Академический Документы
Профессиональный Документы
Культура Документы
Hum. Reprod. Update 2003 Shaw 583 605
Загружено:
randyprayogoАвторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Hum. Reprod. Update 2003 Shaw 583 605
Загружено:
randyprayogoАвторское право:
Доступные форматы
Human Reproduction Update, Vol.9, No.6 pp.
583605, 2003
DOI: 10.1093/humupd/dmg041
Terminology associated with vitrication and other
cryopreservation procedures for oocytes and embryos
J.M.Shaw1 and G.M.Jones
Monash Institute of Reproduction and Development, Monash University, Clayton, Victoria 3168, Australia
1
To whom correspondence should be addressed. E-mail: Jill.Shaw@Med.Monash.edu.au
Key words: cooling/cryopreservation/embryos/oocytes/vitrication
Introduction
Cooling to low subzero temperatures (cryopreservation), is used to
store human oocytes, embryos, sperm and gonadal tissues. There
are several well-established cryopreservation protocols for human
sperm and embryos, most of which were established >15 years ago
and are classied as `slow cooling' or equilibrium cooling
procedures (Elliott and Whelan, 1977; Leibo, 1986; Mazur,
1990). In these slow cooling procedures, cryoprotectant (antifreeze) is added to the solution surrounding the cell and the sample
then cooled at a rate dictated by the size and permeability of the
cell. The thawing rate and protocol for cryoprotectant removal also
needs to be appropriate for the cooling procedure and the cell's
characteristics. The outcome is thought to be best when the rate of
cooling allows an equilibrium to be established between (i) the rate
of water loss from the cell (dehydration) and (ii) the rate at which
this water is incorporated into growing, extracellular, ice crystals.
For human oocytes and embryos, the optimum rate is thought to lie
between 0.3 and 1C/min, for sperm 10C/min, hamster cells
100C/min and for red blood cells >1000C/min, i.e. several orders
of magnitude faster (Elliott and Whelan, 1977; Critser and
Mobraaten, 2000). The period of slow cooling is usually ended
once the temperature has fallen to between 30 and 80C, after
which they are moved to a low temperature storage tank (<
130C). This review does not aim to give details on how to
perform slow cooling procedures or their outcomes, as several
other articles and reviews already do this (Elliott and Whelan,
1977; Friedler at al., 1988; Karow and Critser, 1997; Critser and
Russell, 2000; Shaw et al., 2000b.
Oocytes, embryos and cells can also be cryopreserved by nonequilibrium procedures such as `vitrication', `rapid cooling' and
`ultrarapid cooling'. We will collectively refer to the nonequilibrium protocols as rapid protocols. Rapid protocols all differ
from slow protocols in that (i) most dehydration and cryoprotectant permeation takes place before cooling commences, and (ii)
cooling is usually performed in a single step in which the specimen
is cooled directly from a temperature >0C to low subzero
temperatures (<130C). Rapid protocols are commonly used for
animal embryos as they can give as good or better outcomes than
`slow cooling', yet use less equipment, less liquid nitrogen and less
processing time. Rapid protocols started to be evaluated for human
oocytes and embryos in the 1980s (Trounson, 1986) but were, until
recently, rarely used by human IVF units. Now several large IVF
units have started to publish results for rapidly cooled oocytes and
embryos. The outcomes (survival and pregnancies) of rapid
cooling need further improvement, but the convenience and low
cost of rapid cooling is likely to ensure increasingly widespread
use of these protocols for human embryos.
Because rapid protocols have not been widely used, relatively
few clinicians and embryologists are fully conversant with the
technical terms specically used for rapid cooling protocols. This
can make it difcult to read and understand papers describing any
rapid cooling (`vitrication', `rapid cooling' and `ultrarapid
cooling') protocol. This has in part arisen because many of the
terms used in cryopreservation have been acquired from low
temperature physics, chemistry and engineering. This paper aims
to allow readers to more easily and effectively read and interpret
papers on cryopreservation literature, and in particular papers
Human Reproduction Update Vol. 9 No. 6 European Society of Human Reproduction and Embryology 2003; all rights reserved
583
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Over the past 30 years many cryopreservation procedures have been applied to oocytes, embryos, sperm, ovarian
and testicular tissue. Over this time many, often specialized, terms have been developed for all aspects of these procedures. This can make it difcult for readers who are not familiar with the terminology or protocols to compare
and evaluate different procedures. This paper describes the main cryopreservation procedures, the terminology
associated with them, and briey explains the underlying physical, chemical and biological processes. The aim is to
help readers understand and interpret other papers on slow cooling, rapid cooling, ultrarapid cooling and vitrication.
J.M.Shaw and G.M.Jones
Table I. General classication of current cryopreservation procedures
Slow cooling (equilibrium)
procedures
State during cooling
Ice present (usually seeded
at 5 to 9C)
Cooling rate usually 0.3 to
1C/min to between 30 and
80C then >200C/min
State during warming
Ice present
Warming rate usually 4 to
100C/min
Rapid cooling (non-equilibrium) procedures
Non-vitrifying
Vitrifying
Variable amount of ice
Glass-like state with
Glass-like state with
(spontaneous seeding)
no ice
no ice
Cooling rate depends on container and cooling strategy usually 200 to 20 000C/min (tested range 5 to 130 000C/min)
(e.g. Rall and Fahy, 1985; Rall, 1987; Hochi et al., 2001; Arav et al., 2002)
More ice forms
Ice forms as the glass
Maintains glass-like,
de-vitries
ice-free state
Warming rate depends on container and warming strategy usually 300 to >12 000C/min (e.g. Rall and Fahy, 1985;
Rall, 1987; Hochi et al., 2001; Arav et al., 2002)
Water, ice and physical changes within cryopreservation
solutions
Cooling to low subzero temperatures (e.g. 196C the temperature
of liquid nitrogen) without protection will kill most cells
immediately from damage caused by the growth of intracellular
ice crystals (see Tables VIXI which list denitions of these
terms). Cryopreservation procedures are designed to minimize
damage caused by ice crystal formation and growth. To understand
how cryopreservation procedures achieve this goal, it is helpful to
know how they modify ice crystal formation and growth.
Ice dynamics in pure water
Ice formation is a stochastic process that starts when individual
water molecules join to form microscopic ice nuclei (also called
embryos or germs). In pure water the highest temperature at which
ice can form at normal pressure is 0C, but ice nuclei form only
very reluctantly at this temperature, with the result that nuclei do
not usually form until the temperature falls below 0C. Solutions
that remain free of ice, even though they are below the temperature
at which ice can form, are said to be in a supercooled state. The
lowest temperature at which pure water can be held without
freezing is thought to be ~40C, but ice normally forms at
temperatures between 5 and 15C through spontaneous (due to
homogeneous or heterogeneous nucleation) or induced (by
seeding) ice nucleation. Once an ice nucleus has formed, further
water molecules can very readily bond onto this frozen surface,
allowing it to grow in size. Ice nuclei and the smallest ice crystals
are too small to see even with a microscope, but these usually grow
into rst microscopic and subsequently macroscopic ice crystals.
If ice forms in water at the highest possible temperature it only
spreads slowly and results in relatively few large crystals. When
ice forms in a supercooled solution it grows more rapidly. When
the ice forms there is a release of heat (the latent heat of fusion or
crystallization), as the change from liquid to crystal releases
energy. The amount of energy released may be considerable and
commonly brings the temperature of the whole sample back up to
0C. With pure water the temperature will remain at 0C until the
freezable water has formed ice, after which it will re-equilibrate
with the ambient temperature. Following the crystallization of
pure water, very little remains unfrozen even at temperatures as
high as 5C (>80% ice) or 10C (>90% ice) (Karow, 2001).
When water crystallizes, the space occupied by each individual
water molecule increases with the result that 1 g of ice (density
0.92) occupies more space than 1 g of water (density 1).
Consequently ice oats.
The shape of the ice crystals is largely dictated by the shape and
behaviour of individual water molecules. Normal ice, i.e. that
which develops when water freezes, is also known as hexagonal
ice (or Ice 1h) because multiple groups each comprising six
individual water molecules bond to each other to form sheets.
Although crystals can grow along either the a-axis (parallel to the
hexagonal face or basal plane) or the c-axis (perpendicular to the
hexagonal face or prism plane), the individual molecules most
readily bind to incomplete hexagons, thereby extending existing
sheets. Water molecules do not attach onto the `side' of a sheet. As
a result crystals form characteristic angles. To thaw all this ice the
temperature has to rise above the freezing point. Pure water
therefore melts at 0C, the same as the highest temperature at
which ice crystals can rst form. During the thawing process, the
change from crystal to liquid requires energy input.
Ice dynamics in cells and aqueous solutions containing solutes
Adding salts and other solutes to pure water lowers the temperature at which ice forms/melts. There is no limit to how low the
freezing/melting point can be depressed by solutes, with the result
that solutions with very high concentrations may never form ice,
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
describing rapid cooling procedures. To achieve this aim, in the
following sections we have summarized the terminology for the
ice and physical events associated with cryopreservation (Tables I,
II, VI), the chemicals and solutions (Tables III, VII), and the
procedures and processing steps (Table VIII), briey outlined the
factors that inuence the outcomes of these cryopreservation
procedures (Tables IV, V, IX) and listed the equipment and
materials employed (Figure 1; Appendix 1, Table X). Words
associated with safety issues when working with liquid nitrogen
are also included (Appendix 2, Table XI). This review does not
aim to give details on how to perform rapid cooling procedures or
their outcomes, as several other articles and reviews already do
this (Shaw et al., 2000b; Vajta, 2000; Shaw and Kasai, 2001;
Kuleshova and Lopata, 2002).
584
De-vitrifying
Terminology for cryopreservation techniques
Table II. Temperature table
F
+37
+20
+16
+98
+68
+61
+2 to +6
0
5
+41
+32
+23
10
20
+14
4
30
35
40
22
31
40
80
112
90
130
130
202
150
196
197 to 210
273
238
320
349
459
Nitrogen vapour can be any temperature, but needs to be at low subzero temperatures (under 150C) for storage or rapid cooling.
DMSO = dimethylsulphoxide.
Table III. Glass-forming properties of some chemicals used in cryoprotectant solutions; expressed as the
minimum weight percentage, % wt/v (molarity in parentheses), of the chemical that has to be added to give
a glass-forming solution
Chemical
Fahy (1989)
Ali and Shelton (1993)
Penetrating?
Most effective glass formers
Butylene glycol (2,3-butanediol)a
Propylene glycol (1,2-propanediol)b
1,3-Butanediola
Dimethylsulphoxideb
Glycerol (1,2,3-propanetriol)b
1,3-Propanediola
Ethylene glycol (1,2-ethanediol)b
Acetamidea
Least effective glass formers
46
44
49
49
65
57
55
61
(3.0)
(4.0)
Yes
Yes
(5.0)
(5.0)
Yes
Yes
(6.5)
Yes
No
(5.1)
(5.7)
(5.4)
(6.3)
(7.1)
(7.5)
(8.9)
(10)
aNot
usually used for oocytes or embryos.
are routinely used for oocytes and embryos.
The chemicals are listed in order of effectiveness as glass formers from the most effective to the least
effective.
bThese
rather they will solidify into an ice-free (vitreous) state. Even when
salts and solutes are present, they rarely become incorporated into
the ice crystals themselves, because of the very specic shape and
bonding requirements of the growing ice crystals. Molecules other
than water molecules are instead excluded from the growing ice
crystals, and tend to accumulate between crystals or are physically
pushed ahead of an advancing ice front. The formation and growth
of ice crystals can, however, be modied by compounds such as
antifreeze proteins (O'Neil et al., 1998), ice blocking agents (e.g.
Wowk et al., 2000) and cryoprotectants.
Rapid cooling solutions for oocytes and embryos use ingredients
and procedures which aim to reduce or eliminate both intra- and
585
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Approximate human body temperature
Approximate room temperature
Temperature below which oocytes and embryos of chill-sensitive species can suffer lethal
damage, attributed to membrane phase transitions (e.g. Drobnis et al., 1993; Pollard and Leibo,
1994; Martino et al., 1996). Pure DMSO is frozen at this temperature
Temperature of domestic refrigerator
Freezing/melting point of pure water
Approximate temperature for intracellular ice formation in human oocytes (Trad et al., 1999).
Seeding of the external solution is most commonly performed at between 4 and 9C
Approximate temperature for intracellular ice formation in mouse embryos (Toner et al., 1991)
Temperature of domestic freezer; temperature at which a saturated sodium chloride solution
will freeze. Vitrication solutions stored at this temperature should remain liquid
Highest routinely used plunging temperature for slow cooled specimens
Plunging temperature routinely used for, e.g., materials slow cooled in DMSO
Plunging temperature routinely used for slow cooled ovarian tissue. The lowest temperature at
which pure water can be held as a supercooled liquid without freezing
Temperature of low temperature scientic freezers and approximate temperature of dry ice
(solid CO2), lowest plunging temperature routinely used for slow cooled specimens
Lowest temperature at which growth of damaging ice crystals during warming has been
documented (see e.g. Shaw et al., 1995a,b)
Temperature of ultralow-temperature electric freezers. Most solutions have glass transition
temperatures higher (warmer) than this
Temperature of coldest ultralow-temperature electric freezers currently available
Boiling point of liquid nitrogen at normal air pressure
Boiling point of liquid nitrogen exposed to a reduced pressure (see `nitrogen slush' in text)
Absolute zero; 0 on the Kelvin scale of absolute temperatures
J.M.Shaw and G.M.Jones
Table IV. Survival and development of 8-cell mouse embryos rapidly cooled (>500C/min) in vitrifying or de-vitrifying solutions is more dependent on the
warming rate than on the presence of ice crystals during warminga
Cryoprotectant
Physical state
after cooling
Warming rate and physical state during warming
Fast
(2500C/min)
Moderate
(300C/min)
Slow
(10C/min)
VS1 (~13 molal)
Vitried
Vitreous (glass)
2287.7% blastocysts
3% fetuses
Vitreous (glass)
80.7% blastocysts
De-vitried (ice)
0% blastocysts
90% of VS1 (~12 molal)
Vitried
De-vitried (ice)
8084.6% blastocysts
26% fetuses
De-vitried (ice)
87.4% blastocysts
De-vitried (ice)
0% blastocysts
aFrom Rall and Fahy (1985), Rall et al. (1987), Rall and Wood (1994).
The vitrication solution was termed `VS1' and was used either at full or reduced concentration (here 90% of VS1).
586
thought that extracellular ice remains outside the cell and does not
usually cross/penetrate the cell membrane (possibly because the
membrane has a layer of bound water). However, there is evidence
that the dynamics of ice inside the cell is different depending not
only on whether extracellular ice is present, but also on whether
the cells are present singly in suspension or as monolayers or when
gap junctions are present (Acker and McGann, 2003; Armitage
and Juss, 2003). In rapidly cooled straws, the solution nearest the
walls both cools and warms fastest. The difference can be
biologically signicant as embryos rapidly cooled in straws with 3
mol/l (22%) dimethylsulphoxide (DMSO) showed high survival
and development rates when located close to the edge of the straw
in a region which only formed ice on warming, but very poor
development if they were located in the middle of the straw where
ice formed both on cooling and warming (Shaw et al., 1991a).
