Академический Документы
Профессиональный Документы
Культура Документы
Validasi Vit C PDF
Загружено:
FeslyAnugerahAriestaPayungИсходное описание:
Оригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Validasi Vit C PDF
Загружено:
FeslyAnugerahAriestaPayungАвторское право:
Доступные форматы
Volume: 2: Issue-3: July-Sept-2013
Copyrights@2013
Received: 17th July-2013
Revised: 24th July-2013
ISSN: 2278-0246
Accepted: 25th Aug-2013
Coden: IJAPBS
www.ijapbs.com Research Article
QUANTITATIVE ASSAY EVALUATION OF VITAMIN C FROM FORMULATED TABLETS:
APPLICATION ON RP-HPLC AND UV-SPECTROPHOTOMETRY
Raju Chandra* Sujeet Kumar, Sanjay Singh, Keshav Dutt Sharma, Md. Naushad Alam and Daleep Verma
Department of Pharmaceutical Chemistry, Dolphin PG Institute of Biomedical and Natural Sciences,
Dehradun-248001, Uttarakhand, India
*Author to whom correspondence should be addressed. Email: rajvimalchandra@gmail.com
ABSTRACT: Assay is an important method for the accurate quantity determination of drug analyte in the
formulated products. For which a rapid simple sensitive isocratic reproducible reversed phase-high performance
liquid chromatographic technique has been developed for the quantitative assay evaluation of ascorbic acid. The
chromatographic separation of ascorbic acid samples were performed on a C18-column by isocratic elusion at the
column 250C. The mobile phase was used methanol and water in the ratio of 50:50 (v/v). The flow rate was 1.0
ml/min. The maximum peak area was resolved at 260 nm. The limit of detection and limit of quantification were
0.031 and 0.093 g/mL by RP-HPLC and 0.3 and 0.9 g/mL by UV-Spectrophotometry, respectively. Good results
were obtained with respect to linearity R2=0.997 by RP-HPLC and R2=0.983 by UV-Spectrophotometry. The mean
recoveries in inter-day and intra-day for RP-HPLC were calculated 99.98 % and 100.2 % and for UVSpectrophotometry 99.08 % and 99.37 %. The method was validated for linearity, accuracy, repeatability, limit of
detection (LOD) and limit of quantification (LOQ). The method is simple, accurate and precise and was successfully
applied to the determination of ascorbic acid from formulated products.
Key words: Ascorbic acid, RP-HPLC, UV-Spectrophotometry, Method Validation.
INTRODUCTION
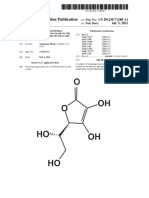
Ascorbic acid is a naturally occurring organic compound (fig. 1.). It is widely distributed in nature. Naturally this is
obtained from fruits and vegetables. It is in pure form a white crystalline powder. Its impure form appears yellow
colour. It dissolves in water to give mildly acidic solutions [1]. Ascorbic acid is one form "vitamer" of vitamin C.
Chemically, it is exists in D-ascorbic acid form which does not occur in nature. It may be synthesized artificially [2].
It is an essential part of human life. The IUPAC name of ascorbic acid is 2,3-dihydro-Lthreohexo-1,4-lactone.
Commonly ascorbic acid is known as vitamin C. The ascorbic acid in the form of vitamin C have various major
biologically activities such as maintenance of the organism, prevention of vitamin C deficiency (scurvy), promotion
of collagen biosynthesis, inhibition of melanogenesis and antioxidation [3-8] etc. Commercially ascorbic acid in
various formulations is available such as tablets, injections, syrups and capsules etc. Assay is an important method
for checking the commercially formulated products. There are various reported methods have been validated for the
simultaneous estimation of vitamin C by high-performance liquid chromatography using an UV detector [9-16]. The
main aim of this study is to develop and validate a new assay method for checking the quality and quantity of
ascorbic acid from formulated tablets.
HO
HO
H
HO
OH
Figure. 1. Chemical structure of ascorbic acid
International Journal of Analytical, Pharmaceutical and Biomedical Sciences
Available online at www.ijapbs.com
Page: 19
Chandra et al
IJAPBS
ISSN: 2278-0246
EXPERIMENTAL
Chemicals and reagents
The 99.7% pure drug of ascorbic acid, HPLC grade methanol, distilled water and 0.45 nylon filter membrane were
purchased from Merck India Ltd. Mumbai.
