Академический Документы
Профессиональный Документы
Культура Документы
Accurately Calculate Nitrogen Requirement For Pressure Purging
Загружено:
Suleyman HaliciogluОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Accurately Calculate Nitrogen Requirement For Pressure Purging
Загружено:
Suleyman HaliciogluАвторское право:
Доступные форматы
Accurately Calculate Nitrogen Requirement for Pressure
Purging
Filed in Technical Papers December 4th, 2011
Olaleye S Adio MEng CEng MIChemE
GECL Oil & Gas Consultant
olaleye.adio@glocalecl.com
The start-up of process plants containing hydrocarbon feed streams are usually preceded by creating
an inert atmosphere within the system. One of the options of creating this inert atmosphere is via
pressure purging, using nitrogen.
Pressure purging is based on using nitrogen to inert a system to low oxygen concentration below
which a flammable atmosphere is not sustainable. To achieve this, nitrogen is used to raise the
pressure of the system (e.g vessel, heat exchanger, piping etc) from initial condition, which is usually
atmospheric, to a chosen value (x barg). With availability of pressure control from the nitrogen header
into the system, the pressure of the system does not have to reach the nitrogen system pressure.
After pressurizing (to x barg), the system is vented back to the initial (atmospheric) condition. This
pressure-venting cycle is repeated until the required inert condition is achieved within the system.
The following steps are used to determine number of pressure-venting cycles and nitrogen
requirement for pressure purging to achieved required inert O2 concentration
1. Choose or calculate inert O2 concentration required (vol %).
2. Calculate number of cycles (pressure-venting) based on initial & inert (final) oxygen
concentration, initial system pressure and chosen nitrogen pressure.
3. Calculate quantity of nitrogen per cycle to determine total nitrogen required.
Step 1. Choose or calculate inert O2 concentration required (vol%)
The first stage is to determine what level of O2 is acceptable within the system for the hydrocarbons
being introduced. As a rule of thumb 9 vol% Oxygen is below the Minimum Oxygen Concentration
(MOC) vol% required for complete combustion of hydrocarbons, whilst 6 vol% is still sufficient for
incomplete combustion. As such most systems are assumed to be safe at approximately 4 5 vol%
O2 .
If the required inert O2 concentration is not known, then the MOC for the hydrocarbon stream must be
calculated. A value below the MOC is then selected as the inert O2 concentration.
The minimum oxygen concentration required by a gas can be calculated from the equation:
Page 1 of 5
Accurately Calculate Nitrogen Requirement for Pressure
Purging
Flammability data for some more common gases is provided below
Flammability Limits of Some Common Hydrocarbons
Compound
LFL
UFL
%vol in Air %vol in Air
Compound
LFL
UFL
%vol in Air %vol in Air
Benzene
1.2
7.8
Ethane
12
Butane, n-Butane
1.6
8.4
Ethanol
19
n-Butanol
1.4
11.2
Ethylbenzene
1.0
7.1
Carbon Monoxide
12
75
Ethylene
2.7
36
Cyclobutane
1.8
11.1
Gasoline
1.4
7.6
Cyclohexane
1.3
7.7
n-Heptane
1.05
6.7
Cyclohexanol
n-Hexane
1.1
7.5
Cyclohexanone
9.5
Isobutane
1.8
9.6
Cyclopentane
1.5
9.5
Kerosene Jet Fuel
0.7
4.9
Cyclopropane
2.4
10.4
Methane (Natural Gas)
4. 5
15
Decane
0.8
5.4
Naphthalene
0.9
5.9
Diesel
0.6
7.5
Octane
Diethanolamine
13
iso-Octane
0.79
5.94
Diethylamine
1.8
10.1
Pentane
1.5
7.8
Toluene
1.2
6.75
Page 2 of 5
Accurately Calculate Nitrogen Requirement for Pressure
Purging
Example 2 Calculate the MOC for a HC mixture; 60 vol% CH4, 30 vol% C2H6 and 10 vol% C3H8.
