solutions of different
concentrations were obtained by diluting the stock solution. To
adjust pH, 0.1N hydrochloric acid and 0.1N sodium hydroxide
solutions were used.
International Journal of Scientific Engineering and Technology (ISSN : 2277-1581)
Volume No.3 Issue No.9, pp : 1198-1202 1 Sep 2014
IJSET@2014 Page 1199
Kinetic experiments
Kinetic experiments were performed using 100ml Cr (VI)
solutions at different concentrations (20-100ppm) and 0.5g of
biosorbent at a pH of 2.0. Samples were collected at different
intervals of time and analyzed for residual Cr (VI)
concentration by atomic absorption spectrometer (GBC932
plus).
Effect of pH on Cr (VI) adsorption
Experiments were conducted under different pH to investigate
the effect on adsorption. The pH was adjusted to a designated
value from 2.0 to 11.0 using 0.1N hydrochloric acid or 0.1N
sodium hydroxide solution for the study.
Effect of temperature on Cr (VI) adsorption
Experiments were conducted at different temperatures using
an incubator (10-40
o
C) to investigate the effect on biosorption
of Cr (VI) ions.
Effect of biomass variation on Cr (VI) adsorption
Varying biomass dosage, experiments were conducted to
investigate its effect on the removal of chromium metal ions.
Biomass dosage was varied from 0.1 to 2.5 grams.
Adsorption equilibrium
Equilibrium studies were conducted by varying the
concentration of Cr (VI) solutions from 20ppm to 100ppm
with the biosorbent dosage being varied from 0.1 to 2.5g. The
pH was adjusted to 2.0 using 0.1N hydrochloric acid solution.
The mixture was constantly shaken on a shaker at 150rpm for
different time intervals at room temperature. Then the
biosorbent was centrifuged at 10,000rpm in a cooling
centrifuge (REMI C-24) for a period of 10 minutes. The
supernatant was collected and filtered through Whatman 41
filter paper. The amount of Cr (VI) uptake by biosorbent was
estimated by checking for residual Cr (VI) ion concentration
using AAS, correspondingly through the mass balance
equation
(1)
Where q is the adsorption amount at equilibrium (mg/g), C
o
the initial concentration of heavy metal (mg/l), C
e
the
concentration remaining in solution at equilibrium (mg/l) and
W is the biosorbent dosage (g/l)[ix].
Characterization of the adsorbent
The biosorbent was characterized using BET analysis. It was
found that the biosorbent had a specific surface area of 4.8477
m
2
/g and pore diameter 56.865
o
A. The surface feature and
morphology of the biosorbent was analyzed using Scanning
Electron Microscope.
III. Results and Tables
Kinetics of biosorption
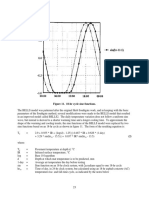
Figure 1 depicts Cr (VI) percentage removal with time (t)
profile by extract spent, at constant initial metal ion
concentration. The removal rate was high in the first 60 min
and then the rate significantly decreased and eventually reached
a stationary phase after 90 min. The result was the same for all
the initial Cr (VI) concentrations. The uptake capacities
increased with increasing initial Cr (VI) concentrations as a
result of higher probability of collision between the ions and
the biosorbent particles. Many models have been developed to
describe the kinetics of heavy metal biosorption [iv-ix]. The
pseudo-second order kinetic model based on sorption capacity
of the solid phase can be used, assuming that measured
concentrations are equal to the cell surface concentrations. The
pseudo second order kinetic rate equation is expressed as
(2)
At boundary conditions q
t
=0 at t=0 and q
t
at time t, the
linearized form of pseudo-second order model is obtained.