Ice nuclei on their own are unlikely to cause damage to a cell,
even if they are located within it because they are so small (they
start as pairs of water molecules). However, water molecules near
an ice nucleus readily bond with it, allowing it to rapidly develop
into a large ice crystal that can cause signicant intracellular
damage (Miller and Mazur, 1976; Elliott and Whelan, 1977; Leibo
et al., 1978; Mazur, 1984, 1990; Acker and McGann, 2003). An
advancing ice front (crystal surface) binds water molecules, while
physically excluding other molecules; it therefore causes damage
in part because it acts like an agricultural plough but also because
this leads to the accumulation of salts, solutes and gases that can
have osmotic and toxic effects. This damage may be exaggerated
by the conformational change that makes individual H2O (water)
molecules each occupy a greater volume in the frozen than in the
liquid state. The magnitude of these forces is exemplied by old
mining practices in which rocks were split by repeated freeze
thaw cycles. Thawing frozen material will cause further damage in
particular if large ice crystals are present because the ice melts to
form areas of pure water, which can cause osmotic shock. There is
clear evidence that oocytes and embryos can be damaged both
during cooling and warming. Ice nucleation, ice crystal growth and
recrystallization does not just occur at around the melting point of
a solution, rather it occurs over a wide temperature range. For
example, if straws containing different solutions are moved from
liquid nitrogen to room temperature air or a waterbath at 37C,
those with vitrifying (ice-free) solutions will warm at a relatively
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
extracellular ice formation. These are discussed in greater detail
later in this review. All slow cooling procedures by contrast use
solutions which form ice, as the formation of ice crystals is integral
to the success of the slow cooling procedure.
An unprotected cell will form intracellular ice when cooled to
low zubzero temperatures. The ice undergoes uncontrolled growth
and forms large ice crystals which kill the cell. Slow cooling
procedures reduce the likelihood of this happening by letting
extracellular ice draw water out of the cell until so little free water
remains in the cell that only small (non-lethal) ice crystals can
form. This form of dehydration can only occur if ice is present in
the extracellular solution and does not form inside the cell. To
ensure that the solution forms ice before the cell, these procedures
rely on the difference in ice nucleation temperature between cells
and their surrounding solutions. Cells contain more solutes and
less free water than most physiological solutions in which they are
held. Ice therefore tends to form more readily in a physiological
solution than in cytoplasm. A solution containing solutes which is
cooled at a steady rate may, however, like pure water, supercool
and not nucleate until it reaches a temperature below that at which
the cytoplasm can nucleate. This is now known to be particularly
likely with oocytes, as those of several species start to form
intracellular ice at unusually high subzero temperatures (e.g. 5C)
both with and without added cryoprotectants (Toner, 1991; Trad
et al., 1999; Koseoglu et al., 2001). To ensure that extracellular ice
always forms at a temperature above that at which the cell can
spontaneously form intracellular ice, most slow cooling protocols
actively seed (induce ice formation in the solution) at temperatures
between 6 and 9C and thereby avoid supercooling. When these
cryoprotectant solutions are seeded the solution freezes and energy
is released (latent heat of fusion), raising the temperature to the
solution's melting point. But the temperature does not (as in pure
water) rise to and remain at this point, rather the it falls as the
solute concentration of the remaining unfrozen fraction rises, as
this lowers the solution's melting point. Human oocytes may be
particularly vulnerable to freezethaw damage during slow
cooling because they start to form intracellular ice at such high
temperatures (e.g. 5C) that they contain ice before the solution is
seeded. It is not known why this occurs but they may contain
structures or molecules that act as ice nucleators. Even now we do
not fully understand the interactions between ice and cells. It is
Terminology for cryopreservation techniques
Table V. Review showing how rapid cooling with vitrifying, de-vitrifying and non-vitrifying solutions/protocols affects the development of 816-cell mouse
embryos (comments in parentheses)
Reference
Cryoprotectant
Blastocyst formation (%)
Vitrifying solutions/protocols
Rall and Fahy (1985)
VS1 (100% = ~13 molal)
87.7 ( >2000C/min)
~82 ( <20C/min)
91 ( >2000C/min)
92.9 ( <20C/min)
8597 (strain dependent)
>95
4560
063 versus 3753
05 versus 010
882 versus 063
096 versus 7092
032 versus 4474
93100
Rall and Wood (1994)
VS3a (BSA)
Dinnyes et al. (1995)
Rall (1987)
Ishida et al. (1997)
Mukaida et al. (1998)
VS3a
VS3
EFS
30 versus 40%
30 versus 40%
30 versus 40%
30 versus 40%
30 versus 40%
7 mol/l EG
Bautista et al. (1997)
90% of VS1 (~12 molal)
~84.6 ( >2000C/min)
~80 ( <20C/min)
~88 ( >2000C/min)
~73 ( <20C/min)
~80 ( >2000C/min)
~60 ( <20C/min)
65
Rall and Fahy (1985)
85% of VS1 (~11 molal)
Rall and Fahy (1985)
80% of VS1 (~10.4 molal)
Trounson et al. (1988)
~26% DMSO (3.5 mol/l)
0.25 mol/l sucrose
33% DMSO (4.5 mol/l)
8.6% sucrose (0.25 mol/l)
30% glycerol, 0.7 mol/l sucrose
(from abstract only)
~26% DMSO (3.5 mol/l)
0.25 mol/l sucrose
25% EG (4.8 mol/l), 35% Ficoll
15% EG (3.08 mol/l), 40% Ficoll
10% EG (2.08 mol/l), 50% Ficoll
96
97
92
DMSO ~15% + sucrose (0.25 mol/l)
~47
Rall and Fahy (1985)
75% of VS1
Rall and Fahy (1985)
70% of VS1
Rall and Wood (1994)
VS3a 6% BSA
48% glycerol (6.5 mol/l)
~48 ( >2000C/min)
0 ( <20C/min)
~48 ( >2000C/min)
0 ( <20C/min)
Shaw et al. (1991b)
Krivokharchenko et al. (1996)
Wilson and Quinn (1989)
Kuleshova et al. (2001)
Kuleshova et al., 2001)
Kuleshova et al. (2001)
Protocols/solutions forming ice on cooling
Trounson et al. (1988)
6573
1871 (strain dependent)
26
8099
32 (per embryo frozen)
42 (per embryo transfer)
7588 (per embryo frozen)
59.390.2
34
87
62
8 overall
10/embryo transfer
75 ( 5C/min 20C/min)
In most studies, straws were warmed rapidly.
= cooling rate, = warming rate; DMSO = dimethylsulphoxide; EG = ethylene glycol; PROH = 1, 2-propanediol; G = glycerol; F = Ficoll; S = sucrose;
FS = Ficoll and sucrose; EFS = ethylene glycol and Ficoll and sucrose; GFS = glycerol and Ficoll and sucrose; BSA = bovine serum albumin.
uniform rate. Straws containing solutions with ice will initially
warm at the same rate as those with vitried contents, but from
temperatures of ~100 or 90C will warm to the solutions melting
point at increasingly slow rates relative to the vitried straw. Once
all ice has melted, the solution in the straw will again warm
rapidly. This pattern develops because, in addition to the energy
(heat) from the surroundings needed to raise the temperature, the
solution with ice has to take up extra energy to convert ice into
water. This implies that the temperature of a slow cooled straw
only has to rise to 100C (which can be reached within 5 s in air)
for changes in the ice to occur. Although there are many different
warming protocols for slow cooled specimens, it is possible to kill
up to 100% of embryos by using a warming protocol that is
inappropriate for the type of cryoprotectant or the cooling protocol
used (see e.g. Elliott and Whelan, 1977; Shaw et al., 1995a,b). In
samples of pure water the difference in density between ice (0.92
g/ml), the water that forms when the ice melts (1 g/ml) and the
remaining non-frozen fraction (>1 g/ml) can lead to separation in
particular in large samples in which the water and ice can oat to
the top, leaving the (now more concentrated) remainder at the
bottom. Gentle agitation of the vial or straw throughout the
thawing step can help minimize the separation. During cooling,
587
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
De-vitrifying solutions/protocols
Rall and Fahy (1985)
DMSO + FS
acetamide + FS
PROH + FS
EFS
GFS
Fetus formation (%)
J.M.Shaw and G.M.Jones
compounds other than the water can crystallize out of the solution.
If phosphate-buffered saline is placed in a normal domestic
freezer, crystallization/precipitation of some non-aqueous
components can occur. Once these components have come out
of solution they may not readily re-dissolve, with the result that the
overall ionic balance is different from that before freezing.
Free versus bound water and dehydration injury
In considering the dynamics of ice crystal growth, it is important to
recognize that in addition to free water, which crystallizes quite
readily, biological systems also contain `bound' water. The
`bound' water molecules are intimately hydrogen-bonded to the
atoms within molecules such as proteins, RNA, DNA or
membrane phospholipid head groups. These water molecules
form a substantial `coat', or hydration shell (one-third of their
weight) around the biological molecule and are essential for
maintaining their structure and function. Cryopreservation has to
try to remove free water to minimize damage from ice crystal
growth, without causing damage by uncontrolled removal of the
bound water providing structural support to proteins, DNA and
membranes.
Both slow and rapid cooling procedures reduce the likelihood of
the free water forming large (damaging) intracellular ice crystals
by enforcing both water loss (dehydration) and cryoprotectant
uptake. The presence of penetrating cryoprotectants and additives
such as sugars can help cells recover from dehydration. The cell
membrane comprises a phospholipid bilayer. Under normal
conditions the hydrophilic headgroups form the inner and outer
surface while the hydrophobic tail group lies between the two.
Drying can disrupt this arrangement (e.g. Bronshteyn and
Steponkus, 1993) by forcing the hydrophilic head groups to
coalesce or the hydrophobic portions to be exposed. Water
molecules form a hydration shell around proteins. The removal of
this shell can cause the irreversible collapse of the protein.
Cryoprotectants and sugars are thought to counteract dehydration
injury by acting as substitutes for water molecules at the surface of
proteins. Sugars can intercalate into the phospholipid head groups,
588
Chemicals and solutions used for cryopreservation
Small (<100 mol. wt) hydrophilic, membrane-penetrating compounds which hydrogen bond with water molecules and can form
hydrogen bonds, with e.g. a protein (in place of a water molecule),
are particularly effective as antifreeze agents. The strength of the
bonding can be measured as the release of heat (heat of solution)
when the cryoprotectant is added to the solution. The presence of
these compounds in an aqueous solution lowers the solution's
tendency to crystallize, and lowers the highest subzero temperature at which ice crystallization occurs. High concentrations of
cryoprotectant compounds make the solution form a wholly ice-
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
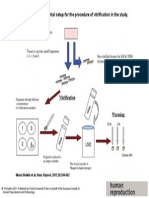
Figure 1. Liquid nitrogen and vapour storage tanks. In liquid storage tanks,
specimens are immersed in liquid nitrogen. In vapour storage tanks,
specimens do not contact liquid nitrogen. Some vapour storage tanks have
the coolant within the walls, not within the storage space. The insulation in
the walls and the size of the opening inuence both the storage capacity
(samples) and insulation properties.
making them less likely to undergo an irreversible phase transition
(Anchordoguy et al., 1987; Crowe et al., 1988). To minimize
dehydration injury, a cell needs to be equilibrated with
cryoprotectant before drying commences. In both slow and rapid
cooling, cells will die from ice formation if they are frozen with
too high water content. Very severe dehydration (<80% of the
original water content) can also cause damage. Optimal cryopreservation procedures therefore have to establish a balance between
(i) too high and too low water contents and (ii) having enough
cryoprotectant present to replace water around the cell molecules
without causing excess toxicity (see e.g. Szell and Shelton, 1987).
The way in which the cryoprotectant is loaded into a cell
appears to inuence the cell's ability to tolerate dehydration and
may explain why protocols in which the concentration of
cryoprotectant is increased gradually can be more effective than
addition in a single step. Most cryoprotectant solutions are at a
signicantly higher osmolarity than physiological solutions
(mouse sperm freezing solutions are the exception). Each time a
cell is placed in a solution with a higher osmolarity than the one at
which the cell is currently equilibrated, it will shrink. The extent to
which it shrinks and the rate at which it recovers reects the
difference in osmolarity between the old and the new solution, but
also the type of cryoprotectant, the presence of cryoprotectant
additives, temperature and cell type (Elliott and Whelan, 1977;
Critser et al., 1997; Paynter et al., 1997a, 1999a; Songsasen et al.,
2002). Critser et al. (1997) placed mouse oocytes in 1 mol/l DMSO
at 20C. These oocytes shrank within seconds, by 25% (to ~75% of
their original volume) through water loss across the membrane,
after which an inux of DMSO entered the cell replacing the lost
water, causing the oocyte to return to its original cell volume
within 2025 min. Mouse oocytes which were placed directly into
higher DMSO concentrations shrink more and those in 6 mol/l
DMSO shrank to 25% of their original size within a few seconds,
and took longer to return towards their normal size. It is often
difcult to assess the degree of dehydration because many cells
collapse or atten rather than shrink symmetrically, but is likely
that this extreme shrinkage results from the loss of both free and
some bound intracellular water, and may as a result be associated
with irreversible damage to the cell. Cells which are preequilibrated with a low cryoprotectant concentration (e.g. 0.51
mol/l) can be placed at very high cryoprotectant concentrations
without exhibiting this extreme shrinkage, and recover better from
the treatment. The bound water around the proteins and other
intracellular structures would already have been partially replaced
(and as a result be partially protected from further water loss) by
penetrating cryoprotectant.
Terminology for cryopreservation techniques
Components of cryopreservation solutions
Most cryopreservation solutions used for oocytes and embryos are
made up in a physiological solution to which is added: one or two
cryoprotectants capable of permeating the cell membranes (usually propanediol, DMSO ethylene glycol or ethylene glycol), one
additive that is non-penetrating to the cell membrane (usually a
sugar), and a protein (which improves handling characteristics,
membrane stability (Anchordoguy et al., 1987; Crowe et al., 1988)
and can reduce toxicity). The permeable cryoprotectants are all
toxic and can cause osmotic shock, particularly at high concentrations. Vitrication solutions commonly also contain a polymer
(e.g. Ficoll) to make the solution more likely to vitrify at a given
concentration of penetrating cryoprotectant. The permeating
cryoprotectants lower the freezing point, and replace some of the
bound water molecules in and around proteins, DNA and other
intracellular components. The non-penetrating additives stay
outside and aid in dehydration before and during preservation;
they may also help stabilize the membrane by stabilizing the
phospholipid headgroups. Compounds such as amino acids vary in
their effect. Studies on liposomes have shown that most amino
acids increased damage; some such as glycine and alanine were
protective in the presence of salts, while amino acids with charged
amine group side-chains were cryoprotective whether salt was
present or not (Anchordoguy et al., 1988). In general, cryoprotectant solutions used for slow cooling contain ~10% penetrating
cryoprotectant, whereas most of those used for rapid cooling or
vitrication contain >30% penetrating cryoprotectant. The concentration of non-penetrating sugar usually ranges from 0.1 to 1
mol/l (in the case of sugar this is 0.342 to 3.42 g/10 ml). It is
important to realize that the permeability of cell membranes to the
penetrating cryoprotectants varies greatly and depends not only on
which chemical it is and the temperature, but also which species
the cell is from and at what stage of development it is at (see e.g.