Instrumentation
A binary pump CYBERLABTM HPLC chromatograph and a double beam UV-Spectrophotometry were used for
analysis. The separation was on C18-column. The analyte were monitored with UV detector at 260 nm. The HPLC
was operated at isocratic eluation mode with 50:50 (v/v) methanol-water mobile phase. The flow rate of eluation was
1.0 min./mL. An ultrasonic sonicator was used for the sonication of mobile phase, standard solution and sample
solution.
Preparation of mobile phase
A methanol /water mobile phase was prepared. The ratio of methanol and water was inthe ratio of 50:50 (v/v). The
mobile phase were filtered through a 0.45m nylon membrane and degassed by ultrasonic sonication.
Preparation of stock solution
A 100 g/mL stock solution was prepared. In a 100 mL capacity volumetric flask 10 mg ascorbic acid was mixed
with mobile phase and make up it to 100 mL volume. The stock solution was filtered with 0.45m nylon filter
membrane and degassed by sonication.
Preparation of sample solution
The 20 tablets (Brand name Celin, label claim: 500 mg ascorbic acid per tablet) were weighed accurately and
crushed by mortar pistol. The crushed tablets were mixed well, and then an equivalent amount of 10 mg was
transferred into a small conical flask and extract with methanol/water 50:50 (v/v) mobile phase. The extract was
filtered into a 100 ml volumetric flask and the volume make up to 100 mL. Achieved aliquots was covered the
working concentration range 100 g/mL.
Preparation of calibration curve
A calibration curve was constructed by injecting the different concentration of serial dilutions (standard drug) 0.5,
1.0, 1.5, 2.0, 2.5 5.0, 10 g/mL in thrice replication. The calibration curve was obtained by plotting the average peak
areas against these different known concentrations.
Method validation
The various method validation parameters were performed according to international conference harmonization
guide lines [17-19] such as system suitability, Linearity, limit of detection (LOD) and limit of quantification (LOQ)
and accuracy.
System suitability test
The system suitability was checked to insure that, the system is working correctly. The system suitability parameters
peak area, retention time, resolution factor, theoretical plates, and tailing factor were checked according to
international conference harmonization guide line. This test was performed during development of the method. This
test was performed by injecting the standard mixture in n=5 replicates.
Linearity
The linearity ranges in both methods were 0.5-10 g/mL. The linearity ranges refers to the highest and lowest
quantity of the analyte that the method can detect with an appropriate amount of accuracy and linearity [20]. The
linearity was calculated by regression analysis.
Limit of detection (LOD) and limit of quantification (LOQ)
The Limit of detection (LOD) and limit of quantification (LOQ) were calculated using the equations, LOD =3.3/s
and LOQ =10./s. Where is the standard deviation of y-intercept and s is the slope of the curve.
Accuracy
Performance of the validated method is conformed by performing inter-day and intra-day recovery study at three
different concentration levels 1.5, 2.5 and 5.0g/mL. The three different concentration diluted from the stock
solution were added to an extract with a known content of ascorbic acid and the percentage recovery of the
respective constituents was calculated as R %=peak area of the drug in sample/peak area of the drug in standard x
100.
RESULTS
The mobile phase was select under reversed phase partition chromatographic condition. The mobile phase
developed was studied in order to achieve suitable system stability. The different ratios of (10:90, 20:80, 30:70,
40:60, 50:50) mobile phases compositions were tested at ambient temperature 25 0C [21]. The mobile phase
methanol/water in the ratio of 50:50 (v/v) was given suitable retention time and better resolution. There is no
interference with excipients (Fig.2).
International Journal of Analytical, Pharmaceutical and Biomedical Sciences
Available online at www.ijapbs.com
Page: 20
Chandra et al
IJAPBS
ASCORBIC ACID
3.55
94.00
[mAU]
ISSN: 2278-0246
71.20
48.40
-20.00
0.00
0.50
1.00
1.50
2.00
2.50
3.00
4.61
4.65
4.68
2.93
2.97
3.09
3.11
3.13
3.15
2.72
2.56
2.33
2.35
2.00
2.04
2.06
2.08
1.57
1.67
1.70
1.05
1.09
1.13
2.80
0.09
0.18
4.15
25.60
3.50
4.00
4.50
5.00
[min]
Figure. 2. Chromatogram of ascorbic acid
The system suitability test is statistically significant. The coefficient of variation percentage of each parameter is less
than 10 (Table 1.).