(3)
Where k
2
is the second order biosorption rate constants (g/mg
min
-1
), q
e
and q
t
are the amounts of the sorbed metal ions on the
biosorbent at equilibrium, at any time t. The kinetic data
analyzed were in terms of pseudo-second order. Figure 2 shows
the variation of t/q
t
with respect to time. The values of
parameters k
2
, q
e
and correlation coefficients are presented in
Table -1. The correlation coefficients obtained were greater
than 0.9753 and suggests that the sorption of Cr (VI) onto
extract spent followed the second order model very well and
based on the assumption; the rate limiting step may be
chemisorption involving forces through the sharing of electrons
between sorbent and sorbate [x].
Figure 1: Adsorption of Cr (VI) on the extract spent biosorbent
at different time(min) at pH 2 and 298K
Effect of pH and biomass on Cr (VI) adsorption
The effect of pH was significant on biosorption of heavy metals
as it affects the protonation of the functional groups on the
biomass as well as their interaction chemistry [x]. The effect of
International Journal of Scientific Engineering and Technology (ISSN : 2277-1581)
Volume No.3 Issue No.9, pp : 1198-1202 1 Sep 2014
IJSET@2014 Page 1200
pH on the Cr (VI) adsorption was studied and the results are
shown in Figure 3. The percentage adsorption at pH 2 was
39.61% and with the increase in pH, the percentage removal
with biosorbent decreased as the pH moved towards the higher
range. Percentage adsorption also varied with varying biomass
dosage. There was an increase in adsorption of heavy metal
ions linearly, up to 2g and with further increase; it was
observed that the adsorption rate was constant as shown in
Figure 4.
Figure 2: Variation of t/t
q
with time at different initial Cr (VI)
concentrations at a pH of 2 and 298K
Effect of Temperature
The effect of temperature on the equilibrium sorption capacity
were conducted within a temperature range of 10-40
o
C. The
pH of each metal solution was fixed at the optimum value
determined from the pH study i.e., at pH 2.0. Figure 5 show
the variation of percentage removal with temperature. It was
observed that the maximum percentage removal occurred at
room temperature. Earlier experiments conducted, explain our
observations that with further increase in temperature (above
30
o
C) there is marked decrease in the percentage removal [xi].
Figure 3: Adsorption of Cr (VI) on the spent biosorbent at
different pH, 50ppm and 298K
Figure 4: Adsorption of Cr (VI) on the spent biomass at
different biomass dosage
This decrease in biosorption efficiency may be due to an
increase in the relative desorption of the heavy metals from the
solid phase to the liquid phase, destruction of active sites on the
biosorbent surface due to bond disruption, or due to the
weakness of the sorbent active site binding forces and the
sorbate species and also between the adjacent molecules of the
sorbed phase. from Figure 5, it is evident that the variation of
temperature from 20-30
o
C has minimal effect on biosorption
process and experiments can be carried out at room temperature
without adjustments.
Figure 5: Adsorption of Cr (VI) on the spent biomass at
different temperature and at pH 2.0.
Table 1: Pseudo second order adsorption constants
C
o
(ppm) q
e
(mg/g) K
2
(x10
3
g/mg min
-1
) R
2
40 1.534 0.098 0.9774
60 2.892 0.057 0.9753
80 5.492 0.120 0.9975
100 6.336 0.095 0.9978
International Journal of Scientific Engineering and Technology (ISSN : 2277-1581)
Volume No.3 Issue No.9, pp : 1198-1202 1 Sep 2014
IJSET@2014 Page 1201
Adsorption Isotherms
Equilibrium sorption isotherm s describe the capacity of
an adsorbent, characterized by certain constants whose values
express the surface properties and affinity of the adsorbent.
The Langmuir sorption isotherm has been the most widely
used adsorption isotherm and has been successfully applied to
many pollutant adsorption processes for the adsorption of a
solute from a liquid solution. Langmuir theorys basic
assumption is that the adsorption takes place at a specific
homogeneous site within the adsorbent. The saturated
monolayer adsorption isotherm can be represented as:
(4)
Where q
m
is the maximum adsorption capacity and b is an
affinity constant related to the energy of adsorption. The value
of b indicates the strength or affinity of the sorbate for the
solute.