Paynter et al., 1997a,b; Paynter et al., 1999a,b).
Some cryopreservation procedures allow cells to be added
directly to the cryoprotectant and to also remove this solution, in a
single step, with physiological media. Most procedures for oocytes
and embryos, and in particular vitrication procedures, require
gradual or stepwise addition and gradual or stepwise dilution. The
composition of the solutions used before cells are placed in the
nal cryopreservation solution varies, but generally the aim is to
replace some of the bound water in the cell's proteins, DNA and
other components with a penetrating cryoprotectant. The concentration of the penetrating cryoprotectant is kept low to minimize
toxicity. The composition of the solutions used after warming also
varies but the aim is to ensure that all the penetrating
cryoprotectant is removed without causing osmotic shock.
It is not easy to control osmotic shock. The semipermeable
membrane that surrounds each cell lets water, cryoprotectants and
some other solutes cross these membranes, but not at the same
speed. Water is a very small molecule and crosses the membrane
more readily than the cryoprotectants currently used in cryopreservation. Cells which are equilibrated with cryoprotectant are
therefore at risk of osmotic injury when they are placed in a less
concentrated solution. In early slow cooling procedures this was
most commonly avoided by stepwise addition and dilution steps.
Rapid cooling and vitrication protocols use signicantly higher
cryoprotectant concentrations than the slow cooling procedures
(>4.5 mol/l as opposed to 12 mol/l). To counteract osmotic shock,
equilibration times for rapid cooling protocols are often short and
dilution is commonly performed in solutions in which an
impermeable non-toxic compound such as sucrose or trehalose
has been added to counteract osmotic stress. Following vitrication, the cells are particularly fragile and vulnerable to osmotic
shock (Pedro et al., 1997; Edashige et al., 1999), and there is
evidence of major changes in membrane permeability with the
result that normally impermeant molecules (e.g. propridium
iodide) can enter the cell (Kaidi et al., 1999; Newton et al.,
1999). It is possible that sodium chloride may enter the cell at this
time because replacing sodium chloride with choline chloride can
improve the post thaw outcome (Stachecki et al., 1998; 2002).
Serum and extracellular physiological solutions contain phosphates and glucose as well as high concentrations of sodium
chloride (e.g. 68126 mmol/l) and calcium chloride (e.g. 0.32.04
mmol/l) and a low concentration of potassium chloride (2.55.5
mmol/l) (Gardner and Lane, 2000). By contrast the cytoplasm of
embryos (see e.g. Baltz et al., 1997), or oocytes (e.g. Dick, 1978)
contains less sodium (~10 mmol/l) and calcium and more
potassium (70266 mmol/l).
Optimal cryopreservation procedures need to minimize osmotic
shock to the cell at all phases of the cryopreservation procedure.
The rate of cryoprotectant inux and efux is inuenced by the
species, cell type, stage of development, solution composition and
temperature. This makes it difcult to extrapolate pre- and postcryopreservation equilibration conditions from one cell type or one
species to another. Our own impression is that an optimized
equilibration procedure shrinks the embryo (often very rapidly)
before cooling, but after warming, the cells exhibit a gradual return
to their original size, and do not swell beyond their original size.
What are vitrication solutions?
Procedures in which a solution/specimen solidies to form a glasslike, or vitreous, state without any ice crystal formation during
cooling and remains in this state throughout the warming step are
called vitrication procedures. Whether vitrication will occur is
589
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
free (glass-like/vitreous) state when cooled. The capacity for
aqueous solutions with high concentrations of cryoprotectants to
reduce, or even abolish, ice nucleation forms the basis for their use
in vitrication solutions. Although there are many compounds
which can act as antifreeze agents, many are too toxic to use with
living cells, in particular at the concentrations which are needed
for vitrication to occur. As a result the only antifreeze chemicals
which are routinely used for vitrication or rapid cooling of cells
are polyols such as glycols (glycerol, propanediol, ethylenediol or
ethylene glycol), and DMSO. Other compounds such as alcohol
can also protect embryos from cryoinjury. However, all these
chemicals are toxic at the concentrations used in cryopreservation
solutions (usually >10%), so it is important that the composition of
the solution and the equilibration times and temperature are
balanced against the permeability characteristics and sensitivity of
each cell type (e.g. Shaw et al., 1991b; Paynter et al., 1997a,b).
Studies on liposomes indicate that the cryoprotective properties of
DMSO may be attributable to an electrostatic interaction between
its polar sulphoxide moiety and phospholipid membranes
(Anchordoguy et al., 1991), but also showed that at increased
temperatures there was a hydrophobic association between DMSO
and phospholipid bilayers (Anchordoguy et al., 1992).
J.M.Shaw and G.M.Jones
Table VI. Terms used to describe ice, physical changes and water movement during cryopreservation procedures
Activation energy
Annealing
Bound water
Bubble formation
Colligative properties
Cooling
Cracking
Critical cooling rate
Cryoinjury
Cuboidal ice
Dehydration
Dehydration curve
Dehydration injury
De-vitrication
Differential scanning
calorimeter
Eutectic point
Exosmosis
Fracture
Freezing
Freezing point
Glass
Glass transition
Heterogeneous
nucleation
Hexagonal ice
Homogeneous
nucleation
Hydration shell
590
The normal form of ice obtained when water freezes, also known as `Ice 1h'. Density 0.92. Protons are disordered but the
molecules form hexameric box-like structures (slight asymmetry 0.25% shorter in the c-direction) arranged as stacked sheets.
Classied in internet sites as `space group P63/mmc, 194; Laue class symmetry 6/mmm with open, all-gauche, low-density
structure, with lower packing efciency than simple cubic (~1/2) or face-centered cubic (~3/4) structures' (adapted from
http://www.lsbu.ac.uk/water/ice1h.html) (terms such as mmm and mmc form part of the nomenclature dening the symmetry of
crystals/space groups)
The spontaneous formation of ice nuclei not triggered by surfaces or impurities
It is estimated that proteins and DNA have to contain ~0.3 g water/g to remain functional. This water is not free, but bound
(e.g. to phosphates and bases) and is integral to the conformation of the biological molecule
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Crystallization
Here, in this context: the minimum amount of energy needed to transport water or other molecules across a cell membrane
Holding a specimen at a specic subzero temperature. Used in studies which aim to achieve a steady state of ice nucleation
(number and distribution) and ice crystal size and structure
Osmotically inactive water, usually bound to cell components such as proteins
On warming, solutions may contain bubbles. These form at advancing ice fronts, but may also develop when fractures or
cracks occur in frozen or vitried samples. Can be symptomatic of poor cryopreservation conditions (Shaw et al., 1988). It is
not known what the bubbles contain but they form as readily in normal and degassed solutions
Properties of a liquid that can be altered by the presence of a solute (molecules or ions), but are not dependent on the type of
solute (these are: vapour pressure, freezing point depression and boiling points, and osmotic pressure). All of these properties
ultimately relate to the vapour pressure
A decrease in temperature
Most frozen and vitried solids and the containers in which they are held become very brittle and prone to fracturing at low
temperatures. As cracking is most evident when a steep temperature gradient (large temperature difference) forms across the
specimen, it is most likely with fast cooling and warming (see e.g. Kasai et al., 1996; Poledna and Berger, 1996; Shi et al.,
1998; Liu et al., 2000; Arav et al., 2002)
Slow cooling at this rate dehydrates the cell without any intracellular ice formation, and can allow the intracellular concentration
of solutes and cryoprotectants to reach such high concentrations that the cell will vitrify when plunged into liquid nitrogen
Cryopreservation, because it inicts such a range of chemical, biological and physical insults, can damage almost any part of a
cell. Although cryopreservation can cause severe injury leading to instant death/lysis, several fully optimized protocols maintain
the cells/embryos viability and developmental potential (no statistical difference as compared to non-treated controls). See also
Table IX and text
In this context, refers to the transition of water molecules from a liquid state into ice. Crystallization releases heat (see heat of
fusion below). Crystals grow as spikes or ngers probably because the tips more effectively disperse the heat of fusion and
therefore grow faster than at surfaces
A metastable form of ice obtained, e.g., when water vapour condenses at very low temperatures (e.g. 130C) also known as
Ice 1c. Density 0.92. One of several less common molecular congurations which water molecules can adopt. All atoms have
four tetrahedrally arranged nearest neighbours and 12 second neighbours. Under some conditions cuboidal ice slowly converts
to hexagonal ice
Reducing the water content of a cell to reduce the likelihood of damaging intracellular ice formation when the cell is cooled
(see text on equilibrium and non-equilibrium methods and free versus bound water)
A graph plotting water content of cells (often assessed by measuring cell volume) against another parameter such as time in
the cryoprotectant solution, or temperature (during a slow cooling ramp)
Proteins and other cellular components are readily damaged by extreme water loss, as this removes water which is integral to
their conformation (see hydration shell). The hydrogen bonding properties of cryo- and lyo- protectants allow them to
reversibly substitute/replace the position/function of water molecules, thereby reducing injury
An event seen in vitried solutions, usually during warming, characterized by the formation of ice nuclei or ice crystals.
The physico-chemical causes and its effect on viability of frozen cells or tissues have not yet been fully established
(see e.g. Karlson, 2001).
Apparatus used to measure small changes in temperature (as calories). Sensitive enough to measure the release of heat
during ice crystallization, and the uptake of heat during melting
The minimum melting temperature of a mixture of two compounds. As an example: When DMSO is added to water, the
melting point becomes lower than for either of the pure ingredients (0C for water and +18C for DMSO). DMSO solutions
at concentrations routinely used for mouse embryo cryopreservation (e.g. 4.5 mol/l DMSO) are liquid even at <30C. It is also
the temperature at which a mixture of two compounds starts to melt, but in practice this rst melting is almost always
undetectable
Movement of water out of the cell by osmosis (because of a chemical gradient)
See cracking
The formation of ice crystals. The term freezing is therefore inappropriate for vitrication solutions which do not form ice
0C is the equilibrium freezing point for water, i.e. the temperature at which water, ice and vapour can coexist at atmospheric
pressure. Ice can nucleate, or be seeded at this temperature, but forms more easily at temperatures <0C
A solid-like phase which does not contain any ice nuclei or ice crystals. Glass formation can occur in aqueous solutions used
in cryopreservation (vitrication) under appropriate cooling conditions. It is solid, not just a supercooled, very viscous liquid
The temperature at which vitrifying solutions change to/from the solid, stable, glass-like state
The formation of ice nuclei triggered by surfaces or impurities
Terminology for cryopreservation techniques
Table VI Continued
Hydraulic conductivity
Hysteresis
Ice
Ice-axis
Ice nucleation
KedemKatchalsky
coefcient
Lp
Melting
Membrane phase
transition
Modelling
Nucleation/nucleation
temperature
Osmotic shock
Permeability
Phase diagram: phase
transition diagram
Re-crystallization
Reection coefcients
Supercooling
Td
Tg
Thawing
Thawing rate/warming
rate
Timetemperature
transformation curve
Tm
Triple point
Vapour pressure
Vapour pressure
depression
Vitrication
Warming
See `Glass' (above)
An increase in temperature
591
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Melting point/melting
point range
Movement of water across a membrane (Lp). Permeability
A solution is said to exhibit hysteresis if it melts at a temperature that is different from it's highest possible ice nucleation
temperature. Compounds such as antifreeze proteins can cause hysteresis. Not inuenced by supercooling
Ice can form in any of 12 different crystal structures, plus two amorphous states. The oxygen atom of each molecule is strongly
covalently bonded to two hydrogen atoms, while the molecules are weakly hydrogen-bonded to each other. In all solid ice the
water molecules are hydrogen-bonded to four neighbouring water molecules: there are two hydrogen atoms near each oxygen,
one hydrogen atom on each OO bond. When ice and water coexist at the freezing point, the amount of ice remains constant as
long as no heat is added or removed (e.g. Schulson, 1999). Heat is released as water changes from the liquid to crystal phase.
Energy needs to be added to convert ice back to water
Ice crystals have structure dictated by the shape of each water molecule and the way it bonds to neighbouring water
molecules. This leads to crystals with hexagonal symmetry with an a- and a c-axis
See homo- and heterogeneous nucleation and crystallization above (c.f. manual `seeding' as dened in Table VIII)
Membrane transport can be modelled using principles of non-equilibrium thermodynamics. KedemKatchalsky equations model
the ux of both water and cryoprotectant through a cell membrane when driven by concentration gradients. See e.g. Liu et al. (2000)
Hydraulic conductivity. See e.g. Liu et al. (2000)
Put simply, the process in which ice turns into water. As a solution melts there is a constant interchange of water molecules
between ice and water. The rate of exchange is lowest where the molecules form a at (planar) ice surface and highest where
the crystal structure is incomplete or damaged (e.g. at corners). Corners, or smaller crystals, usually melt at a temperature at
which a large planar crystal surface is stable. Energy (heat) has to be added in order for each molecule to change from ice
(a low energy state) to water (a higher energy state)
The temperature at which the solid and liquid phases of a given compound or mixture have the same vapour pressure. Melting
point range: the temperature at which a solid sample begins to melt and the temperature at which melting is complete. For water
the melting point range is from 100 to 0C
Membranes are phospholipid bilayers. A phase transition alters the membrane uidity because individual components or
regions of the membrane change between a liquid and a solid state. The transition temperature is dictated by the chain length
and saturation level of the individual fatty acids
Many important aspects of cryopreservation can be modelled mathematically (see e.g. Liu et al., 2000). Parameters to take
into account include: cell diameter, cryoprotectant molarity (initial and nal), ambient temperature. The cell's osmotically
inactive volume, the reection coefcient in KedemKatchalsky equations, cell membrane water (hydraulic) and
cryoprotectant permeability, molar volume of the cryoprotectant, and time
(e.g. http://www.me.umn.edu/divisions/tht/bhmt/tutorial/CPAload)
The rst event in ice crystal formation. At the molecular level, individual water molecules join. The change from a liquid to a
crystalline state is associated with a loss of energy (latent heat of crystalization). Nucleation is a stochastic process
Cell shrinkage and swelling which can lead to irreversible damage or lysis caused by excess water loss/gain (see text for full
description). See e.g. Pedro et al. (1997)
How much and how fast chemicals and water move across the cell membrane (can be denoted as P). Water permeability is
expressed as hydraulic conductivity
A plot (commonly of temperature against pressure or temperature against solute concentration) showing when the solution
undergoes a specic physical change e.g. glass transition, melting, de-vitrication, nucleation
A complex process, usually associated with warming, in which individual ice crystals within a solution change size. Small
crystals commonly shrink while large crystals become larger (see also crystallization and melting)
Also written as `s'(sigma), gives a measure of how well solutes cross a membrane
A solution which remains liquid, even though it is below the temperature at which it can support ice, is said to be in a
supercooled state. Ice crystallization will occur rapidly if a supercooled solution is seeded
Temperature at which de-vitrication (if any) occurs. The composition of the solution will inuence the temperature and
extent of this event
Temperature at which glass transition occurs; characterized by a sharp exotherm because of heat loss when metastable clusters
form. Below Tg, it is not merely a viscous liquid, but a solid in a stable thermodynamic state. The glass transition temperature
varies considerably and is dependent on the composition of the solution
Melting of ice crystals. The term thawing is therefore inappropriate for vitrication solutions which do not form ice
The rate at which the specimen is warmed. Specimens slow cooled to between 30 and 40C are usually warmed rapidly
(>200C/min) while those slow cooled to <60C are usually warmed slowly (<20C/min). Specimens containing ice do
not warm as uniformly or rapidly as ice-free samples as energy is taken up by the melting ice crystals
A plot of crystallization temperature against time. Generally gives a <-shaped plot which shows the temperature at
which crystallization is fastest. Inuenced by concentration and viscosity (see e.g. Sutton, 1992)
Melting temperature. The melting temperature varies considerably and is dependent on the composition of the solution
The temperature and pressure at which water can be present as a liquid, crystal or gas. Adding solutes alters the triple point.