Table 1. Summary of system suitability test (n=5) for RP-HPLC
Parameters
Peak area
Retention Time
Resolution
Theoretical Plate
Tailing Factor
Mean
69964.26
3.56
1.544
2785.42
1.646
SD
1.6562
0.036007
0.027019
0.0983362
0.027016
CV%
0.002367
1.003818
1.749904
0.035304
1.641465
SE
() 0.74
() 0.01
() 0.01
() 0.43
() 0.01
SD=standard deviation CV%=coefficient of variation percentage SE=standard error ()
The linearity was measured by regression analysis. The linearity range of ascorbic acid was 0.5-50 g/mL for both
methods. Both methods reversed phase-high performance liquid chromatography (RP-HPLC) and UVSpectrophotometry results were showed good linearity with regression coefficient (R2) 0.997 and 0.983. The linear
regression equations for ascorbic acid are described in Table 1.
Table 2. Summary of linearity (n=3) and limit of detection (LOD) and limit of quantification (LOQ)
Parameters
Correlation range (g/mL)
Regression equation
Regression coefficient
LOD (g/mL)
LOQ (g/mL)
HPLC Method
0.5-10
Y=2726.x+475.2
0.997
0.031
0.093
UV Method
0.5-10
Y=0.033.x+0.079
0.983
0.3
0.9
The limit of detection (LOD) and limit of quantification (LOQ) for reversed phase-high performance liquid
chromatography (RP-HPLC) were reported 0.031 and0.093 g/mL and for UV-Spectrophotometry were reported 0.3
and 0.9 g/mL (Table 1.), respectively. The mean recoveries in inter-day and intra-day with RP-HPLC were
calculated 99.98 % and 99.77 % (Table 3.) and with UV-Spectrophotometry method were calculated 99.08 % and
99.37 % (Table 4.).
International Journal of Analytical, Pharmaceutical and Biomedical Sciences
Available online at www.ijapbs.com
Page: 21
Chandra et al
IJAPBS
ISSN: 2278-0246
Table 3. Recovery study in inter-day
UV-Spectrophotometry
Method
Recovery (%) Recovery
Recovery (%)
Recovery in g/mL
(w/w)
in g/mL
(w/w)
1.49
99.33%
1.48
98.66%
2.49
99.6%
2.47
98.80%
5.05
101%
4.99
99.80%
Mean=99.98%
Mean=99.08%
SD=0.89
SD=0.61
CV%=0.89
CV%=0.62
SD=standard deviation CV%= coefficient variation percentage
Added pure drug in
g/mL
1.5
2.5
5.0
RP-HPLC Method
Table 4. Recovery study in intra-day
UV-Spectrophotometry
method
Recovery in
Recovery (%) Recovery
Recovery (%)
g/mL
(w/w)
g/mL
(w/w)
1.5
1.499
99.33%
1.49
99.33%
2.5
2.48
99.2%
2.48
99.2%
5.0
5.01
100.2%
4.98
99.6%
Mean=99.77%
Mean=99.37%
SD=0.51
SD=0.20
CV%=0.51
CV%=0.20
SD=standard deviation CV%=coefficient variation percentage
Added pure drug in
g/mL
RP-HPLC method
DISCUSSION
The system suitability study is indicates that the applied method was suitable for the analysis. Wave length
selection is the primary need for the chromatographic analysis. To selection the wave length for ascorbic acid were
investigated in order to determine a suitable wavelength for the assay evaluation. The suitable wave length was found
260 nm. The selection of mobile phase is an important secondary basic need for chromatographic analysis. The
mobile phase was select under reversed phase-partition chromatographic conditions. The recovery results were
showed good accuracy with less than one coefficient variation percentage in both methods. Hence, the interday and
intraday results were significant.
CONCLUSION
In this study, an isocratic RP-HPLC and UV-Spectrophotometry method were developed and validated for the
simultaneous estimation of ascorbic acid from formulated ascorbic acid products. The experimental conditions in
both methods were optimized to provide high resolution and reproducible absorbance and peak area. The results
were statistically significant. In both methods coefficient variation and standard deviation were found less than one
from interday and intraday recovery study. From the obtained results this was observed the reverse phased- high
performance liquid chromatography (RP-HPLC) is an advance technique while UV-Spectrophotometry has few
limitations. Hence, both methods are suitable for the quantitative estimation. Any one method can be used for
quantitative estimation of ascorbic acid formulated products in pharma industries.