Freundlich isotherm is an empirical isotherm that is used for
non-ideal adsorption and is represented by the equation:
(5)
Where K
f
and n are Freundlich constants and b is the affinity
constant related to the adsorption capacity and the adsorption
intensity of the adsorbent respectively.
Redlich- Peterson Isotherm incorporates features of both the
Langmuir and Freundlich equations, given by the equation:
(6)
A linear analysis is not possible for a three parameter isotherm
and the three isotherm constants can be evaluated from the
pseudo linear plot using a trial and error optimization
method.
Tempkin Isotherm assumes that the fall in the heat of sorption
is a linear rather than the logarithmic, as implied in the
Freundlich equation. The equation is given by
eq T
T
eq
C A
b
RT
q ln (7)
Biosorption isotherms are characterized by certain constants,
the values of which express the surface properties and affinity
of the sorbent and can also be used to compare the adsorption
capacity of biomass for different metal ions. Out of several
isotherm equations available, four have been applied for this
study, the Langmuir, Freundlich, Redlich-Peterson and
Temkin isotherms. The equilibrium constants and correlation
co-efficient (R
2
) are represented in the Table -2. The
maximum adsorption capacity increased with increase in
metal ion concentration. The value of q
m
at room temperature
were 14.92 mg/g at 298 K at pH of 2.0.
Table 2: The Langmuir, Freundlich, Redlich- Peterson and
Tempkin sorption constants for biosorption of Cr (VI) at 298K
and at pH of 2
Langmuir constants Freundlich constants
q
m
(mg/g) b(l/mg) R
2
K
F
n R
2
14.92 0.067 0.5702 0.1887 1.1376 0.9992
Redlich-Petersen
constants
Tempkin constants
A G R
2
A
T
b
t
*10
3
R
2
0.001 0.14 0.961 3.4403 1.58 0.9592
Morphological changes and surface structure of the adsorbent:
The surface texture of the biosorbent after adsorption was
imaged using scanning electron microscope (SEM, JEOL, JSM-
6490LV) with magnification of 2000x. It was clearly observed
(Figure: 6) that the surface tearing takes place after metal
biosorption, confirming the site participation in biosorption.
Analysis through BET (NOVA 1000) indicates the biosorbent
to be mesoporous with a specific surface area of 4.8477 m
2
/g
and pore diameter 56.865
o
A.
(a)
(b)
Figure 6: Scanning electron microscopy (SEM) micrographs of
spent biomass before (a) and after (b) biosorption of Cr (VI).
International Journal of Scientific Engineering and Technology (ISSN : 2277-1581)
Volume No.3 Issue No.9, pp : 1198-1202 1 Sep 2014
IJSET@2014 Page 1202
IV. Conclusion
This adsorption study indicates that the spent industrial
biomass which is usually discarded as waste, in large
quantities, can be used as an efficient adsorbent material for
the removal of Cr(VI) ions from industrial effluents of various
origin. The rate of adsorption was very fast in the first 60
minutes and then showed a steady phase. The biosorption
process was endothermic andfollowed pseudo-second order
kinetic model and the adsorption isotherms were well
described by Freundlich, Redlich-Peterson and Tempkin
isotherm models. The maximum adsorption was 14.12 mg/g
at 298K and pH 2.0. This study led to the conclusion that the
industrial spent biomass with their rapid adsorption rate,
makes them well suited for the removal of chromium (VI)
from wastewater.
Acknowledgement
The authors wish to sincerely thank Sami Labs Limited,
Biotechnology division manufacturing facility, Nelamangala,
Bangalore with head office at 19/1, 19/2, 1
st
main, 2
nd
Phase
Peenya Industrial area, Bangalore, Karnataka 560058 for
providing industrial extract spent as biosorbent to carry out
our work. We also sincerely thank the department of Chemical
Engineering, M S Ramaiah institute of Technology and
Department of Biotechnology and Sir M Visvesvaraya
Institute of Technology for providing the laboratory facilities
to carry out this research work.