Assume complete combustion 0.6CH4 + 0.3C2H6 + 0.1C3H8 + 2.75O2 > 1.5CO2 + 2.5H2O
Note: Volume fractions ratio of 6:3:1 is equal to mole fractions ratio of 6:3:1 on ideal gas molar volume
basis
Methane LFL 4.5%vol in Air, Ethane LFL 3%vol in Air, Propane LFL 2.15%vol in Air
Step 2. Calculate number of cycles required to achieve chosen oxygen
concentration
It can be proved analytically that the number of cycles (n) is related to the initial and final oxygen mole
fractions (yo and yn) and the initial (LOW) and HIGH pressure used for purging by the equation
Note: The above relationship assumes pure nitrogen is used for inerting. It should also be noted that
the number of cycles is independent of the vessel volume. As such a 2m3 vessel will require the same
number of cycles as a 50m3 vessel.
Example 3 Calculate the number of cycles required to inert an atmospheric vessel down to
2%vol O2, using a Nitrogen pressure of 2.27barg
An atmospheric vessel will be at PL = 1.01325bara and yo = 0.21; based on Air of 79% N2 and 21% O2.
with yn = 0.02 and PH = 3.283bara,
From a design point of view, nitrogen pressure (e.g 2.27barg) can always be chosen to ensure that
number of cycle is a whole number, such as 2, 3, 4 etc. If nitrogen pressure is constrained and
number of cycle is not a whole number then cycles should be rounded up to the next whole number,
such as a 1.4 cycles should be designed as 2 cycles.
Step 3. Calculate quantity of nitrogen required to achieve chosen oxygen
concentration
To determine the total amount of nitrogen required, the nitrogen per cycle needs to be
calculated. Total nitrogen is then calculated as the number of cycles multiplied by nitrogen/cycle.
Page 3 of 5
Accurately Calculate Nitrogen Requirement for Pressure
Purging
Example 4 Calculate the total quantity of nitrogen in Nm3/hr required to achieve the inert
atmosphere in example 3 for a vessel and associated piping of 15m3 at 25C and Nitrogen
temperature of 37C
One approach that has been used to calculate the quantity of nitrogen is to determine the quantity per
cycle as
However, this approach is only a good estimation when the system and nitrogen temperature are
equal but not equal to zero degrees Celsius (given that Normal gas volume is defined at 1.01325bara
& 0C) and is even less accurate when the nitrogen temperature is not equal to the temperature of the
system to be inerted.
A more accurate approach will be to apply a material & energy balance to determine the number of
moles of nitrogen required to achieve the required pressure in the vessel.
To determine the quantity of nitrogen more accurately, use the following steps
Step 3.1 Calculate the number of kmols in the vessel and piping system at initial
conditions
Using PV = nRT
Step 3.2 Iteratively determine the mixture temperature and number of moles in the
vessel at high pressure of 3.283 bara
This step involves calculating the number of kmols in the vessel at the high pressure end of the
cycle. However, as the equation is dependent on temperature in the system at the high pressure and
this final temperature is not known due to difference in Nitrogen and system pressure, the calculation
becomes iterative.
The basis of the iteration is to
3.2.1 Guess a value for final temperature
3.2.2 Calculate number of kmols in vessel based on high pressure and (guessed) final temperature
3.2.3 Determine number of kmols N2 introduced into system (kmols in 3.2.2 initial kmols)
Page 4 of 5
Accurately Calculate Nitrogen Requirement for Pressure
Purging
3.2.4 Check energy balance heat gained by initial kmols in system (0.61314 kmols) = heat lost by
kmols of N2 introduced.
3.2.5 If energy balance in 3.2.4 is satisfied, then correct value of temperature and N2 kmols is
obtained. If not, repeat steps 3.2.1 to 3.2.4 with new value of temperature.