ACKNOWLEDGEMENT
Present study was supported by Dolphin Institute of Biomedical and Natural Sciences, Dehradun, Uttarakhand,
INDIA.
REFERENCES
[1] Velisek J, Cejpek K; Biosynthesis of food constituents: Vitamins. Water-soluble vitamins, part 1-a
review. Czech; Journal of Food Science 2007, 25: 49-64.
[2] Reichstein T, Grussner A, Oppenauer R; Synthesis of d- and l-Ascorbic Acid (Vitamin C); Nature
1933, 132-280.
International Journal of Analytical, Pharmaceutical and Biomedical Sciences
Available online at www.ijapbs.com
Page: 22
Chandra et al
IJAPBS
ISSN: 2278-0246
[3] Murad S, Grove D, Lindberg K.A, Reynolds G, Sivarajah A; Regulation of collagen biosynthesis by ascorbic
acid; Natlional Academy of Sciences 1981, 78: 2879-2882.
[4] Shao L, Li JT, Guo S, Wang SE, Guo DM, Determination of reduced and total vitamin C in beverage by high
performance liquid chromatography; Acta Nutrimenta Sinica 2007, 29: 192-194.
[5] McEoy GK, Drug 2000. American society of Health-System Pharmacists, Inc, Bethesda, MD 2000, 3328-3330.
[6] Pugliese PT, Lampley CB; Biochemical assessment of the anti-aging effects of cos- metic products; Journal of
Applied Cosmetology 1985, 3:129-138.
[7] Hagen TM, Ingersoll RT, Lykkesfeldt J, Liu J, Wehr CM, Vinarsky V; (R)--Lipoic acid-supplemented old rats
have improved mitochondrial function, decreased oxidative damage, and increased metabolic rate; FASEB
Journal 1999, 13:411-418.
[8] Liandong H, Li L, Zhaoliang L, Jianxue Y, Wei L; Determination of trace vitamin C by ion-pair HPLC with UV
detection in calcium gluconate and vitamin C compound oral solution; Journal of Chromatographic Science
2012, 50: 102-107.
[9] Din MSE, Eid M, Zeid AM; Development and Validation of RP- HPLC Method for Simultaneous Determination
of Ascorbic Acid and Salicylamide in their Binary Mixtures: Application to Combined Tablets; Journal of
Chromatogrphy and Separation Techniqes 2012, 3(5): 1-7.
[10] Adriana MM, Andre RB, Wilson JY, Eunice S, Telma MK, Maria VRV; Validation of HPLC stabilityindicating method for Vitamin C in semisolid pharmaceutical/cosmetic preparations with glutathione and
sodium metabisulphite, as antioxidants; journal of Talanta 2007, 71: 639-643.
[11] Marco I, Aurelia DT; Monitoring on the Presence of Ascorbic Acid in Not Prepacked Fresh Meat Preparations
by a Validated HPLC Method; Journal of Food Research 2012, 1: 22-31.
[12] Bogdan K, Danka O, Dobrina T; Validation of HPLC method for determination of antioxidant vitamin c
andvitamin B6 in food supplements and drugs; International Journal of Pharmacy and Pharmaceutical
Sciences 2012, 4(1): 300-304.
[13] Barbara A, Fernando C, Lidon A, Eduardo L; Ascorbic acid quantification in melon samples the importance of
the extraction medium for HPLC analysis; General Applied Plant physiology 2005, 31: 247-251.
[14] Waheed UZ, Mehwish A, Rabia R; Effect of Temperature Variations during Cooking and Storage on Ascorbic
Acid Contents of Vegetables: A Comparative Study; Journal of Chemical Society of Pakistan 2013, 35: 1-4.
[15] Artur M, Urszula P; Changes of dehydro ascorbic acid contentin relation to total content of vitamin c in
selected fruits and vegetables; Acta Sciintiarium Polonorum-Hortorum Cultus 2012; 11 (6): 169-177.
[16] Ana V, Tania GA, Ana SS, Helena SC; Ascorbic acid content in exotic fruits: A contribution to produce quality
data for food composition databases; Food Research International 2011, 44: 22372242.
[17] International Conference on Harmonization, Validation of Analytical Procedures: Text and Methodology,
Geneva, Switzerland, 2005, ICH Q2 (R1).