References
i. Jianlong Wang, Can Chen (2009), Biosorbents for heavy
metalsremoval and their future, Biotechnology Advances
27(2009)195-226.
ii. T. R. Muraleedharan, Leela Iyengar and C. Venkobachar,
Biosorption, An alternative for metal removal and recovery,
General article.
iii. Ahalya N., Kanamadi R D and Ramachandra T V., 2008.
Biosorption of Chromium (VI) Tamarindus indica pod shells,
Journal of Environmental Science Research International, 1 (2):77-
81
iv. Eric Guibal, Javier Guzman, Ricardo Navarro, Montserrat
Ruiz, Ana Sastre,(2004), Influence of the speciation of metal ions
on the sorption on Chitosan, Fundamentals and applications of
anion separations. pp 225-247.
v. Volesky B. Holan ZR (1995), Biosorption of Heavy
Metals. Biotech, Prog 11:235-259.
vi. J. Yang, B. Volesky, Biosorption of Uranium on
Sargassum biomass, (1999) water Res.33
vii. M. A. Hashim, K. H. Chu, Biosorption of
cadmium by brown, green and red sea weeds, Chem. Engg. J. 97
(2004) 249-255.
viii. V. K. Gupta, A. K. Shrivastava, N. Jain,
"Biosorption of chromium (VI) from aqueous solutions by green
Spirogyra species, Water Res. 35 (2001) 4079-4085.
ix. V. K. Gupta, A. Rastogi, V. K. Saini, N. Jain, Biosorption
of Cu (II) form aqueous solutions by green Spirogyra species, J.
Colloid Interf. Sci.296 (1) (2006) 59-63.
x. Liping Deng, Yingying Su, Hua Su, Xinthing Wang,
Xiaobin Zhu, Sorption and desorption of lead (II) from waste water
by green algae Cladophora fassicularis, Journal of Hazardous
Materials 143 (2007) 220-225.
xi. Z. R. Holan, B. Volesky, I. Prasetyo, Biosorption of
cadmium by biomass of marine algae, Biotech, Bioeng.
41(1993)819-825
xii. Sulaymon AH, Mohammed AA, Al-Musawi TJ
(2013) Column Biosorption of Lead, Cadmium, Copper, and
Arsenic ions onto Algae. J Bioprocess Biotech 3: 128.
Вам также может понравиться
- Power Transformer Protection Using Fuzzy Logic Based ControllerДокумент5 страницPower Transformer Protection Using Fuzzy Logic Based ControllerInnovative Research PublicationsОценок пока нет
- Case Study: Implementation of 5S Methodology in JCB Assembly Business Unit Material StorageДокумент6 страницCase Study: Implementation of 5S Methodology in JCB Assembly Business Unit Material StorageInnovative Research PublicationsОценок пока нет
- The Experimental Study of Eccentric Loadingfor Piled Raft Foundations Settling On Slope CrestДокумент5 страницThe Experimental Study of Eccentric Loadingfor Piled Raft Foundations Settling On Slope CrestInnovative Research PublicationsОценок пока нет
- Surveillance NetworksДокумент2 страницыSurveillance NetworksInnovative Research PublicationsОценок пока нет
- Computational EconomicsДокумент2 страницыComputational EconomicsInnovative Research PublicationsОценок пока нет
- 3D Modeling of Complex Structure Based On AutoCAD VBAДокумент3 страницы3D Modeling of Complex Structure Based On AutoCAD VBAInnovative Research PublicationsОценок пока нет
- Information EngineeringДокумент2 страницыInformation EngineeringInnovative Research PublicationsОценок пока нет
- Computational ElectromagneticsДокумент3 страницыComputational ElectromagneticsInnovative Research PublicationsОценок пока нет
- Wearable ComputingДокумент3 страницыWearable ComputingInnovative Research PublicationsОценок пока нет
- Role of Coaching Institutes in IndiaДокумент5 страницRole of Coaching Institutes in IndiaInnovative Research PublicationsОценок пока нет
- Comparative Print Quality Analysis of Screen Printing and Liquid Ink Based Digital Printing MachinesДокумент3 страницыComparative Print Quality Analysis of Screen Printing and Liquid Ink Based Digital Printing MachinesInnovative Research PublicationsОценок пока нет