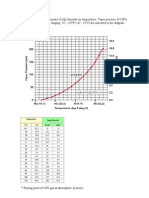
3.2.6 Calculate Nm3 of N2 using Normal molar gas volume of 22.4 Nm3/kmol. The calculation can be
set up in excel and solved for temperature using goal seek. An iteration table is also showed below for
the case being considered here:
Data: N2 : Cp = 1.04 kJ/kg & RMM = 28kg/kmol, Air: Cp= 1.006 kJ/kg K & RMM =
28.96 kg/kmol
Guess T (C) kmols @ kmols of N2
3.283bara
Heat gained Heat lost by N2
by Air (kJ)
(kJ)
Energy
balance (kJ)
31.00
1.9474
1.3343
107.18
233.13
125.95
34.00
1.9284
1.3153
160.77
114.90
-45.86
32.50
1.9379
1.3247
133.97
173.59
39.62
33.25
1.9331
1.3200
147.37
144.14
-3.23
33.19
1.9335
1.3204
146.30
146.49
0.19
Using the last line of data at temperature of 33.19C each calculation step from 3.2.2 to 3.2.6 is
detailed below
Compare the 59.16Nm3 to the nitrogen value of 68.10Nm3 determined from the first approximate
method in this example, there is a conservative 15% over estimation.
This approach thus provides an accurate way of determining the nitrogen requirement for pressure
purging.
Page 5 of 5
Вам также может понравиться
- Accurately Calculate Nitrogen Requirement For Pressure PurgingДокумент7 страницAccurately Calculate Nitrogen Requirement For Pressure PurginglouayОценок пока нет
- Calculation Sheet For PurgingДокумент7 страницCalculation Sheet For PurgingAkintoye Asaolu100% (1)
- N2 PurgingДокумент5 страницN2 PurgingSubbarayan SaravanakumarОценок пока нет
- Nitrogen CalcДокумент1 страницаNitrogen CalcSomayajula Suryaram100% (2)
- Nitrogen Calculation AirgasДокумент1 страницаNitrogen Calculation AirgasJack Sapp100% (1)
- Drying of Pipelines Prior To Commissioning - CEPS A.SДокумент6 страницDrying of Pipelines Prior To Commissioning - CEPS A.Sim4uim4uim4uim4uОценок пока нет
- Pressure Testing and Purging CalculatorДокумент2 страницыPressure Testing and Purging Calculatoramacathot06100% (5)
- Calculation Nitrogen Consumption For Nitrogen BlanketingДокумент1 страницаCalculation Nitrogen Consumption For Nitrogen Blanketingasep saefulloh100% (1)
- Blowdown Calc FormulaeДокумент1 страницаBlowdown Calc FormulaeWayne MonneryОценок пока нет
- Nitrogen PurgingДокумент4 страницыNitrogen PurgingSaeid Rahimi MofradОценок пока нет
- Volume of Nitrogen Required For Testing & Purging ContainersДокумент2 страницыVolume of Nitrogen Required For Testing & Purging ContainersHgagselim SelimОценок пока нет
- Line Pack CalculationДокумент7 страницLine Pack CalculationRachan EngchanilОценок пока нет
- Calculation of Purge RateДокумент42 страницыCalculation of Purge RateMuhammad_Saim_Ahmed100% (8)
- Purging of PipelineДокумент7 страницPurging of PipelineJenniferNealОценок пока нет
- Line Sizing ReportДокумент7 страницLine Sizing Reportmichael chiwuezeОценок пока нет
- PSV Sizing - Two Phase (Two-Points)Документ2 страницыPSV Sizing - Two Phase (Two-Points)Saeid Rahimi MofradОценок пока нет
- Erosional Velocity Ratio CalculationДокумент1 страницаErosional Velocity Ratio Calculationvijay_jvОценок пока нет
- 16" Utue Ka-Ubit PP Gas Pipeline Nitrogen Purging ProcedureДокумент17 страниц16" Utue Ka-Ubit PP Gas Pipeline Nitrogen Purging ProcedureRichard Nwachukwu92% (12)
- Blowdown Calculation Final Rev B1 PDFДокумент23 страницыBlowdown Calculation Final Rev B1 PDFAwwalin Bocah Ilang100% (1)