Increased pressure can also lower the triple point
The pressure (e.g. in Pascals) that needs to be applied in order to maintain the amount of water vapour constant in the
presence of pure water or ice
Change in vapour pressure (in Pascals) caused by adding e.g. solutes to the water
J.M.Shaw and G.M.Jones
Table VII. Terminology for chemicals and solutions used for cryopreservation
Albumin
Antifreeze
Antifreeze protein
Buffer
Choline chloride
Colloidal pressure
CPA
Cryoprotectant,
non-permeating
Cryoprotectant
Permeating
Dimethylsulphoxide
(DMSO) (Me2SO)
EFS
Ethylene glycol (EG)
Formaldehyde
Glycerol
Glycol
Heat of solution
Hydrogen bonding
Molar (M, mol/l)
Molecular weight
(MW, mol. wt);
formula weight (FW)
Polyethylene glycol
Polymer
Propylene glycol
Propanediol (PROH)
592
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Composition
A protein ~68 000 Da. Used to reduce the `stickiness' of oocytes and embryos during handling, but it can also promote the
vitrication of cryoprotectant solutions (Rall and Wood, 1994; Shaw and Trounson, 1998)
Most cryoprotectants act as `antifreezes' in that they depress the ice nucleation temperature
Molecules which reduce the growth of ice crystals. A common source is from Arctic or Antarctic sh. Some act by having
a tertiary structure that produces one hydrophobic face which preferentially binds to crystalline ice, (i.e. the ice front) and a
hydrophilic face on the opposite side which preferentially binds liquid water. This `coating' reduces the likelihood of water
molecules in the liquid phase becoming crystalline by attaching to an existing ice surface. Antifreeze proteins have been added
to slow and rapid cooling procedures with highly variable success (Shaw et al., 1995a,b; Wang, 2000)
The solutions in which cells are cryopreserved include commonly available physiological buffers (e.g. phosphate-buffered
saline, HEPES, or MOPS buffered media), media (T6, Medicult, HTF), special media (e.g. choline chloride-based buffer
Stachecki et al., 1998, 2002), serum or even pure water. To this are added permeating and non-permeating cryoprotectants
and other additives. The choice of buffer can affect the outcome (Vasuthevan et al., 1992). Most buffers stabilize the pH.
In the semen cryopreservation literature the solutions (usually buffered and with cryoprotectant) that are added to the semen
before/after cryopreservation are referred to as `extenders'
A salt, mol. wt 139.6. It has been used instead of sodium chloride (NaCl) in slow cooling and vitrication procedures
(Stachecki et al., 1998, 2002) as this may help reduce the adverse effects of high NaCl concentrations on the hydration
shell of proteins and reduce the risk of raising cytosol NaCl levels
The osmotic pressure exerted by colloids (e.g. proteins). Colloid osmotic pressure (Donnan pressure) is the pressure
difference which has to be established between a colloidal system and its equilibrium liquid to prevent material transfer
between the two phases when they are separated by a membrane, permeable to all components of the system, except the
colloidal ones
The composition of a cryoprotectant solution can be expressed on the basis of weight (wt%, wt/wt, g etc.) or by volume
(ml, v/v) or a combination of the two (e.g. wt/v). A mole is a mass equal to the mol. wt in g; molar solutions have 1 mol/l of
solution; molal solutions have 1 mol/1000 g of solvent
In this context; cryoprotectant solution
Any of a range of compounds which do not readily cross the cell membrane but which none the less provide cells with
protection against cryoinjury. The ones most commonly used for cells include sugars, polymers and proteins
Any of a range of low mol. wt hydrophilic compounds which can hydrogen bond with water. The ones most commonly used
for cells because of their comparatively low toxicity are DMSO, ethylene glycol, glycerol and propylene glycol
Mol. wt 78.13. Antifreeze compound widely used for oocytes and embryos at a concentration of 1.5 mol/l (11% v/v) for slow
cooling, and >3.5 mol/l for rapid cooling. It is commonly used in combination with glycol-type compounds as the
combination interacts with water and biological materials in a slightly different way to glycols alone
E = ethylene glycol, F = Ficoll, S = sucrose. This combination has been found to be useful for mammalian embryos of a
wide range of species. First developed by Kasai et al. (1990)
Also called 1,2-ethanediol, mol. wt 62.07. Antifreeze compound widely used for oocytes and embryos. It is commonly used
in combination with DMSO. It has relatively low toxicity, and usually, because of its small size, penetrates membranes rapidly
A naturally occurring compound which at high concentrations (1 to 4%) is used as a xative. Has been found, at varying
concentrations, in batches of propanediol, thus it is advisable to screen or test propanediol before use
(see e.g. Mahadevan et al., 1998)
Glycerine; also called 1,2,3-propanetriol; mol. wt 92.1. Antifreeze compound widely used for sperm and blastocysts. It has
low toxicity, but is slow to cross membranes which can contribute to osmotic shock
A chemical classication of a group of chemicals which includes glycerol, propylene glycol (1,2-propanediol), ethylene glycol
(ethane diol), polyethylene glycol, butanediol (some of which may be methoxylated; Wowk et al., 1999). Most of these have
cryoprotectant properties but many are very toxic
The heat released when a cryoprotectant is dissolved, for example into water to make an aqueous solution
A chemical bond between a hydrogen atom on one molecule and a nitrogen, oxygen, sulphur or halogen. The hydrogen atom
is usually is covalently bonded in its molecule to oxygen, nitrogen or sulphur
A solution made up based on the mol. wt of a compound. For a compound such as sucrose (mol. wt 342) a 1 molar solution is
made by weighing out 342 g of sugar and adding liquid to make it up to a total volume of 1 l. (Not the same as molality or
mole fraction.)
Mol. wt is the sum of the weights of each atom in each molecule of this compound, calculated from the atoms which it contains
(number and atomic weight). Water for example contains one oxygen atom (16 in the periodic table) and two hydrogen atoms
(1 in the periodic table). Its mol. wt is calculated as 16 + 1 + 1 = 18. For salts it is more common to give the formula weight
(e.g. anhydrous MgCl has a FW of 95.21, and MgCl hexahydrate has a FW of 203.3)
A polymer of ethylene glycol available in a range of mol. wt. Inuences membrane permeability and fusability. Has proved
benecial in some glycerol (Rall, 1987), DMSO (O'Neil et al., 1997) and DMSO + PG (Rall and Fahy, 1985)-based vitrication
solutions for several species including the mouse and cow (Ohboshi et al., 1997)
Large mol. wt compounds such as Ficoll, Dextran, polyvinyl alcohol, polyvinylpyrrolidone, polyethylene glycol. Large mol. wt
proteins can have analogous effects. Can increase the viscosity and physical vitrication characteristics (Wowk and Fahy, 2002;
Shaw et al., 1997). Used in some solutions for slow cooling and vitrication (Rall, 1987; Kasai et al., 1990; Carroll et al., 1993;
Rall and Wood, 1994)
See `Propanediol' (commonly abbreviated as PG or PROH)
1,2-Propanediol, FW 76.1. Antifreeze compound very widely used for human embryos. It readily forms vitrifying solutions.
Commonly abbreviated as PG or PROH. A potential problem is that one of its natural breakdown products is formaldehyde
which has the potential to damage gametes (see e.g. Mahadevan et al., 1998)
Terminology for cryopreservation techniques
Table VII Continued
Proton bonding
PVA
PVP
Rafnose
Solute concentration
Solution control
Solution effect
Solvent
Sucrose
Sugars
Trehalose
v/v (vol/vol)
Vitrication solution
VS1
VS3/VS3a
wt/wt (wt%)
dictated by the composition of the solution but also other factors,
the most important being the cooling rate and the warming rate.
Thus, a solution which vitries in one protocol may form ice
crystals when used under other conditions; these crystals may
develop only during warming (de-vitrication) or during both
cooling and warming. Vitrication occurs most readily at high
cooling and warming rates. Because of the interactions between
ice formation and the cooling and warming conditions, it is
possible to have both vitrifying and ice-forming regions within the
same solution. A solution with a set composition but cooled and
warmed at different rates, or solutions with differing compositions
but cooled at a set cooling and warming rate, can as a result form
anything from a wholly transparent (vitried, ice-free) and glasslike state, through having some microscopic ice crystals (usually
seen as a `haze'), to being fully opaque in the presence of many
macroscopic ice crystals. However, even transparent solutions
may contain countless ice nuclei and ice crystals, because the ice
crystals only become detectable optically once they become larger
than the wavelength of light. Sensitive equipment such as
differential scanning calorimeters can measure the release and
uptake of energy associated with ice crystal formation and melting,
but even with this equipment it can be difcult to differentiate
between a fully vitried ice-free state and a nearly vitried state in
which only some ice nuclei or small ice crystals are present. If a
specimen is not uniformly cooled or the solution is not well mixed
before cooling, then it may form both vitried and non-vitried
regions. Thus vitrifying, partially vitrifying, de-vitrifying and iceforming conditions, although they are clearly different physically,
are part of a continuum rather than each having a clear boundary.
Unlike slow cooling solutions that expand during cooling because
ice occupies a larger volume than water, vitrication solutions
appear to remain the same volume or shrink. In the absence of ice
there is also no redistribution of salts, solutes or cells. These
attributes should in theory make solutions that remain vitreous
593
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Total solute
concentration
Toxicity
See `Hydrogen bonding'
Polyvinyl alcohol. A polymer used to seal straws, but also to substitute for proteins. Recently shown to be highly effective
in promoting vitrication (reducing ice nucleation) (Wowk and Fahy, 2002)
Polyvinylpyrrolidone. A polymer used to seal straws, but also in media, buffers and cryoprotectants to substitute for proteins.
Can be used to promote vitrication or to replace some penetrating cryoprotectant (Shaw et al., 1997). Some batches are toxic to
embryos even after dialysis against water
A sugar, mol. wt 594.5. Used to promote dehydration before, during and/or after cryopreservation. Normally does not cross the
cell membrane (non-permeating/impermeant) and of low toxicity to oocytes and embryos (Kuleshova et al., 1999b;
dela Pena et al., 2002). Used for mouse sperm cryopreservation
How much solute (salts, sugars, cryoprotectants, and other additives) a solution contains. Vitrication solutions commonly
have a high solute concentration (>40% wt/vol) (Fahy et al., 1987)
Oocytes, embryos or cells that have been exposed to the solutions used in a cryopreservation procedure but have not actually
been cryopreserved. Gives an indication of the toxicity of the solutions
Damage caused in a slow cooling procedure by the non-frozen fraction (Mazur et al., 1972)
In cryopreservation, water is usually the solvent in which other chemicals are dissolved
A sugar, mol. wt 342. Used to promote dehydration before, during and/or after cryopreservation. Normally membrane
impermeant and of low toxicity to oocytes and embryos
Monosaccharides (fructose, galactose, glucose); disaccharides (maltose, sucrose, trehalose, lactose); polysaccharides
(e.g. rafnose) (Abas Mazni et al., 1989; McWilliams et al., 1995; Kuleshova et al., 1999b)
How much solute (salts, sugars, cryoprotectants, and other additives) a solution contains. Vitrication solutions commonly have
a high solute concentration (>40% wt/vol)
General term, referring to any reduction in viability or developmental potential in cryopreserved and solution control
embryos/cells, but not non-treated controls. Degree of toxicity assessed under any particular set of conditions will depend on
the particular criteria used in the assessment. Strongly inuenced by cryoprotectant type and concentration, temperature and
time of exposure, cell type and species. Unrelated to chill sensitivity and osmotic shock. Can affect both cytoplasm and the
nucleus (see e.g. Kono et al., 1988)
A sugar, mol. wt 378.3. Used to promote dehydration before, during and/or after cryopreservation. Normally membrane
impermeant and of low toxicity to oocytes and embryos (Kuleshova et al., 1999b; Ergolu et al., 2002)
The composition of a cryoprotectant solution expressed by volume (volume of the additive/volume of the total solution).
Useful when compounds differ in density.
Solutions which, on cooling, can form a solid, non-crystalline phase. Whether a solution vitries is governed by its
composition and the cooling and warming rate. Solutions may vitrify on cooling, but crystallize (de-vitrify) during warming.
Procedures which allow the solution to retain its glass-like state throughout cooling and warming are `true' vitrication
procedures. Procedures which allow any crystals (ice) to form in the solution at any stage during cooling and warming are
not vitrication procedures but can be classied as rapid cooling procedures
The vitrication solution used by Rall and Fahy (1995), the 100% VS1 (~ 13 molal) contained 10% PROH (~1.5 mol/l),
20% DMSO (~3 mol/l) + 15% acetamide + 6% polyethyleneglycol
VS3 = 48% glycerol (6.5 mol/l) + 6% polyethylene glycol; VS3a = 48% glycerol (6.5 mol/l) + 6% bovine serum albumin
The composition of a cryoprotectant solution expressed by weight (weight of the additive/weight of the total solution).
Useful when compounds differ in density. The composition of a cryoprotectant solution can be expressed as its percentage
of the weight of the solution. Thus when 100 g of the nal solution contains 10 g of a chemical, this chemical constitutes
10 wt%. [Could also be written as 10% (wt/wt).]
J.M.Shaw and G.M.Jones
Table VIII. Terminology for processing steps/procedures
Break point
Cooling rate
Cryoinjury
Denaturation
Non-equilibrium
cooling
Plug
Ramp
Rehydration
Rinse
Seeding
Soaking time
Ultrarapid
Length of time that the specimen is exposed to a given solution (see e.g. Shaw et al., 1992)
A term used for cryopreservation protocols in which the water content of a cell remains in osmotic balance with the surrounding solution
during cooling in the presence of ice crystals. Exemplied by slow cooling procedures
Length of time that the specimen is exposed to a given solution (see e.g. Shaw et al., 1992)
A step in a slow cooling programme. A period of time over which the temperature is held constant. (Also called soaking time.)