- Online Laboratory: Roy G. Perry College of Engineering Prairie View A&M University Prairie View, TX 77446Документ2 страницыOnline Laboratory: Roy G. Perry College of Engineering Prairie View A&M University Prairie View, TX 77446Innovative Research PublicationsОценок пока нет
- Experimental Study About Soil Improvement With Glass FibersДокумент5 страницExperimental Study About Soil Improvement With Glass FibersInnovative Research PublicationsОценок пока нет
- A Smart Monitoring of Faults in Power Transformers and Maintenance Based On Wi-FiДокумент6 страницA Smart Monitoring of Faults in Power Transformers and Maintenance Based On Wi-FiInnovative Research PublicationsОценок пока нет
- Information Security in Cloud Computing: A Systematic Literature Review and AnalysisДокумент6 страницInformation Security in Cloud Computing: A Systematic Literature Review and AnalysisInnovative Research PublicationsОценок пока нет
- CFD Analysis of A 210 MW Tangential Fired BoilerДокумент6 страницCFD Analysis of A 210 MW Tangential Fired BoilerInnovative Research PublicationsОценок пока нет
- The Yellow House: A Memoir (2019 National Book Award Winner)От EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Рейтинг: 4 из 5 звезд4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeОт EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeРейтинг: 4 из 5 звезд4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingОт EverandThe Little Book of Hygge: Danish Secrets to Happy LivingРейтинг: 3.5 из 5 звезд3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureОт EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureРейтинг: 4.5 из 5 звезд4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryОт EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryРейтинг: 3.5 из 5 звезд3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceОт EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceРейтинг: 4 из 5 звезд4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnОт EverandTeam of Rivals: The Political Genius of Abraham LincolnРейтинг: 4.5 из 5 звезд4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItОт EverandNever Split the Difference: Negotiating As If Your Life Depended On ItРейтинг: 4.5 из 5 звезд4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerОт EverandThe Emperor of All Maladies: A Biography of CancerРейтинг: 4.5 из 5 звезд4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaОт EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaРейтинг: 4.5 из 5 звезд4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaОт EverandThe Unwinding: An Inner History of the New AmericaРейтинг: 4 из 5 звезд4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersОт EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersРейтинг: 4.5 из 5 звезд4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyОт EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyРейтинг: 3.5 из 5 звезд3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreОт EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreРейтинг: 4 из 5 звезд4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)От EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Рейтинг: 4.5 из 5 звезд4.5/5 (121)
- EPFL Lectures On Conformal Field Theory in D 3 DimensionsДокумент68 страницEPFL Lectures On Conformal Field Theory in D 3 DimensionsMojeime Igor NowakОценок пока нет
- 2005 - Active Listening Room Compensation For Spatial Sound Reproduction SystemsДокумент293 страницы2005 - Active Listening Room Compensation For Spatial Sound Reproduction Systemsarthi_ramaniamОценок пока нет