[18] ICH Harmonized Triplicate Guidelines ICH, Validation of Analytical Procedures: Consensus Guidelines, 1994,
ICH Q2A.
[19] ICH Harmonized Triplicate Guidelines, Validation of Analytical Procedures: Methodology, Consensus
Guidelines, 1996, ICH Q2B.
[20] Bognar A, Daood HG; simple in-line postcolumn oxidation and derivatization for the simultaneous analysis of
ascorbic acid and dehydroascorbic acids in foods; Journal of Chromatographic Science 2000; 38: 162-168.
[21] Harish CU, Ram KV, Santosh KS; Quantitative determination of bioactive 4-Hydroxy-alpha-Tetralone,
Tetralone-4-O-beta-D-glucopyranoside and ellagic acid in ammania baccifera (Limn.) by reverse-phase
high-performance liquid chromatography; Journal of Chromatographic Science 2013, 51: 21-25.
International Journal of Analytical, Pharmaceutical and Biomedical Sciences
Available online at www.ijapbs.com
Page: 23
Вам также может понравиться
- Carbohydrate Analysis: High Performance Liquid Chromatography and Capillary ElectrophoresisОт EverandCarbohydrate Analysis: High Performance Liquid Chromatography and Capillary ElectrophoresisОценок пока нет
- Calculation and Preparation of Standard Solutions in Food AnalysisДокумент5 страницCalculation and Preparation of Standard Solutions in Food AnalysisMahi HarixОценок пока нет
- Determination of Vitamin B6 in Foods by HPLCДокумент6 страницDetermination of Vitamin B6 in Foods by HPLCDaniel Dávila MartinezОценок пока нет
- Semester 3 Syllabus B PharmaДокумент18 страницSemester 3 Syllabus B Pharmaabhishek sharmaОценок пока нет
- IC - Principles and Troubleshooting DionexДокумент64 страницыIC - Principles and Troubleshooting Dionexwwwyyyzzz100% (2)
- Sigma Aldrich Grading ChartДокумент2 страницыSigma Aldrich Grading Chartjm06100% (1)
- GC-MS-MS Analysis of Pesticide Residues in Green Tea Extracted by QuEChERSДокумент1 страницаGC-MS-MS Analysis of Pesticide Residues in Green Tea Extracted by QuEChERSAmerican Lab100% (1)
- Analysis Method For Starch: Japan Customs Analysis MethodsДокумент3 страницыAnalysis Method For Starch: Japan Customs Analysis Methodsrusli308Оценок пока нет
- Absorbance and Fluorescence Spectroscopies of Green Fluorescent ProteinДокумент24 страницыAbsorbance and Fluorescence Spectroscopies of Green Fluorescent ProteinMadel Tutor ChaturvediОценок пока нет
- Dissolution MethodsДокумент216 страницDissolution MethodsPUBG MOBILE - FANS CLUBОценок пока нет
- HPLC Column Cleaning and RegenerationДокумент2 страницыHPLC Column Cleaning and RegenerationOrc PharОценок пока нет
- D 1387 - 89 R02 - Rdezodc - PDFДокумент2 страницыD 1387 - 89 R02 - Rdezodc - PDFJail MartinezОценок пока нет
- Quantitative Analysis of Reducing Sugars in Sugar Preparations Consisting of Sugar and DextrinДокумент6 страницQuantitative Analysis of Reducing Sugars in Sugar Preparations Consisting of Sugar and DextrinMuztika Andriana RahmawatiОценок пока нет
- Advanced Pharmaceutical AnalysisДокумент4 страницыAdvanced Pharmaceutical AnalysisRezaul RazibОценок пока нет
- Finlay MacRitchie - Concepts in Cereal Chemistry-CRC Press (2010) PDFДокумент192 страницыFinlay MacRitchie - Concepts in Cereal Chemistry-CRC Press (2010) PDFEdward PinedaОценок пока нет
- Determination of Trichloroacetic Acid Using HPLCДокумент11 страницDetermination of Trichloroacetic Acid Using HPLCLucia PuspitasariОценок пока нет
- Integrating My Results in ChemStation - 121008Документ50 страницIntegrating My Results in ChemStation - 121008William QuiroaОценок пока нет