Loading and sealing a straw so that it contains both the dilution solution and the cryoprotectant solution containing the cells. These
are separated by an air bubble. After warming the straw is shaken to move the air bubble, allowing the two solutions to mix
Latent heat of crystallization is the heat (energy) released by water molecules when they change from a liquid to a solid (ice)
In this context this refers to the size and number of air bubbles/columns of solution within a straw. Semen is commonly loaded into a
straw with either no, or only a very small, airgap before being sealed. When embryos or oocytes are cryopreserved by slow cooling,
a small amount of air (e.g. 0.05 ml) is usually placed both in front of and behind the cryoprotectant solution containing the cells. This
prevents the cells from becoming lodged in the plug or sealed ends of the straw. A further amount of cryoprotectant or dilution solution
is also commonly placed beyond the air. If more than one solution is placed in a straw, it is important that the bubble or interface
separating them is not disturbed during the cooling and warming steps
A term used for cryopreservation protocols in which cellular dehydration and cryoprotectant penetration are enforced by solutions with
a high osmolarity before cooling commences, and change very little during the cooling step. Exemplied by all rapid-cooling procedures
There are usually two small pieces of material on either side of a water-absorbent polymer (e.g. PVP) at the end of cryopreservation
straws. The water-absorbent material swells and forms an impervious barrier when an aqueous solution is drawn into it. In slow cooling,
the straw is usually loaded in such a way that the plug becomes sealed by the rst aliquot or column of solution. The straw may in
addition be heat, or impulse sealed. A bubble of air between the specimen and the solution containing cells is thought to reduce the
likelihood of cellular damage by heat being transferred into the liquid
A step in a slow cooling programme, which starts at one temperature and ends at a different temperature. The cooling or warming rate
is constant between the two points e.g. 0.3C/min
Cells are normally dehydrated by cryopreservation and need to be re-hydrated before use. Cells are usually rehydrated and at the
same time have the cryoprotectant removed, by placing them in one or more solutions with successively lower osmolarity ending
with the return to a physiological solution (buffer or media) or transfer to a recipient
Straws are commonly rinsed with the cryoprotectant solution before being loaded with the cells to remove debris and possible
contaminants. We have noticed that conventional cryopreservation straws can change the pH of the rinse solution
Manual or automated initiation of ice formation. Usually as part of a slow cooling procedure. Is initiated for example by touching
the edge of the solution with a cold implement (e.g. forceps at 196C). The cryoprotectant is seeded at a high subzero temperature
(between 4C and 9C) to initiate dehydration of the specimen
Alternative term to `holding time' (see above)
A term which has (confusingly) been applied to a very wide range of cooling procedures and cooling rates. It is therefore best
considered as a `catch' phrase
throughout cooling and warming less damaging to cells than ones
that form ice. Whether this is true or not remains unclear.
In the literature the term `vitrication' has on occasion been
applied to both wholly ice-free and ice-forming conditions. The
terms cooling, freezing, warming, melting and thawing have been
used relatively interchangeably whether ice is present or not. The
term `freezing' specically relates to ice crystal formation whereas
`thawing' specically refers to the melting of ice crystals. These
terms are therefore only relevant to solutions with ice. Under
conditions in which no ice forms, the terms `cooling' and
`warming' are more correct. Several papers also describe the use
of `ultrarapid' cooling procedures, but this may be thought of as a
catchphrase and not a cryopreservation procedure since this term
has been loosely and inconsistently applied to a range of protocols.
As it can be difcult to determine from a description of a procedure
594
whether the specimen actually vitries and remains vitried, this
review includes rapid cooling (e.g. >20C/min) procedures
irrespective of whether these would result in vitrifying, devitrifying or non-vitrifying conditions.
Procedures/processing steps in cryopreservation for
assisted reproductive technology
Most human IVF units use slow cooling rather than rapid cooling
procedures to store embryos. In the mouse there has, over the past
10 years, been a shift away from slow cooling procedures to
ethylene glycol (Kasai et al., 1990; Zhu et al., 1993; Kuleshova
et al., 1999b), glycerol (Rall and Wood, 1994; Dinnyes et al.,
1995; Zhu et al., 1996) or DMSO (Shaw et al., 1991a,b)-based
rapid cooling and vitrication procedures. For many mouse strains
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Equilibration
temperature
Equilibration
time
Equilibrium
cooling
Exposure time
Holding time
In-straw
dilution
Latent heat
Loading
pattern
The temperature at which a slow cooling ramp is ended (most commonly between 30 and 80C), and a more rapid cooling step (usually
3300C/min), usually to the nal storage temperature, starts
The known or estimated change in temperature over a set time. In slow cooling, the rate of cooling most commonly is 0.3 to 1C/min from
6 to ~35C, and then >10C/min to 196C. With rapid protocols, cooling rates of between 200 and 20 000C/min are most common
Damage to cell components caused by cryopreservation. Virtually any component can suffer cryoinjury
A change in the 3-dimensional conformation of a protein. Cryoprotectants such as DMSO can, if added too quickly to a solution
containing a protein, make it milky or opaque. It is likely that the protein becomes denatured by the DMSO. It is recommended that
the DMSO be added stepwise
The temperature of the solution in which cells are placed/equilibrated (see e.g. Shaw et al., 1992; Mukaida et al., 1998)
Terminology for cryopreservation techniques
Table IX. Terms describing processes associated with cells (other than those already covered in previous tables)
Apoptosis
Chill injury
Dilution effect
Extender
Free radical damage
Lipid peroxidation
Metabolism
Oocyte
Phase transition
Reactive oxygen species
(ROS)
Saturation
Spindle
disassembly/reassembly
Storage time
Thermal shock
Zona hardening
the in-vitro and in-vivo development of embryos cryopreserved by
these rapid cooling procedures is the same (statistically) as control
non-frozen embryos and signicantly better than for slow cooled
embryos. Highly efcient protocols have also been developed for
other species including the cow (Vajta et al., 1998, 2000), sheep
(Naitana et al., 1997; Martinez and Matkovic, 1998; Baril et al.,
2001; Zhu et al., 2001; Al-aghbari and Menio, 2002;
Papadopoulos et al., 2002; Isachenko et al., 2003) and rabbit
(Lopez-Bejar and Lopez-Gatius, 2002). The animal research
indicates that there is no single `correct' or single optimum
cryopreservation protocol because the outcome is inuenced by
many factors (see e.g. Table V for 8-cell mouse embryos). Thus
there is now a variety of different solutions, equilibration and
dilution strategies (volumes, times and temperatures), and cooling
and warming procedures that can give excellent results (for the
mouse >90% blastocysts, >60% fetuses). However, for each
effective combination there are many other combinations of
solutions, times and temperatures that give poor or erratic
outcomes (Table V). The slow cooling procedures used for
human embryos do generally give consistent results, but the
embryo survival rates (usually <80%) are not ideal. Early rapid
cooling trials for human oocytes and embryos (e.g. Trounson,
1986) obtained some promising results, but there was also
considerable concern as several early papers indicated that rapid
cooling could harm oocytes and embryos. The tangible benets of
rapid protocols (cost savings and convenience) have, however,
ensured that some groups have persevered in trying to optimize
rapid protocols for human oocytes and embryos. Several very
signicant advances have recently been made in rapid cooling of
human embryos, and as a result we may now be at the point where
595
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Parthenogenic activation
Cell suicide. An active process in which the cell undergoes an orderly sequence of events leading to its disintegration.
Cryopreservation can trigger apoptosis in cells
Damage to cells that occurs at temperatures >0C (see e.g. Pollard and Leibo, 1994; Martino et al., 1996a)
The change in sperm motility when semen is diluted (this is often a sharp fall)
Term used widely in the sperm cryopreservation literature. A solution added to semen. It can have a very simple composition
but most contain cryoprotectant, and additives such as oocyte yolk or milk powder
Most cellular components can be damaged by free radicals (unpaired oxygen atoms, free hydroxyl groups and hydrogen peroxide)
as these compounds are highly reactive and cause damage by oxidation or reduction. See `ROS' below
Membranes and other lipid-containing cellular components are degraded by free radicals (see `ROS'). Removal of lipid stores
can greatly improve survival following cryopreservation (Ushijima et al., 1999; Nagashima et al., Dobrinsky 2001)
The biochemical reactions occurring in a cell. Placing cells at temperatures below that of the normal body temperature perturbs
metabolic functions. The cell's capacity to fully recover after rewarming to body temperature may be affected by these changes
(see e.g. Hammerstedt and Andrews, 1997; Parks, 1997)
Oocytes of all species so far tested are more difcult to cryopreserve than embryos of the same species (see e.g. Nakagata, 1989;
Van Blerkom, 1989; Carroll et al., 1993; Bernard and Fuller, 1996; Martino et al., 1996; O'Neil et al., 1997, 1998;
Paynter et al., 1997 a,b; 1999a,b; Stachecki et al., 1998, 2002; Trad et al., 1999; Kuleshova et al., 1999a; Papis et al., 2000;
Vajta, 2000; Chen et al., 2001, 2003; Hochi et al., 2001; Lane and Gardner, 2001; Ledda et al., 2001; Matsumoto et al., 2001;
Arav et al., 2002; Songsasen et al., 2002; Begin, 2003)
Development of an unfertilized oocyte. This can be triggered by the chemicals and procedures associated with cryopreservation.
Parthenogenically activated oocytes can be haploid or diploid and develop to somite stage fetuses following transfer. Care is
therefore needed to not confuse them with normally fertilized oocytes (two-pronuclear and with two polar bodies)
See denitions in Table VI for membrane phase transition and phase transition diagram, `Saturation' (below) and `Free radical
damage' (above)
Oxygen molecules are normally paired. If the two molecules are separated they form highly reactive charged ions that can
readily damage cellular components by oxidation. Not all cell types and components of the cell are equally vulnerable to ROS
injury (see `Metabolism', `Saturation'). Free radical scavengers (including cryoprotectants, sugars, vitamins such as vitamin C
and E, and enzymes such as superoxide dismutase) may help protect against damage by ROS
Hydrocarbon chains in fatty acids may have a variable number of double bonds. These chains are said to be saturated if there
are no double bonds, monounsaturated if there is a single double bond and polyunsaturated if there are multiple double bonds.
The number of double bonds affects two main properties of the fatty acid. Double bonds represent potential sites for
biochemical reactions, particularly with ROS or other tree radicals. They also limit the exing of the carbon skeleton of the
fatty acid, which affects the melting temperaturethe greater the number of double bonds the greater the inexibility, and the
higher the temperature at which melting occurs (see e.g. Arav, 1996, 2000; Zeron et al., 2001, 2002)
In metaphase II oocytes the chromosomes are held in place by very ne bres (the spindle). The spindle can break down in
response to cooling and cryoprotectants. On warming, the spindle will reassemble but may not do so correctly. This can lead
to the loss of chromosomes when the cell extrudes the polar body (see e.g. Chen et al., 2001, 2003)
At 196C it should be possible to store cells for considerable lengths of time without spoilage (decades, possibly centuries).
For human gametes and zygotes, legal constraints usually impose a shorter storage time than this
Damage caused by reduced temperature. Membrane shrinkage and rearrangement of membrane components are important,
particularly when this increases membrane permeability
Changes that increase crosslinking of the glycoproteins of the zona pellucida, making it more impenetrable. Important for
oocytes as it can reduce sperm penetration. Caused for example by cryoprotectants, sugars or cooling (see e.g.
Matson et al., 1997)
J.M.Shaw and G.M.Jones
596
osmotic shock and toxicity when the cells are cooled to low
subzero temperatures.
Slow cooling
Slow cooling and rapid cooling procedures tend to differ in
cryoprotectant concentration and cooling rate, and it is possible
(but not neccessarily optimal) to use either high and low
cryoprotectant concentrations and high and low cooling rates
with either procedure. Thus the principal difference between the
two lies in when and how the specimen becomes loaded with
antifreeze compounds and dehydrated. Equilibrium, or slow
cooling procedures usually use solutions containing 12 mol/l
concentrations of cryoprotectant and reduce the likelihood of
intracellular ice formation by initiating extracellular ice crystal
formation (seeding) at a high subzero temperature (e.g. 6C). The
rate of cooling is then used to govern how quickly these
extracellular ice crystals grow. The best results are thought to
occur at the fastest cooling rate at which a balance (equilibrium) is
maintained between water loss (by diffusion) from the cell and the
incorporation of this water into the extracellular ice crystals
surrounding the cell (for a fuller description of the mathematics
and principles of equilibrium cooling procedures see Mazur,
1990). The cooling rate used for oocytes and embryos (between
0.3 and 1C/min) reects their size, permeability and diffusion
characteristics and is signicantly slower than for somatic cells,
erythrocytes or sperm (1 to >100C/min). Even though equilibrium
protocols use relatively low concentrations of cryoprotectant, it is
still necessary to control damage from cryoprotectant toxicity and
osmotic shock by selecting the most appropriate cryoprotectants as
well as the time, temperature, and number of addition and removal
steps. A small number of equilibrium slow cooling procedures are
very widely used for human preimplantation embryos at all stages
of development. These procedures generally result in 70% survival
of embryos and the pregnancy rate and development to term
following transfer are similar to those of non-frozen embryos. It
would be highly desirable to have a slow cooling protocol that is
100% efcient for human oocytes and embryos, but it is not clear
whether this will ever be possible. There are groups who have
consistently obtained excellent slow cooling outcomes (>90%
continued development) for mouse embryos. Mouse oocytes and
embryos are more tolerant of cryopreservation than most other
species because of their smaller size, low intracellular ice
nucleation temperature and low chill sensitivity. Oocytes and
early embryos of domestic species such as the pig and cow are by
contrast much more chill sensitive than those of the human.
Despite the lack of animal models, there have recently been
changes to the protocols for both human oocytes (Fabbri et al.,
2001) and blastocysts (Gardner et al., 2003) which improve the
post-thaw outcomes. Further advances are therefore possible. Thus
the main disadvantages of slow cooling are that the equipment can
be expensive to purchase and maintain and that each `run'
(equilibration and cooling) takes >1 h.
Rapid cooling
Rapid cooling procedures began to be developed in the 1970s (e.g.
Wilmut, 1972; Elliott and Whelan, 1977; Leibo et al., 1978).
These are non-equilibrium procedures in that the water loss
(dehydration) and cryoprotectant gain by the cell does not reach an
equilibrium with the surrounding solution before cooling com-
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
a transition from slow to rapid cooling procedures will start to take
place also for the human.
Rapid cooling protocols are very inexpensive and easy to
establish, but it is important that the procedure is fully optimized
for the material which is to be preserved. The cheapest and
simplest rapid cooling procedures to set up are those which allow
the materials to be cooled in the vapour phase of liquid nitrogen or
in liquid nitrogen itself. Specimens placed deep within a stable
vapour phase cool at rates of between 10C/min for large
specimens (e.g. goblets of straws containing sperm) and >300C/
min (individual straws, or small samples). Specimens placed
directly into liquid nitrogen cool at rates of 2000C/min (e.g. for
straws) to >20 000C/min (e.g. for small grids and pulled straws).