- Void Content of Reinforced Plastics: Standard Test Methods ForДокумент3 страницыVoid Content of Reinforced Plastics: Standard Test Methods ForStéfany Vier SteffenОценок пока нет
- Law of OnersiaДокумент2 страницыLaw of Onersiaวงกลม มิ.ย.Оценок пока нет
- DIY Temperature Controlled Solder StationДокумент6 страницDIY Temperature Controlled Solder StationAnonymous UNG1t7lxОценок пока нет
- 2000 (LTPP) FHWA-RD-98-085 Temperature Predictions and Adjustment Factors For Asphalt Pavemen PDFДокумент49 страниц2000 (LTPP) FHWA-RD-98-085 Temperature Predictions and Adjustment Factors For Asphalt Pavemen PDFTim LinОценок пока нет
- Fatigue Crack Propagation Analysis of Repaired Pipes With Composite Patch Under Cyclic PressureДокумент9 страницFatigue Crack Propagation Analysis of Repaired Pipes With Composite Patch Under Cyclic PressureAdam StevensonОценок пока нет
- NAKED Singularities - PenroseДокумент10 страницNAKED Singularities - PenroseLeon FosiltОценок пока нет
- Combur 10 M 100 STR - EsДокумент2 страницыCombur 10 M 100 STR - EsVictor Manuel Parra Torres50% (4)
- Chemistry TДокумент2 страницыChemistry TMoni ShaОценок пока нет
- Momentum, Impulse and Energy: Pre-Reading: KJF 9.1 and 9.2Документ27 страницMomentum, Impulse and Energy: Pre-Reading: KJF 9.1 and 9.2lady jazzmineОценок пока нет
- Logarithmic and Trigonometric TablesДокумент104 страницыLogarithmic and Trigonometric Tableshillerphoebe100% (1)
- ASTM E1012 - 2005 - For Axial Force ApplicationДокумент11 страницASTM E1012 - 2005 - For Axial Force ApplicationSarvesh MishraОценок пока нет
- Department of Mathematics & Statistics MATH 0863 - PrecalculusДокумент6 страницDepartment of Mathematics & Statistics MATH 0863 - PrecalculusRax UtaОценок пока нет
- Rock Class CriepiДокумент3 страницыRock Class Criepirizka rahmadani100% (1)
- Junior High School Department: Caldwell Adventist AcademyДокумент3 страницыJunior High School Department: Caldwell Adventist Academyrosanie remotinОценок пока нет
- Calibration of A Pressure GaugeДокумент7 страницCalibration of A Pressure Gaugeneutron_8788% (17)
- Cunge1969 PDFДокумент27 страницCunge1969 PDFArun GangwarОценок пока нет
- Motion Mountain: The Adventure of PhysicsДокумент1 366 страницMotion Mountain: The Adventure of PhysicsGoembaОценок пока нет
- Kinetic Energy Equals: A. Mass X Velocity B. Mass X Velocity C. Mass X Acceleration D. Mass X VelocityДокумент108 страницKinetic Energy Equals: A. Mass X Velocity B. Mass X Velocity C. Mass X Acceleration D. Mass X Velocitynathaniel villanuevaОценок пока нет
- Flash Point by Tag Closed Cup Tester: Standard Test Method ForДокумент12 страницFlash Point by Tag Closed Cup Tester: Standard Test Method ForAngel MonteroОценок пока нет
- WRC368Документ33 страницыWRC368fahid masoodОценок пока нет
- Science States of Matter JeopardyДокумент34 страницыScience States of Matter Jeopardyapi-254830778Оценок пока нет
- (1993) Compressible Flow in A Capillary (Tube) PDFДокумент20 страниц(1993) Compressible Flow in A Capillary (Tube) PDFJing-Wu DongОценок пока нет
- Sgsits ManualДокумент91 страницаSgsits ManualJoyita BiswasОценок пока нет
- Free Standing Tension StructuresДокумент255 страницFree Standing Tension StructuresJoaquin VieraОценок пока нет
- MercerizationДокумент2 страницыMercerizationLohit MohapatraОценок пока нет
- Sss Royal Touch Laundry BrochureДокумент2 страницыSss Royal Touch Laundry BrochureMd. Badrul IslamОценок пока нет
- Control Theory II - Homework 1-Chapter 1: 1 ProblemsДокумент2 страницыControl Theory II - Homework 1-Chapter 1: 1 ProblemsSantiago Orellana CОценок пока нет