- 15 Whatman Pricelist 2014 INRДокумент93 страницы15 Whatman Pricelist 2014 INRguita riefОценок пока нет
- ISO 17025 IntroДокумент6 страницISO 17025 Introakash batraОценок пока нет
- Theory of HPLC Quantitative and Qualitative HPLC PDFДокумент24 страницыTheory of HPLC Quantitative and Qualitative HPLC PDFArRashidRashidОценок пока нет
- SM 4500 Cloro ResidualДокумент14 страницSM 4500 Cloro ResidualDiego AngeloОценок пока нет
- A Review On Step-by-Step Analytical Method ValidationДокумент13 страницA Review On Step-by-Step Analytical Method ValidationMd MoinulОценок пока нет
- Making Sense of The LCMS Data Differences - David WeilДокумент65 страницMaking Sense of The LCMS Data Differences - David WeilKhoranaОценок пока нет
- TOCV WS操作说明书 英文版Документ262 страницыTOCV WS操作说明书 英文版scribd birdОценок пока нет
- EuSalt AS008-2005 Potassium - Flame Atomic Absorption Spectrometric MethodДокумент4 страницыEuSalt AS008-2005 Potassium - Flame Atomic Absorption Spectrometric MethodRuth Patinggi LPОценок пока нет
- Limit Test For Heavy MetalsДокумент19 страницLimit Test For Heavy MetalsnutrimakeОценок пока нет
- 15%PerAcetic Acid Birlox-15P Rev.1Документ2 страницы15%PerAcetic Acid Birlox-15P Rev.1Sushil Jadhav100% (1)
- Vitamin Analysis: Definition and ImportanceДокумент18 страницVitamin Analysis: Definition and ImportanceManjusha KondepudiОценок пока нет
- Analisis Quimica Cuantitativa - Daniel HarrisДокумент15 страницAnalisis Quimica Cuantitativa - Daniel HarrisLucia CoxОценок пока нет
- Technical Specifications HPLC - ProminenceДокумент4 страницыTechnical Specifications HPLC - ProminenceMaría José OrtegaОценок пока нет
- A Review On Stability Indicating HPLC Method DevelopmentДокумент19 страницA Review On Stability Indicating HPLC Method DevelopmentppiccoliniОценок пока нет
- Synthesis and Antibacterial Activity of 1-MonolaurinДокумент5 страницSynthesis and Antibacterial Activity of 1-Monolaurini love chubzОценок пока нет
- USP Medicines Compendium - Eflornithine Topical Cream - 2013-01-16Документ3 страницыUSP Medicines Compendium - Eflornithine Topical Cream - 2013-01-16amin138irОценок пока нет
- Fulltext TbarsДокумент115 страницFulltext TbarsWny SimatupangОценок пока нет
- Volumetric Determination of Total Aldehydes in Citrus OilДокумент4 страницыVolumetric Determination of Total Aldehydes in Citrus OilAnida Maria Moraes Gomes100% (1)
- Erweka Wet Granulator FGS - ManualДокумент28 страницErweka Wet Granulator FGS - ManualSilvia BaruchОценок пока нет
- Ftir and Gc-Fid CharacterizationДокумент8 страницFtir and Gc-Fid CharacterizationIpeghan Otaraku100% (1)
- Solvent Polarity Table - Miller's HomeДокумент6 страницSolvent Polarity Table - Miller's HomeJoseph CannilloОценок пока нет
- Agilent AAS Calibration MethodДокумент6 страницAgilent AAS Calibration Methodashif28Оценок пока нет
- HPLCДокумент170 страницHPLCmaddukuri jagadeesh babuОценок пока нет
- Cleaning Reverse Phase ColumnДокумент2 страницыCleaning Reverse Phase ColumnhbordasmОценок пока нет
- Instrument Analysis: Analysis Carbohydrate Contained in Palm Sugar by High Performance Liquid Chromatography (HPLC)Документ7 страницInstrument Analysis: Analysis Carbohydrate Contained in Palm Sugar by High Performance Liquid Chromatography (HPLC)Muhammad FahmiОценок пока нет
- Spectrophotometric Determination of Drugs & Pharmaceuticals Using Cerium (IV) As Oxidant and Amaranth Dye As Analytical ReagentДокумент9 страницSpectrophotometric Determination of Drugs & Pharmaceuticals Using Cerium (IV) As Oxidant and Amaranth Dye As Analytical ReagentIOSRjournalОценок пока нет