- Converstion For KG Per HR in To MMSCFDДокумент8 страницConverstion For KG Per HR in To MMSCFDJagan BoseОценок пока нет
- PSV Blocked Fire Relief Valve CasesДокумент21 страницаPSV Blocked Fire Relief Valve CasesTun Naing WinОценок пока нет
- Pressure Testing and Purging CalculatorДокумент2 страницыPressure Testing and Purging Calculatordeion29Оценок пока нет
- Drying and Nitrogen ProcedureДокумент5 страницDrying and Nitrogen ProcedureAdaghara67% (3)
- Thermal Relief Rate Calculation: Page 17 of 38Документ1 страницаThermal Relief Rate Calculation: Page 17 of 38Ascend032100% (1)
- Ognition PSV Sizing Calculation SpreadsheetДокумент2 страницыOgnition PSV Sizing Calculation Spreadsheetger80Оценок пока нет
- 10 Gas Conditioning SystemДокумент24 страницы10 Gas Conditioning SystemRavi Sankar VenkatesanОценок пока нет
- Pipe and Vessel PurgingДокумент4 страницыPipe and Vessel PurgingAnonymous v5uipH100% (2)
- Depressuring - A Practical GuideДокумент30 страницDepressuring - A Practical GuideCastorlexОценок пока нет
- Calculation of Orifice Plate Hole DiameterДокумент3 страницыCalculation of Orifice Plate Hole DiameterhiuОценок пока нет
- Blowdown CalculationДокумент1 страницаBlowdown CalculationecavalinОценок пока нет
- Leak Rate CalculationДокумент1 страницаLeak Rate Calculationtenjoe_38Оценок пока нет
- Vertical Knock Out Drum With Wire Mesh For NДокумент1 страницаVertical Knock Out Drum With Wire Mesh For Nteegala_1950% (2)
- API - STD - 521 Fire Gas ExpansionДокумент3 страницыAPI - STD - 521 Fire Gas Expansioneliealtawil100% (1)
- Gas - Line SizingДокумент2 страницыGas - Line SizingSeema SiddiquaОценок пока нет
- KG Per HR To Nm3 Per HR To MMSCFD & Vice VersaДокумент3 страницыKG Per HR To Nm3 Per HR To MMSCFD & Vice VersaAbhay BhoirОценок пока нет
- Pipeline Pressurisation & de Pressurisation CalculationДокумент4 страницыPipeline Pressurisation & de Pressurisation Calculationhatakerobot50% (2)
- Purge FlareДокумент15 страницPurge FlareMuhammadОценок пока нет
- Purge With Nitrogen-1Документ5 страницPurge With Nitrogen-1Yawar QureshiОценок пока нет
- Gas Pipeline Blowdown TimeДокумент6 страницGas Pipeline Blowdown TimeWalid Ben HuseinОценок пока нет
- Nitrogen BlanketingДокумент21 страницаNitrogen Blanketingrvkumar61Оценок пока нет
- Cold Venting As API 521 To Discharge HCДокумент4 страницыCold Venting As API 521 To Discharge HCkenoly123100% (1)
- Purging Hydrocarbon PipelinesДокумент7 страницPurging Hydrocarbon Pipelinesgan_raoОценок пока нет
- Smith Industries WaterBath Indirect Heater PDFДокумент29 страницSmith Industries WaterBath Indirect Heater PDFcassindromeОценок пока нет
- Velocity in Pipeline For Natural GasДокумент8 страницVelocity in Pipeline For Natural GasNakkolop100% (1)
- PSV SizingДокумент3 страницыPSV SizingpavanОценок пока нет
- Venting As Per Api 2000Документ6 страницVenting As Per Api 2000Joshue Adele100% (1)
- Depressurization Calculation..Документ12 страницDepressurization Calculation..Faber TrujilloОценок пока нет
- Pressure Safety Valve Sizing Calculation Rev 01Документ3 страницыPressure Safety Valve Sizing Calculation Rev 01Athiphap SrisupareerathОценок пока нет
- Relief Load Calculation For Fire Case - As Per API 521Документ1 страницаRelief Load Calculation For Fire Case - As Per API 521ktejankarОценок пока нет