Which cooling rate and which cryopreservation solution is best for
a given cell type is dependent on several parameters.
Many studies have investigated the effect of different cooling
and warming rates. Some early studies compared cooling rates of
between 5 and 2500C/min and warming rates of between 10 and
2000C/min. Moderate or high cooling and warming rates gave
more consistent and better results than the slowest cooling and
warming rates, but the magnitude of the effect differed markedly
depending on the composition and concentration of the solution.
Two glycerol-based vitrication solutions, VS3 and VS3a, gave
high (>80%) or very high (>90%) survival rates of mouse embryos
over most of the tested cooling and warming rates, and even the
combination of the very slowest cooling and warming rates did not
reduce survival to <75%. By contrast, vitrication solutions VS1
and VS2, and dilutions of VS1, were strongly inuenced by the
cooling and warming rate, in that the slowest cooling and warming
rates could kill all embryos. Solutions which formed ice during
cooling usually give suboptimal results, but in other respects the
results did not correlate closely with physical changes. Many
recent studies have used much higher (e.g. >20 000C/min)
cooling and warming rates than in the earlier studies. The very
high cooling rates and warming rates have been achieved by (i)
reducing the cryoprotectant volume, (ii) modifying the container,
(iii) cooling in liquid nitrogen slush (210C), or (iv) cooling on
metal blocks. Increasing the cooling and warming rates is very
benecial for the lipid-rich oocytes and embryos of domestic
species (cow, pig), as it helps to overcome chill sensitivity (Pollard
and Leibo, 1994; Martino et al., 1996a,b). The benet of using
faster rates must, however, be balanced against the increased
likelihood of stress fractures (most materials shrink on cooling)
(Kasai et al., 1996; Poledna and Berger, 1996; Shi et al., 1998; Liu
et al., 2000; Arav et al., 2002). This is best exemplied by a study
in which repeated cooling and warming of mouse embryos at
intermediate cooling and warming rates gave signicantly better
survival than at high rates. Reducing the volume of cryoprotectant
reduces the likelihood of stress fractures (by reducing the
temperature difference across the specimen), but this needs to be
balanced against the increased likelihood of osmotic shock in the
rst dilution solution.
Slow cooling (equilibrium) and rapid cooling (non-equilibrium)
procedures all comprise four basic steps: exposing the cell/tissue
to a cryoprotectant, cooling to the storage temperature (usually
196C, the temperature of liquid nitrogen), warming and
cryoprotectant removal. These steps aim to reduce chill and
cryoinjury, e.g. intracellular ice formation, dehydration injury,
Terminology for cryopreservation techniques
(toxic) cryoprotectant needs to be added to the solution. The
sugars, in addition to contributing to glass formation, also aid
dehydration by exerting an osmotic effect on the cells, and can
help to stabilize membranes (Anchordoguy et al., 1987; Crowe
et al., 1988). Together they can greatly reduce the amount of toxic
penetrating cryoprotectant needed to protect the cell from
cryoinjury and the length of time needed to dehydrate the cells
before cooling and restore the cell to an iso-osmotic solution after
warming. Even very small amounts of additive can have a
dramatic effect on the vitrication properties of a solution (Wowk
and Fahy, 2002). Rall (1987) and Rall and Wood (1994) showed
that adding either polyethylene glycol or BSA promoted the
vitrication of their glycerol-based solution. Adding BSA could
reduce the likelihood of ice formation in glycerol based solutions
cooled at 5 or 200C/min from 100% to 0.
Processing steps with the potential to compromise cell viability
Both slow and rapid cooling procedures can kill or damage cells
(oocytes and embryos). Unfortunately even effective procedures
can be compromised by inexperienced users or inappropriate
handling. The most common errors which we see among
inexperienced users are failure to pre-plan, failure to adhere to
the specied temperatures, times and volumes and the failure to
load, cool, warm and dilute as specied. They may also
inadvertently compromise the specimens by causing partial
warming when they lift, hold, move or inspect cold specimens
(or the canes/goblets/or canisters that they are in). In the steps
before cooling it is important that the pre-equilibration and nal
cryoprotectant solutions are made up as directed, used in the
specied quantities and not diluted. Thus the cells (oocytes/
embryos) should be moved with a minimal volume of attendant
solution (e.g. <1 ml). For oocytes and embryos rapidly cooled in
large volumes, e.g. >10 ml, it appears that the best outcomes are
obtained if they are always handled gently (i.e. slowly and gently
expelled into the diluent and from there gently moved on in a
generous amount e.g. 20 ml of uid) as the cells are very fragile and
susceptible to osmotic shock. All steps should be performed
rapidly and smoothly, in particular the insertion and removal of the
specimen into/out of vapour or liquid nitrogen. Specimens cooled
and warmed in larger volumes (>10 ml) appear to benet from
steps aimed at minimizing fractures, e.g. by holding the specimen
very deep in the vapour phase for several minutes before and after
nitrogen storage (Kasai et al., 1996).
Equipment/materials
To perform cryopreservation requires materials, equipment and
the knowledge how to use them. In addition to standard
embryology equipment (microscopes, incubators etc.), the only
additional materials needed by a functional IVF laboratory to
perform rapid, ultrarapid or vitrication procedures are a large
vessel in which to store samples (liquid nitrogen or vapour
storage), small containers for the individual oocytes or embryos, a
waterproof pen (or equivalent for labelling), safety equipment
(gloves, goggles and long forceps) and a container (Dewar) for
bench-top work (see Appendix 1).
597
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
mences. Early studies investigated a very wide range of cooling
rates, warming rates, containers, cryoprotectants and cryoprotectant additives and formed the basis for subsequent work (Rall et al.,
1980, 1983; Takeda et al., 1984; Krag et al., 1985; Scheffen et al.,
1986; Williams and Johnson, 1986). Rall and Fahy (1985) were the
rst to apply vitrifying conditions to embryos. This approach,
which was based on the pioneering work of Lujet, reasoned that
because ice formation is a major contributor to freezethaw injury,
cryopreservation conditions which prevented any ice from forming
should be benecial. The concept is attractive, which may explain
why vitrication appears to receive more attention/mention than
other rapid cooling procedures.
Whether procedures in which the solutions remain vitreous
throughout the cooling and warming procedure are better or worse
than those in which solutions vitrify during cooling but de-vitrify
during warming is not wholly clear. In the mouse, conditions in
which ice forms during both the cooling and warming step of a
rapid cooling procedure are usually damaging (Tables IV, V),
while those rapid cooling procedures in which the specimen
remains vitried or only forms ice (de-vitrify) during warming can
both give either excellent or poor results (Tables IV, V). In this
case the best outcome (developing fetuses) was achieved when devitrifying rather than vitrifying conditions were used. It is possible
that the more concentrated solution gave this result by being more
toxic or by causing greater loss of bound water. Hochi et al. (2001)
showed that with a single vitrifying solution the fastest cooling and
warming rates did not necessarily give the best results.
Vitrication and a rapid warming rate is thus not a guarantee of
success. Rather it is now evident that embryo/oocyte survival is
dependent on optimization of the whole cryopreservation procedure (see e.g. Kasai et al., 1980, 1990, 1996; Rall and Fahy, 1985;
Rall, 1987; Szell and Shelton, 1987; Shaw et al., 1991a,b; Ali and
Shelton, 1993a,b; Mukaida et al., 1998; Vajta et al., 1998; Vajta,
2000; Arav et al., 2002). Adding the cryoprotectant in several
steps, starting with a low concentration, can be benecial as this
ensures that the cells gain enough cryoprotectant to protect them
from dehydration injury without increasing toxic effects. Early
vitrication procedures aimed for relatively full equilibration with
the nal cryoprotectant solution, but it now appears that a short
exposure time to the nal (cryoprotectant) solution is better
because less cryoprotectant enters the cell, thereby reducing both
toxicity and cryoprotectant removal problems. Providing that the
osmolarity of the nal solution is high, and it contains some
cryoprotectant, even a very short exposure time (<1 min) appears
to remove enough water from a cell (by osmosis) to minimize
intracellular ice formation. Although vitrication and other rapid
cooling procedures can be made without any polymers, proteins or
sugars, it does appear that these compounds can be benecial in
both vitrifying and de-vitrifying procedures. Polymers such as
Ficoll, Dextran, polyvinylpyrrolidone, hylauronic acid, polyvinyl
alcohol, polyethylene glycol, or proteins such as bovine serum
albumin (BSA) or sugars such as sucrose or trehalose, raise the
total solute concentration of a solution without increasing the
toxicity of the solution (providing that the additive is not itself
embryotoxic). The large polymers do not penetrate a normal cell
membrane but may markedly increase the viscosity of the solution
and can contribute to, or modify, the formation of a stable glasslike state (vitrication) (see e.g. Sutton et al., 1992; Shaw et al.,
1997; Kuleshova et al., 2001) with the result that less penetrating
J.M.Shaw and G.M.Jones
Storage vessels
It is possible to drip or deposit a drop of cryoprotectant solution
containing cells, oocytes or embryos directly, as a drop, into liquid
nitrogen (Landa and Tepla, 1990; Papis et al., 2000), but this
technique is rarely used, and instead most workers place their
specimens within a specialized holder or container. A number of
different containers can be used for rapid cooling and vitrication
of oocytes and embryos. These include: (i) whole plastic straws,
e.g. 0.25 or 0.5 ml (e.g. Rall and Fahy, 1985; Kasai et al., 1990;
Shaw et al., 1991a,b; Vanderzwalmen et al., 2002); (ii) pulled
plastic straws (Vajta et al., 1998; Lopez-Bejar and Lopez-Gatius,
2002; Chen et al., 2003); these are made by stretching conventional straws to give them a smaller diameter and thinner walls
(pulled straws); (iii) plastic straws which have been cut in half
(Benia et al., 2000; Vandervorst et al., 2001); (iv) nylon loops
(Lane et al., 1999; Yeoman et al., 2001; Begin et al., 2003;
Mukaida et al., 2003); (v) copper or gold electron microscope
grids (Martino et al., 1996b; Chen et al., 2001); and (vi) pipette
tips (Liebermann et al., 2002; Hochi et al., 2003), nylon mesh
(Matsumoto et al., 2001), glass capillaries (Kong et al., 2000;
Hochi et al., 2001), pieces of aluminium foil (J.M.Shaw,
unpublished data), `cryotop' minimum volume lament (Hochi
et al., 2003), solid metal surfaces (Begin et al., 2003), vials
(Nakagata, 1989) and tiny drops placed on another surface (e.g.
Misumi et al., 2003) (Table X). Many of these have only been used
for rapid cooling of oocytes or embryos of animals. Each type has
specic characteristics which inuence the total size and
cryoprotectant volume, cooling rate warming rate, likelihood of
loosing the specimen, risk of fracture, risk of explosion, risk of
contamination, ease of use, ease of labelling, ease of storing, ease
of sterilization, loading cost and availability. Straws are the most
widely used container type. They come in many varieties,
including different sizes, compositions (plastics, polymers) and
colours. The risk of a straw shattering, breaking or leaking is
inuenced by its composition and how it is sealed. Straws for
agricultural use (semen preservation) are generally low cost and
supplied in large packets. Straws for human use are generally of
higher quality and supplied pre-sterilized in small packets. All can
be used for slow cooling and rapid/direct cooling protocols, but
some types of plastic become unsuitable for rapid cooling
procedures if they are sterilized by irradiation (Shaw et al., 1988).
The function of a storage vessel is to hold specimens (cryopreserved by either rapid or slow protocols) at a low subzero
temperature. Most groups use liquid nitrogen storage vessels
(variously referred to as tanks, Dewars or asks) which keep
specimens at 196C, but very low temperature electrical freezers
(130 or 150C), or nitrogen vapour tanks (ideally as close to
196C as possible) can also be used. Within each of these three
categories there are many types of vessel, differing in their overall
dimensions, storage volume (e.g. 20500 l), storage capacity
(number of samples), type of storage (commonly vials, straws,
canes, goblets or boxes), and running costs. Running costs are
governed by the insulation, neck diameter, use, and the age of the
tank (nitrogen consumption rates in a new tank lie between 0.1 and
1 l/day). The vessel conguration should be matched to the user's
requirements. Most IVF units cryopreserve oocytes or embryos
individually, or in small groups, in some form of small container
(e.g. straws, grids, loops, or vials) which in turn is placed on a cane
or in a goblet (the goblet may then be afxed to a cane). Canes or
goblets are then most commonly inserted into a canister in a large
storage vessel. An alternative is to place specimens in boxes which
are held in a rack. Boxes are more widely used for vials and cell
lines than for oocytes, embryos or sperm. Most equipment and
accessories (e.g. racks for boxes, canisters for canes or goblets) for
liquid nitrogen storage are available from general scientic
suppliers (e.g. Sigma Aldrich), and other outlets e.g. BOC, CIG,
and manufacturers (e.g. MVE, Taylor Wharton). Electrical
freezers and vapour storage units are also available from scientic
suppliers.
The main advantage of liquid nitrogen storage over vapour
tanks or others is that the specimens are at a very low and constant
temperature. The main concerns, over and above the safety issues
associated with any procedures using liquid nitrogen, are that: (i)
the specimens will warm up if the level of nitrogen is not
monitored and maintained (Siegel-Itzkovich, 2003); (ii) older
vessels (>5 years) will use increasing amounts of liquid nitrogen
and may suffer outright failure (loss of all nitrogen within a few
hours); (iii) more than one specimen is housed in each canister,
thus they may be partially warmed each time a specimen in the
same canister is accessed; (iv) with time, labelling may smudge or
become indistinct; (v) specimens and contaminants can escape into
the liquid nitrogen; (vi) liquid nitrogen can enter the specimen
container, which may cause the container to explode if it is
removed rapidly from the liquid nitrogen, and the possibility of
contaminants entering with the nitrogen. Movement of pathogens
between samples (cross-contamination) has resulted in clinical
disease once, when hepatitis C virus from a broken blood bag was
transmitted by the liquid nitrogen to other blood bags (Tedder
et al., 1995; Hawkins et al., 1996). Experimental cross-contamination was not seen for vials (Kyuwa et al., 2003) but could occur
with open straws (Benia et al., 2000; Bielanski et al., 2000;
Letur-Konirsch et al., 2003). There are no reports of transmission
of pathogens between specimens by Units performing assisted
reproduction, but steps to reduce the possibility of cross-contamination are recommended (Tomlinson and Sakkas, 2000;
Kuleshova and Shaw, 2000). Storage in freezers or in nitrogen
vapour eliminates the possibility of cross-contamination and
specimens oating into the tank, but there is a higher risk of
Nitrogen slush
Most direct cooling protocols can be performed without any large
or costly items of equipment using normal liquid nitrogen or
nitrogen vapour. However, there are claims that using liquid
nitrogen slush, which gives a faster cooling rate, can give better
outcomes than ordinary liquid nitrogen. We do not know if this
applies to the human oocytes or embryos. Liquid nitrogen slush is
made by applying a vacuum to liquid nitrogen. A vacuum lowers
the boiling point of the liquid nitrogen and raises the evaporation
rate. As the nitrogen boils off, evaporative cooling makes the
temperature fall. As the temperature of the remaining liquid
nitrogen falls, the viscosity of the remaining liquid appears
greater, and it boils off less readily around specimens immersed
into it. It is possible to buy equipment designed to make liquid
nitrogen slush.