- PRESERVATIVESДокумент25 страницPRESERVATIVESimran aliОценок пока нет
- Patent Liposomas Vitamina CДокумент20 страницPatent Liposomas Vitamina CJomertron100% (1)
- Indoor Marijuana Lab Results (Dogwalker & Blackdog)Документ8 страницIndoor Marijuana Lab Results (Dogwalker & Blackdog)Teresa Mahoney100% (1)
- Aspirin SynthesisДокумент48 страницAspirin SynthesisPaolo PepsОценок пока нет
- Waters AquityДокумент92 страницыWaters AquityPeter KrasnovОценок пока нет
- FTIR, NIR and Raman SpectrosДокумент7 страницFTIR, NIR and Raman SpectrosananedallОценок пока нет
- Quantitative Determination of Citric and Ascorbic Acid WДокумент3 страницыQuantitative Determination of Citric and Ascorbic Acid Wblackjiriu100% (1)
- LaMotte 7191-01 Peracetic Acid Drop Count Titration Kit InstructionsДокумент3 страницыLaMotte 7191-01 Peracetic Acid Drop Count Titration Kit InstructionsPromagEnviro.comОценок пока нет
- Peracetic Acid Assay MerckДокумент73 страницыPeracetic Acid Assay Merckhmtlion100% (1)
- PDFДокумент9 страницPDFCésar CuadraОценок пока нет
- Applications of Solid-Phase Microextraction in Food AnalysisДокумент28 страницApplications of Solid-Phase Microextraction in Food AnalysisAnonymous xGc8MRRysОценок пока нет
- Erythropoietin Concentrated Solution (1316)Документ5 страницErythropoietin Concentrated Solution (1316)Mulayam Singh YadavОценок пока нет
- Insrumental Analysis ManualДокумент62 страницыInsrumental Analysis ManualFemola97Оценок пока нет
- Care Maintenance and HPLC Column TroubleshootingДокумент52 страницыCare Maintenance and HPLC Column TroubleshootingducngoctrinhОценок пока нет
- Tips and Tricks HPLC Troubleshooting AgilentДокумент30 страницTips and Tricks HPLC Troubleshooting AgilentverajaОценок пока нет
- Water ActivityДокумент19 страницWater ActivitySiri NalОценок пока нет
- Jurnal Sediaan Nasal PDFДокумент4 страницыJurnal Sediaan Nasal PDFFeslyAnugerahAriestaPayungОценок пока нет
- PCT Tablet PDFДокумент8 страницPCT Tablet PDFFeslyAnugerahAriestaPayungОценок пока нет
- Jurnal Sediaan Mata PDFДокумент15 страницJurnal Sediaan Mata PDFFeslyAnugerahAriestaPayung100% (1)
- Isb Jurnal Alkaloid PDFДокумент5 страницIsb Jurnal Alkaloid PDFFeslyAnugerahAriestaPayung100% (1)
- 2008 Qi Yulan PDFДокумент287 страниц2008 Qi Yulan PDFFeslyAnugerahAriestaPayungОценок пока нет
- 1214 Full PDFДокумент6 страниц1214 Full PDFFeslyAnugerahAriestaPayungОценок пока нет
- Research Article: Development and Evaluation of Microemulsions For Transdermal Delivery of InsulinДокумент7 страницResearch Article: Development and Evaluation of Microemulsions For Transdermal Delivery of InsulinFeslyAnugerahAriestaPayungОценок пока нет
- Formulation and Evaluation of Mucoadhesive Buccal Tablets of Ondansetron HCLДокумент15 страницFormulation and Evaluation of Mucoadhesive Buccal Tablets of Ondansetron HCLFeslyAnugerahAriestaPayungОценок пока нет
- Suppositories PHR308Документ20 страницSuppositories PHR308FeslyAnugerahAriestaPayungОценок пока нет
- Thesis On Fast Dissolving TabletsДокумент4 страницыThesis On Fast Dissolving Tabletsmichellespragueplano100% (1)
- Marketing DemoДокумент14 страницMarketing DemoShamim Ahamed ShakilОценок пока нет
- Kythera S-1 Ex. 99.1Документ471 страницаKythera S-1 Ex. 99.1darwinbondgrahamОценок пока нет