- UFC-85 Plants For Urea Fertilizer Projects: Recommended DosageДокумент8 страницUFC-85 Plants For Urea Fertilizer Projects: Recommended Dosagevaratharajan g rОценок пока нет
- Accurately Calculate Nitrogen RequirementДокумент6 страницAccurately Calculate Nitrogen RequirementRachel BaileyОценок пока нет
- Geclearning: Step 1. Choose or Calculate Inert O Concentration Required (Vol%)Документ4 страницыGeclearning: Step 1. Choose or Calculate Inert O Concentration Required (Vol%)sandy4u2k2Оценок пока нет
- Material/Energy Balance of Combustion at Steady StateДокумент26 страницMaterial/Energy Balance of Combustion at Steady StateIlyas HassanОценок пока нет
- Flare Emission CalculationsДокумент8 страницFlare Emission CalculationsMubarik AliОценок пока нет
- Removing NitrogenДокумент6 страницRemoving NitrogenKhuram MaqsoodОценок пока нет
- Technical Note TN-106: Correction Factors, Ionization Energies, and Calibration CharacteristicsДокумент12 страницTechnical Note TN-106: Correction Factors, Ionization Energies, and Calibration Characteristicssrinivas1108Оценок пока нет
- 1 A Novel ApproachДокумент10 страниц1 A Novel Approachamir8100Оценок пока нет
- AUTO 303 PPT 1 Combustion EngineeringДокумент69 страницAUTO 303 PPT 1 Combustion EngineeringpatiencetinotendajengwaОценок пока нет
- Impact Testing RequirementsДокумент24 страницыImpact Testing Requirementschuckhsu124850% (2)
- ASME Impact Test RequirementДокумент6 страницASME Impact Test RequirementJ.SIVIRAОценок пока нет
- ASME-Notch-Toughness & Supplementary Essential VariablesДокумент16 страницASME-Notch-Toughness & Supplementary Essential VariablesSuleyman Halicioglu100% (2)
- WPS PQR Checklist 2014Документ2 страницыWPS PQR Checklist 2014Sebastian Rajesh100% (1)
- Pressure Testing (Hydrostatic Pneumatic) Safety GuidelinesДокумент7 страницPressure Testing (Hydrostatic Pneumatic) Safety GuidelinesSuleyman HaliciogluОценок пока нет
- Effect of Calcium and Magnesium Treatment On Steel WeldabilityДокумент7 страницEffect of Calcium and Magnesium Treatment On Steel WeldabilitySuleyman HaliciogluОценок пока нет
- PWHT Procedure For Petrol Steel - CoreДокумент9 страницPWHT Procedure For Petrol Steel - CoreSuleyman HaliciogluОценок пока нет
- Calcium Treatment of SteelДокумент4 страницыCalcium Treatment of SteelSuleyman HaliciogluОценок пока нет
- Inclusions in Steel by Calcium TreatmentДокумент89 страницInclusions in Steel by Calcium TreatmentSuleyman HaliciogluОценок пока нет
- MQA CaTreatmentInLCAKSteelДокумент16 страницMQA CaTreatmentInLCAKSteelSuleyman HaliciogluОценок пока нет
- Heat Exchanger Tube To Tubesheet ConnectionsДокумент19 страницHeat Exchanger Tube To Tubesheet ConnectionsSuleyman Halicioglu100% (2)
- Tube Expansion Issues & MethodsДокумент4 страницыTube Expansion Issues & MethodsSuleyman HaliciogluОценок пока нет
- Handbook of Sulphuric Acid ManufacturingДокумент25 страницHandbook of Sulphuric Acid ManufacturingSuleyman Halicioglu50% (2)
- FEM StandardsДокумент330 страницFEM StandardsVic Sargent100% (2)
- Worksheet#1Документ2 страницыWorksheet#1Vince HernándezОценок пока нет
- Tables and Index Thermodynamics Cengel 7E-2Документ118 страницTables and Index Thermodynamics Cengel 7E-2tomtom9649Оценок пока нет
- Propane Gas and Liquefied GasДокумент10 страницPropane Gas and Liquefied GasJ.SIVIRAОценок пока нет
- Section 5 - LPG266 - 154562Документ11 страницSection 5 - LPG266 - 154562handoyo_eko20017573Оценок пока нет