598
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Containers for oocytes and embryos
Terminology for cryopreservation techniques
accidental warming/thawing of specimens during activities such as
checking for specimen ID, because the storage temperature is
closer to that at which ice crystals can grow. To prevent warming
at times of power failure, electric freezers may come with a liquid
nitrogen back-up.
Conclusions
Acknowledgements
We thank Dr G.Shaw, Prof. D.MacFarlane and Prof. D.Galloway for
helpful input. This review was prepared while in receipt of funding
from Monash IVF, and the NH&MRC (J.M.S.).
References
AbasMazni, O., Takahashi, Y., Valdez, C.A., Hishinuma, M. and Kanagawa,
H. (1989) Effects of various cryoprotectants on the survival of mouse
embryos cryopreserved by the quick freezing method. Jpn. J. Vet. Res.,
37, 2939.
Acker, J.P. and McGann, L.E. (2003) Protective effect of intracellular ice
during freezing? Cryobiology, 46, 197202.
Al-aghbari, A.M. and Menino, A.R. (2002) Survival of oocytes recovered
from vitried sheep ovarian tissues. Anim. Reprod. Sci., 71, 101110.
Ali, J. and Shelton, J.N. (1993a) Vitrication of preimplantation stages of
mouse embryos. J. Reprod. Fertil., 98, 459465.
Ali, J. and Shelton, J.N. (1993b) Design of vitrication solutions for the
cryopreservation of embryos. J. Reprod. Fertil., 99, 471477.
Anchordoguy, T.J., Rudolph, A.S., Carpenter, J.F. and Crowe, J.H. (1987)
Modes of interaction of cryoprotectants with membrane phospholipids
during freezing. Cryobiology, 24, 32431.
Anchordoguy, T., Carpenter, J.F., Loomis, S.H. and Crowe, J.H. (1988)
Mechanisms of interaction of amino acids with phospholipid bilayers
during freezing. Biochim. Biophys Acta, 946, 299306.
Anchordoguy, T.J., Cecchini, C.A., Crowe, J.H. and Crowe, L.M. (1991)
Insights into the cryoprotective mechanism of dimethyl sulfoxide for
phospholipid bilayers. Cryobiology, 28, 467473.
Anchordoguy, T.J., Carpenter, J.F., Crowe, J.H. and Crowe, L.M. (1992)
599
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Vitrication and other rapid cooling protocols provide an alternative to slow cooling. These cooling protocols now generally use
short equilibration times (down to a few seconds), fast cooling
rates (>200C/min) and no expensive equipment other than the
storage tanks for the cryopreserved specimens. Rapid cooling
procedures use the same cryoprotectant chemicals as slow cooling
protocols, but a high total solute concentration (cryoprotectants
and additives) is used to ensure that sufcient dehydration and
cryoprotectant permeation occurs before cooling to 196C
commences. There are now many different rapid cooling protocols
differing in how much and which chemicals are used in the
solutions, how and when the cryoprotectant is added and/or
removed (number of steps, equilibration temperatures and times)
and how the specimen is cooled and warmed (volume, rate,
container). In species in which rapid cooling procedures have been
optimized, cryopreservation causes almost no loss (e.g. Shaw et al.,
1991b, 2000a; Dinnyes et al., 1995; Vajta, 2000; Kuleshova et al.,
2001; Zhu et al., 2001). Optimizing any cryopreservation procedure (slow or rapid) for human oocytes or embryos would be a
signicant advance, which would greatly benet human IVF
clinics.
Further information on cryopreservation can be obtained from
the websites of companies and universities e.g. the IMV site (http://
www.cryobiosystem-imv.com/Cryobiology/CONSCBS.htm), and
Xytex (www.xytex.com/cryobiology).
Temperature-dependent perturbation of phospholipid bilayers by
dimethylsulfoxide. Biochim. Biophys Acta, 1104, 11722.
Arav, A., Zeron, Y., Leslie, S.B., Behboodi, E., Anderson, G.B. and Crowe,
J.H. (1996) Phase transition temperature and chilling sensitivity of bovine
oocytes. Cryobiology, 33, 589599.
Arav, A., Pearl, M. and Zeron, Y. (2000) Does membrane lipid prole explain
chilling sensitivity and membrane lipid phase transition of spermatozoa
and oocytes? CryoLetters, 21, 179186.
Arav, A., Yavin, S., Zeron, Y., Natan, D., Dekel, I. and Gacitua, H. (2002)
New trends in gamete's cryopreservation. Mol. Cell. Endocrinol., 187,
7781.
Armitage, W.J. and Juss, B.K. (2003) Freezing monolayers of cells without
gap junctions. Cryobiology, 46, 194196.
Baltz, J.M., Smith, S.S., Biggers, J.D. and Lechene, C. (1997) Intracellular ion
concentrations and their maintenance by Na+/K(+)-ATPase in
preimplantation mouse embryos. Zygote, 5, 19.
Baril, G., Traldi, A.L., Cognie, Y., Leboeuf, B., Beckers, J.F. and Mermillod,
P. (2001) Successful direct transfer of vitried sheep embryos.
Theriogenology, 56, 299305.
Bautista, J.A., Takahashi, Y. and Kanagawa, H. (1997) In vitro viability of
mouse 8-cell embryos vitried in a simple solution of ethylene glycol.
Jpn. J. Vet. Res., 45, 6773.
Begin, I., Bhatia, B., Baldassarre, H., Dinnyes, A. and Keefer, C.L. (2003)
Cryopreservation of goat oocytes and in vivo derived 2- to 4-cell embryos
using the cryoloop (CLV) and solid-surface vitrication (SSV) methods.
Theriogenology, 59, 18391850.
Benia, J.L., Letur-Konirsch, H., Collin, G., Devaux, A., Kuttenn, F.,
Madelenat, P., Brun-Vezinet, F. and Feldmann, G. (2000) Safety of
cryopreservation straws for human gametes or embryos: a preliminary
study with human immunodeciency virus-1. Hum. Reprod., 15, 2186
2189.
Bernard, A. and Fuller, B.J. (1996) Cryopreservation of human oocytes: a
review of current problems and perspectives. Hum. Reprod. Update, 2,
193207.
Bielanski, A., Nadin-Davis, S., Sapp, T. and Lutze-Wallace, C. (2000) Viral
contamination of embryos cryopreserved in liquid nitrogen. Cryobiology,
40, 110116.
Bronshteyn, V.L. and Steponkus, P.L. (1993) Calorimetric studies of freezeinduced dehydration of phospholipids. Biophys. J., 65, 18531865.
Carroll, J., Wood, M.J. and Whittingham, D.G. (1993) Normal fertilization
and development of frozenthawed mouse oocytes: protective action of
certain macromolecules. Biol. Reprod., 48, 606612.
Chen, S.U., Lien, Y.R., Cheng Y.Y., Chen, H.F., Ho, H.N. and Yang, Y.S.
(2001) Vitrication of mouse oocytes using closed pulled straws (CPS)
achieves a high survival and preserves good patterns of meiotic spindles,
compared with conventional straws, open pulled straws (OPS) and grids.
Hum. Reprod., 16, 23502356.
Chen, S.U., Lien, Y.R., Chao, K.H., Ho, H.N., Yang, Y.S. and Lee, T.Y.
(2003) Effects of cryopreservation on meiotic spindles of oocytes and its
dynamics after thawing: clinical implications in oocyte freezinga
review article. Mol. Cell. Endocrinol., 202, 101107.
Critser, J.K. Agca, Y. and Gunasena, K.T. (1997) The cryobiology of
mammalian oocytes. In Karow, A.M. and Critser, J.K. (eds), Reproductive
Tissue Banking: Scientic Principles. Academic Press, New York, pp.
329357.
Critser, J.K. and Russell, R.J. (2000) Cryobiology of embryos germcells, and
ovaries. ILAR J., 41, 183237.
Critser, J.K. and Mobraaten, L.E. (2000) Cryopreservation of murine
spermatozoa. ILAR J., 41, 197206.
Crowe, J.H., Crowe, L.M., Carpenter, J.F., Rudolph, A.S., Wistrom, C.A.,
Spargo, B.J. and Anchordoguy, TJ. (1988) Interactions of sugars with
membranes. Biochim. Biophys. Acta, 947, 367384.
de la Pena, E.C., Takahashi, Y., Katagiri, S., Atabay, E.C. and Nagano, M.
(2002) Birth of pups after transfer of mouse embryos derived from
vitried preantral follicles. Reproduction, 123, 593600.
Dick, D.A. (1978) The distribution of sodium, potassium and chloride in the
nucleus and cytoplasm of Bufo bufo oocytes measured by electron
microprobe analysis. J. Physiol., 284, 3753.
Dinnyes, A., Wallace, G.A. and Rall, W.F. (1995) Effect of genotype on the
efciency of mouse embryo cryopreservation by vitrication or slow
freezing methods. Mol. Reprod. Dev., 40, 429435.
Dobrinsky, J.R. (2001) Cryopreservation of pig embryos: adaptation of
vitrication technology for embryo transfer. Reproduction, 58 (Suppl.),
325333.
Drobnis, E.Z., Crowe, L.M., Berger ,T., Anchordoguy, T.J., Overstreet, J.W.
J.M.Shaw and G.M.Jones
600
toxic effects on nuclei and cytoplasm of mouse zygotes. Cryobiology, 25,
197202.
Koseoglu, M., Eroglu, A., Toner, M. and Sadler, K.C. (2001) Starsh oocytes
form intracellular ice at unusually high temperatures. Cryobiology, 43,
248259.
Krag, K.T Koehler, I.-M. and Wright Jr, R.W. (1985) A method for freezing
early murine embryos by plunging directly into liquid nitrogen.
Theriogenology, 23, 199.
Krivokharchenko, A.S., Serobian, G.A., Shakhbazian, A.K. and Sadovnikov,
V.B. (1996) [Ultrafast freezing of the embryos of outbred and inbred
mouse strains] Ontogenez, 27, 300304 (from Pub Med abstract only).
Kuleshova, L.L. and Lopata, A. (2002) Vitrication can be more favorable
than slow cooling. Fertil. Steril., 78, 449454.
Kuleshova, L.L. and Shaw, J.M. (2000) A strategy for rapid cooling of mouse
embryos within a double straw to eliminate the risk of contamination
during storage in liquid nitrogen. Hum. Reprod., 15, 26042609.
Kuleshova, L., Gianaroli, L., Magli, C., Ferraretti, A. and Trounson, A.
(1999a) Birth following vitrication of a small number of human oocytes:
case report. Hum. Reprod., 14, 30773079.
Kuleshova, L.L. MacFarlane, D.R., Trounson, A.O. and Shaw, J.M. (1999b)
Sugars exert a major inuence on the vitrication properties of ethylene
glycol-based solutions and have low toxicity to embryos and oocytes.
Cryobiology, 38, 119130.
Kuleshova, L.L., Shaw, J.M. and Trounson, A.O. (2001) Studies on replacing
most of the penetrating cryoprotectant by polymers for embryo
cryopreservation. Cryobiology, 43, 2131
Kyuwa, S., Nishikawa, T., Kaneko, T., Nakashima, T., Kawano, K.,
Nakamura, N., Noguchi, K., Urano, T., Itoh, T. and Nakagata, N.
(2003) Experimental evaluation of cross-contamination between
cryotubes containing mouse 2-cell embryos and murine pathogens in
liquid nitrogen tanks. Exp. Anim., 52, 6770.
Landa, V. and Tepla, O. (1990) Cryopreservation of mouse 8-cell embryos in
microdrops. Fol. Biol. (Praha), 36, 153158.
Lane, M. and Gardner, D.K. (2001) Vitrication of mouse oocytes using a
nylon loop. Mol. Reprod. Dev., 58, 342347.
Lane, M., Schoolcraft, W.B. and Gardner, D.K. (1999) Vitrication of mouse
and human blastocysts using a novel cryoloop container-less technique.
Fertil. Steril., 72, 10731078.
Ledda, S., Leoni, G., Bogliolo, L. and Naitana, S. (2001) Oocyte
cryopreservation and ovarian tissue banking. Theriogenology, 55, 1359
1371.
Leibo, S.P. (1986) Cryobiology: preservation of mammalian embryos. Basic
Life Sci., 37, 251272.
Leibo, S.P., McGrath, J.J. and Cravalho, E.G. (1978) Microscopic observation
of intracellular ice formation in unfertilized mouse ova as a function of
cooling rate. Cryobiology, 15, 257271.
Letur-Konirsch, H., Collin, G., Sifer, C., Devaux, A., Kuttenn, F., Madelenat,
P., Brun-Vezinet, F., Feldmann, G. and Benia, J.L. (2003) Safety of
cryopreservation straws for human gametes or embryos: a study with
human immunodeciency virus-1 under cryopreservation conditions.
Hum. Reprod., 18, 140144.
Liebermann, J., Tucker, M.J., Graham, J.R., Han, T., Davis, A. and Levy, M.J.
(2002) Blastocyst development after vitrication of multipronuclear
zygotes using the Flexipet denuding pipette. Reprod. Biomed. Online, 4,
146150.
Liu, J., Woods, E.J., Agca, Y., Critser, E.S. and Critser, J.K. (2000)
Cryobiology of rat embryos II: A theoretical model for the development
of interrupted slow freezing procedures. Biol. Reprod., 63, 13031312.
Lopez-Bejar, M. and Lopez-Gatius, F. (2002) Nonequilibrium
cryopreservation of rabbit embryos using a modied (sealed) open
pulled straw procedure. Theriogenology, 58, 15411552.
Mahadevan, M.M., McIntosh, Q., Miller, M.M., Breckinridge, S.M., Maris, M.
and Moutos, D.M. (1998) Formaldehyde in cryoprotectant propanediol
and effect on mouse zygotes. Hum. Reprod., 13, 979982.
Martinez, A.G. and Matkovic, M. (1998) Cryopreservation of ovine embryos:
slow freezing and vitrication. Theriogenology, 49, 10391049.
Martino, A., Pollard, J.W. and Leibo, S.P. (1996a) Effect of chilling bovine
oocytes on their developmental competence. Mol. Reprod. Dev., 45, 503
512.
Martino, A., Songsasen, N. and Leibo, S.P. (1996b); Development into
blastocysts of bovine oocytes cryopreserved by ultrarapid cooling. Biol.
Reprod., 54, 10591069.
Matson, P.L., Graeing, J., Junk, S.M., Yovich, J.L. and Edirisinghe, W.R.
(1997) Cryopreservation of oocytes and embryos: use of a mouse model
to investigate effects upon zona hardness and formulate treatment
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
and Crowe, J.H. (1993) Cold shock damage is due to lipid phase
transitions in cell membranes: a demonstration using sperm as a model. J.