- Sales Consultant in Columbia SC Resume Stephen KingДокумент2 страницыSales Consultant in Columbia SC Resume Stephen KingStephenKing8Оценок пока нет
- ParkinsonДокумент24 страницыParkinsonSandy AgustianОценок пока нет
- Sulfonamidas PDFДокумент58 страницSulfonamidas PDFPablo AguilarОценок пока нет
- Gopal Satyanarayan Karva: Formulation Research ScientistДокумент4 страницыGopal Satyanarayan Karva: Formulation Research ScientistgopalОценок пока нет
- Leadership and Management in Pharmacy Practice-Taylor & Francis, CRC Press (2014)Документ340 страницLeadership and Management in Pharmacy Practice-Taylor & Francis, CRC Press (2014)SolomonОценок пока нет
- Dulcolax PDFДокумент12 страницDulcolax PDFAnica BesmonteОценок пока нет
- DistrofiaДокумент48 страницDistrofiaMD IurieОценок пока нет
- Metabical: Pricing, Packaging, and Demand Forecasting For A New Weight-Loss DrugДокумент4 страницыMetabical: Pricing, Packaging, and Demand Forecasting For A New Weight-Loss DrugKishan BhalotiyaОценок пока нет
- Pharma Manufacturing Company in LucknowДокумент8 страницPharma Manufacturing Company in LucknowAvail HealthcareОценок пока нет
- Seizure Freedom With More Than One Antiepileptic Drug: Linda J. Stephen & Martin J. BrodieДокумент3 страницыSeizure Freedom With More Than One Antiepileptic Drug: Linda J. Stephen & Martin J. BrodieAbi ZakiОценок пока нет
- Final Business Plan Q3D: Elemental Impurities IWG Dated 1 October 2014Документ3 страницыFinal Business Plan Q3D: Elemental Impurities IWG Dated 1 October 2014hamoumОценок пока нет
- Potassium - Sparing DiureticsДокумент12 страницPotassium - Sparing Diureticsrhimineecat71Оценок пока нет
- Mefenamic Acid Is A Non-SteroidalДокумент4 страницыMefenamic Acid Is A Non-SteroidalAnkan PalОценок пока нет
- Tapentadol ParacetamolДокумент2 страницыTapentadol ParacetamolMohammad Shahbaz Alam100% (1)
- NO Produk: List Harga Dexa Per Oktober 2020Документ6 страницNO Produk: List Harga Dexa Per Oktober 2020Rina RaehanaОценок пока нет
- Index From Desperate HousewivesДокумент9 страницIndex From Desperate HousewivesPickering and ChattoОценок пока нет
- Asthma: Rochelle M. Nolte, MD CDR Usphs Family MedicineДокумент56 страницAsthma: Rochelle M. Nolte, MD CDR Usphs Family MedicineJoan Marie Lechado InoviaОценок пока нет
- 2013 Colon Targeted Drug Delivery System of TizanidineДокумент7 страниц2013 Colon Targeted Drug Delivery System of TizanidineGanesh BharskarОценок пока нет
- Economic Analysis PharmaДокумент19 страницEconomic Analysis PharmaPooja SajanОценок пока нет
- Generic Name: Quinupristin/Dalfopristin - Injection (Kwin-ue-PRIS-tin/DAL-foe-PRIS-tin)Документ4 страницыGeneric Name: Quinupristin/Dalfopristin - Injection (Kwin-ue-PRIS-tin/DAL-foe-PRIS-tin)Jrose CuerpoОценок пока нет
- Plasma Protein BindingДокумент2 страницыPlasma Protein BindingKavish ChhajedОценок пока нет
- Dosis MimsДокумент21 страницаDosis MimsYulia Putri CarlianaОценок пока нет
- Drug Study AmoxicillinДокумент4 страницыDrug Study AmoxicillinKaren T. Celetaria75% (12)
- Nama - NenG SITI)Документ2 страницыNama - NenG SITI)Siti SyarifahОценок пока нет
- Clinical Pharmacy PHR 405: Chapter 1: General ConsiderationsДокумент24 страницыClinical Pharmacy PHR 405: Chapter 1: General ConsiderationsSamiul Alam Rajib100% (1)
- Jaipuria Institute of Management, Vineet Khand, Gomti Nagar Lucknow - 226 010Документ9 страницJaipuria Institute of Management, Vineet Khand, Gomti Nagar Lucknow - 226 010Aman Srivastava100% (1)
- Misoprostol PDFДокумент5 страницMisoprostol PDFEduardОценок пока нет