- Comparison Between Gasoline Diesel CNG LPG 1664300610Документ19 страницComparison Between Gasoline Diesel CNG LPG 1664300610richard100% (1)
- Liquefied Gas Tanker Familiarisation CourseДокумент266 страницLiquefied Gas Tanker Familiarisation CourseGeorge Ponpyka100% (6)
- 16 - High Hydrogen Fuels in Fired HeatersДокумент17 страниц16 - High Hydrogen Fuels in Fired HeatersBamrung SungnoenОценок пока нет
- Flow Assurance Training Course-1 Underlined (IOGPT)Документ108 страницFlow Assurance Training Course-1 Underlined (IOGPT)Abid Hussain100% (5)
- Requestn About Power Plant Project (Included Technical & Commercial For Informatio Part)Документ6 страницRequestn About Power Plant Project (Included Technical & Commercial For Informatio Part)Chakravarthi NagaОценок пока нет
- Manufacturing of MAN PDFДокумент126 страницManufacturing of MAN PDFChetal BholeОценок пока нет
- Natural Gas PresentationДокумент30 страницNatural Gas PresentationEng Said ElsayedОценок пока нет
- 001 Carbon Dioxide General DIOM Manual PDFДокумент46 страниц001 Carbon Dioxide General DIOM Manual PDFargenis alezonesОценок пока нет
- Ardrox 907p Aerosol Sds Ver1Документ7 страницArdrox 907p Aerosol Sds Ver1Aneesh KumarОценок пока нет
- The Natural Gas Industry in Trinidad and TobagoДокумент22 страницыThe Natural Gas Industry in Trinidad and TobagoRich Ian100% (1)
- Cambridge IGCSE™ Chemistry 4th Edition (Bryan Earl, Doug Wilford) (Z-Library)Документ18 страницCambridge IGCSE™ Chemistry 4th Edition (Bryan Earl, Doug Wilford) (Z-Library)Muhammad TayyabОценок пока нет
- PS Checalc 2 PrelimsДокумент10 страницPS Checalc 2 PrelimsPrincess Janine CatralОценок пока нет
- Thermal Conductivity of GasesДокумент2 страницыThermal Conductivity of GasesFlorian_AngererОценок пока нет
- Student Handout - Advanced Gas Tanker Training PDFДокумент136 страницStudent Handout - Advanced Gas Tanker Training PDFPrassanna Venkatesh100% (2)
- Gas Processors Suppliers Association GPSA Eng (Bookos - Org) - 701-821-1!60!41-60Документ20 страницGas Processors Suppliers Association GPSA Eng (Bookos - Org) - 701-821-1!60!41-60Karen VlОценок пока нет
- Gpsa 2Документ6 страницGpsa 2David Cortez PeraltaОценок пока нет
- Gas Processing 4.1 Natural Gas and Gas Processing OperationДокумент14 страницGas Processing 4.1 Natural Gas and Gas Processing OperationJulia HartОценок пока нет
- MQДокумент7 страницMQalaa icesОценок пока нет
- Lab Report Experiment 1 PDFДокумент4 страницыLab Report Experiment 1 PDFJanelle MelchorОценок пока нет
- LPG Properties RKSДокумент33 страницыLPG Properties RKSmukund madhav100% (2)
- Flow Coefficient (CV) and Calculation of Flow Through ValvesДокумент7 страницFlow Coefficient (CV) and Calculation of Flow Through ValvesMikeCroОценок пока нет
- Freeze R600a MSDSДокумент12 страницFreeze R600a MSDSmazhar13191Оценок пока нет
- Tabla - Masa MolecularДокумент2 страницыTabla - Masa MolecularWulfranoSánchezОценок пока нет
- The General Properties of HydrocarbonsДокумент9 страницThe General Properties of Hydrocarbonsjimbob8888Оценок пока нет
- Comparison Study Between DWSIM and Aspen Plus SoftwareДокумент11 страницComparison Study Between DWSIM and Aspen Plus Softwarepavanchem61Оценок пока нет
- Vol 2 SNG Peak Shaving HandbookДокумент38 страницVol 2 SNG Peak Shaving HandbookCaterine Jara CeaОценок пока нет