Exp. Zool., 265, 432437.
Edashige, K., Asano, A., An, T.Z. and Kasai, M. (1999) Restoration of
resistance to osmotic swelling of vitried mouse embryos by short-term
culture. Cryobiology, 38, 273280.
Elliott, K. and Whelan, J. (eds) (1977) The Freezing of Mouse Embryos. CIBA
Foundation Symposium 52. Elsevier, Amsterdam. 340 pp.
Eroglu, A., Toner, M. and Toth, T.L. (2002) Benecial effect of microinjected
trehalose on the cryosurvival of human oocytes. Fertil. Steril., 77, 152
158.
Fabbri, R., Porcu, E., Marsella, T., Rocchetta, G., Venturoli, S. and Flamigni,
C. (2001) Human oocyte cryopreservation: new perspectives regarding
oocyte survival. Hum. Reprod., 16, 411416.
Fahy, G.M. (1989) Vitrication. In McGrath, J.J. and Diller, K.R. (eds), Low
Temperature Biotechnology: Emerging Applications and Engineering
Contributions. BED Vol. 10/HTD Vol. 98, pp. 113146.
Fahy, G.M., Levy, D.I. and Ali, S.E. (1987) Some emerging principles
underlying the physical properties, biological actions, and utility of
vitrication solutions. Cryobiology, 24, 196213.
Friedler, S., Giudice, L.C. and Lamb, E.J. (1988) Cryopreservation of embryos
and ova. Fertil. Steril., 49, 74364.
Gardner, D.K. and Lane, M. (2000) Embryo culture systems. In Trounson,
A.O. and Gardner, D. (eds), Handbook of In Vitro Fertilization, 2nd edn.
CRC Press, Boca Raton, FL, USA. Chap. 11, pp. 205264.
Gardner, D.K., Lane, M., Stevens, J. and Schoolcraft, W.B. (2003) Changing
the start temperature and cooling rate in a slow-freezing protocol
increases human blastocyst viability. Fertil. Steril., 79, 40710.
Hammerstedt, R.H. and Andrews, J.C. (1997) Metabolic support of
normothermia. In Karow, A.M. and Critser, J.K. (eds), Reproductive
Tissue Banking: Scientic Principles. Academic Press, New York, pp.
139166.
Hawkins, A.E., Zuckerman, M.A., Briggs, M., Gilson, R.J., Goldstone, A.H.,
Brink, N.S. and Tedder, R.S. (1996) Hepatitis B nucleotide sequence
analysis: linking an outbreak of acute hepatitis B to contamination of a
cryopreservation tank. J. Virol. Methods, 60, 8188.
Hochi, S., Akiyama, M., Minagawa, G., Kimura, K. and Hanada, A. (2001)
Effects of cooling and warming rates during vitrication on fertilization
of in vitro-matured bovine oocytes. Cryobiology, 42, 6973.
Hochi, S., Terao, T., Kamei, M., Hirao, M. and Hirabayashi, M. (2003)
Vitrication of pronuclear stage rabbit zygotes by different ultra-rapid
cooling procedures Theriogenology, 59, 303, abstract.
Isachenko, V., Alabart, J.L., Dattena, M., Nawroth, F., Cappai, P., Isachenko,
E., Cocero, M.J., Olivera, J., Roche, A., Accardo, C., Krivokharchenko,
A. and Folch, J. (2003) New technology for vitrication and eld
(microscope-free) warming and transfer of small ruminant embryos.
Theriogenology, 59, 12091218.
Ishida, G.M., Saito, H., Ohta, N., Takahashi, T., Ito, M.M., Saito, T.,
Nakahara, K. and Hiroi, M. (1997) The optimal equilibration time for
mouse embryos frozen by vitrication with trehalose. Hum. Reprod., 12,
12591262.
Kaidi, S., Van Langendonckt, A., Massip, A., Dessy, F. and Donnay, I. (1999)
Cellular alteration after dilution of cryoprotective solutions used for the
vitrication of in vitro-produced bovine embryos. Theriogenology, 52,
515525.
Karlsson, J.O. (2001) A theoretical model of intracellular devitrication.
Cryobiology, 42, 154169.
Karow, A.M. (2001) Cryobiology 2001 for mammalian embryologists.
www.xytex.com/cryobiology (%202001%20-%202.pdf)
Karow, A.M. and Critser, J.K. (eds) (1997) Reproductive Tissue Banking:
Scientic Principles. Academic Press, New York, 472pp.
Kasai, M., Niwa, K. and Iritani, A. (1980) Survival of mouse embryos frozen
and thawed rapidly. J. Reprod. Fertil., 59, 5156.
Kasai, M., Komi, J.H., Takakamo, A., Tsudera, H., Sakurai, T. and Machida,
T. (1990) A simple method for mouse embryo cryopreservation in a low
toxicity vitrication solution, without appreciable loss of viability. J.
Reprod. Fertil., 89, 9197.
Kasai, M., Zhu, S.E., Pedro, P.B., Nakamura, K., Sakurai, T. and Edashige, K.
(1996) Fracture damage of embryos and its prevention during vitrication
and warming. Cryobiology, 33, 459464.
Kong, I.K., Lee, S.I., Cho, S.G., Cho, S.K. and Park, C.S. (2000) Comparison
of open pulled straw (OPS) versus glass micropipette (GMP) vitrication
in mouse blastocysts. Theriogenology, 53, 18171826.
Kono, T. and Tsunoda, Y. (1988) Ovicidal effects of vitrication solution and
the vitricationwarming cycle and establishment of the proportion of
Terminology for cryopreservation techniques
Rall, W.F. (1987) factors affecting the survival of mouse embryos
cryopreserved by vitrication Cryobiology, 24, 387402.
Rall, W.F. and Fahy, G.M. (1985) ice-free cryopreservation of mouse embryos
at 196C by vitrication. Nature, 313, 573574.
Rall, W.F. and Wood, M.J. (1994) High in vitro and in vivo survival of day 3
mouse embryos vitried or frozen in a non-toxic solution of glycerol and
albumin. J. Reprod. Fertil., 101, 681688.
Rall, W.R. Reid, D.S. and Farrant, J. (1980) Innocuous biological freezing
during warming. Nature, 286, 511514.
Rall, W.F., Mazur, P. and McGrath, J.J. (1983) Depression of the icenucleation temperature of rapidly cooled mouse embryos by glycerol and
dimethyl sulfoxide. Biophys. J., 41, 112.
Scheffen, B., Van Der Zwalmen, P. and Massip, A. (1986) A simple and
efcient procedure for preservation of mouse embryos by vitrication.
CryoLetters, 7, 260269.
Schulson, E.M. (1999) The structure and mechanical behavior of ice. J. O. M.,
51, 2127.
Shaw, J.M. and Kasai, M. (2001) Embryo cryopreservation for transgenic
mouse lines. In Kola, I. and Tymms, M. (eds), Methods in Molecular
Biology, Gene Knockout Protocols, 158. Humana Press, Totowa, NJ,
USA. Chap. 25, 397419.
Shaw, J.M. and Trounson, A.O. (1989) Effect of dimethyl sulfoxide and
protein concentration on the viability of two-cell mouse embryos frozen
with a rapid freezing technique. Cryobiology, 26, 413421.
Shaw, J.M., Diotallevi, L. and Trounson, A.O. (1988) Ultrarapid embryo
freezing: effect of dissolved gas and pH of the freezing solutions and
straw irradiation. Hum. Reprod., 3, 905909.
Shaw, J.M., Kola, I., MacFarlane, D.R. and Trounson, A.O. (1991a) An
association between chromosomal abnormalities in rapidly frozen 2-cell
mouse embryos and the ice-forming properties of the cryoprotective
solution. J. Reprod. Fertil., 91, 918.
Shaw, J.M., Diotallevi, L. and Trounson, A.O. (1991b) A simple rapid
dimethyl sulphoxide freezing technique for the cryopreservation of onecell to blastocyst stage preimplantation mouse embryos. Reprod. Fertil.
Dev., 3, 621626.
Shaw, J.M., Ward, C. and Trounson, A.O. (1995a) Survival of mouse
blastocysts slow cooled in propanediol or ethylene glycol is inuenced by
the thawing procedure, sucrose and antifreeze proteins. Theriogenology,
43, 12891300.
Shaw, J.M., Ward, C. and Trounson, A.O. (1995b) Evaluation of propanediol,
ethylene glycol, sucrose and antifreeze proteins on the survival of slow
cooled mouse pronuclear and 4-cell embryos. Hum. Reprod., 10, 396
402.
Shaw, J., Kuleshova, L., MacFarlane, D. and Trounson, A.O. (1997)
Vitrication properties of solutions of ethylene glycol in saline
containing PVP, coll or dextran. Cryobiology, 35, 219229.
Shaw, J.M., Oranratnachai, A. and Trounson, A.O. (2000a) Fundamental
cryobiology of mammalian oocytes and ovarian tissues. Theriogenology,
53, 5972.
Shaw, J.M., Oranratnachai, A. and Trounson, A.O. (2000b) Cryopreservation
of oocytes and embryos. In Trounson, A.O. and Gardner, D. (eds),
Handbook of In Vitro Fertilization, 2nd edn. CRC Press, Boca Raton, FL,
USA. Chap. 16, pp. 373412.
Shaw, P.W., Bernard, A.G., Fuller, B.J., Hunter, J.H. and Shaw, R.W. (1992)
Vitrication of mouse oocytes using short cryoprotectant exposure:
effects of varying exposure times on survival. Mol. Reprod. Dev., 33,
210214.
Shi, X., Datta, A.K. and Mukherjee, Y. (1998) Thermal stresses from large
volumetric expansion during freezing of biomaterials. J. Biomech. Engng,
120, 720726.
Siegel-Itzkovich, J. (2003) Tel Aviv hospital destroys frozen embryos by
mistake. Br. Med. J., 326 (7379), 12.
Songsasen, N., Ratterree, M.S., VandeVoort, C.A., Pegg, D.E. and Leibo, S.P.
(2002) Permeability characteristics and osmotic sensitivity of rhesus
monkey (Macaca mulatta) oocytes. Hum. Reprod., 17, 18751884.
Stachecki, J.J., Cohen, J. and Willadsen, S.M. (1998) Cryopreservation of
unfertilized mouse oocytes: the effect of replacing sodium with choline in
the freezing medium. Cryobiology, 37, 346354.
Stachecki, J.J., Cohen, J., Schimmel, T. and Willadsen, S.M. (2002) Fetal
development of mouse oocytes and zygotes cryopreserved in a
nonconventional freezing medium. Cryobiology, 44, 513.
Sutton, R.L. (1992) Critical cooling rates for aqueous cryoprotectants in the
presence of sugars and polysaccharides. Cryobiology, 29, 585598.
Szell, A. and Shelton, J.N. (1987) Osmotic and cryoprotective effects of
601
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
strategies in an in-vitro fertilization programme. Hum. Reprod., 12, 1550
1553.
Matsumoto, H., Jiang, J.Y., Tanaka, T., Sasada, H. and Sato, E. (2001)
Vitrication of large quantities of immature bovine oocytes using nylon
mesh. Cryobiology, 42, 139144.
Mazur, P. (1984) The freezing of living cells: mechanisms and implications.
Am. J. Physiol., 247 (3 Pt 1), C125142.
Mazur, P. (1990) Equilibrium, quasi-equilibrium, and nonequilibrium freezing
of mammalian embryos. Cell Biophys., 17, 5392.
Mazur, P., Leibo, S. and Chu, E.H.Y. (1972) A two-factor hypothesis of
freezing injury: evidence from Chinese hamster tissue culture cells. Exp.
Cell Res., 71, 345355.
McWilliams, R.B., Gibbons, W.E. and Leibo, S.P. (1995) Osmotic and
physiological responses of mouse zygotes and human oocytes to monoand disaccharides. Hum. Reprod., 10, 11631171.
Miller, R.H. and Mazur, P. (1976) Survival of frozenthawed human red cells
as a function of cooling and warming velocities. Cryobiology, 13, 404
414.
Misumi, K., Suzuki, M., Sato, S. and Saito, N. (2003) Successful production of
piglets derived from vitried morulae and early blastocysts using a
microdroplet method. Theriogenology, 60, 253260.
Mukaida, T., Wada, S., Takahashi, K., Pedro, P.B., An, T.Z. and Kasai, M.
(1998) Vitrication of human embryos based on the assessment of
suitable conditions for 8-cell mouse embryos. Hum. Reprod., 13, 2874
2879.
Mukaida, T., Nakamura, S., Tomiyama, T., Wada, S., Oka, C., Kasai, M. and
Takahashi, K. (2003) Vitrication of human blastocysts using cryoloops:
clinical outcome of 223 cycles. Hum. Reprod., 18, 384391.
Naitana, S., Ledda, S., Loi, P., Leoni, G., Bogliolo, L., Dattena, M. and
Cappai, P. (1997) Polyvinyl alcohol as a dened substitute for serum in
vitrication and warming solutions to cryopreserve ovine embryos at
different stages of development. Anim. Reprod. Sci., 48, 247256.
Nakagata, N. (1989) High survival rate of unfertilized mouse oocytes after
vitrication. J. Reprod. Fertil., 87, 479483.
Newton, H., Pegg, D.E., Barrass, R. and Gosden, R.G. (1999) Osmotically
inactive volume, hydraulic conductivity, and permeability to dimethyl
sulphoxide of human mature oocytes. J. Reprod. Fertil., 117, 2733.
Ohboshi, S., Fujihara, N., Yoshida, T. and Tomogane, H. (1997) Usefulness of
polyethylene glycol for cryopreservation by vitrication of in vitroderived bovine blastocysts. Anim. Reprod. Sci., 48, 2736.
O'Neil, L., Paynter, S.J. and Fuller, B.J. (1997) Vitrication of mature mouse
oocytes: improved results following addition of polyethylene glycol to a
dimethyl sulfoxide solution. Cryobiology, 34, 295301.
O'Neil, L., Paynter, S.J., Fuller, B.J., Shaw, R.W. and DeVries, A.L. (1998)
Vitrication of mature mouse oocytes in a 6 M Me2SO solution
supplemented with antifreeze glycoproteins: the effect of temperature.
Cryobiology, 37, 5966.
Papadopoulos, S., Rizos, D., Duffy, P., Wade, M., Quinn, K., Boland, M.P.
and Lonergan, P. (2002) Embryo survival and recipient pregnancy rates
after transfer of fresh or vitried, in vivo or in vitro produced ovine
blastocysts. Anim. Reprod. Sci., 74, 3544.
Papis, K., Shimizu, M. and Izaike, Y. (2000) Factors affecting the survivability
of bovine oocytes vitried in droplets. Theriogenology, 54, 651658.
Parks, J.E. (1997) Hypothermia and mammalian gametes In Karow, A.M. and
Critser, J.K. (eds), Reproductive Tissue Banking: Scientic Principles.
Academic Press, New York, pp. 229261.
Paynter, S.J., Fuller, B.J. and Shaw, R.W. (1997a) Temperature dependence of
mature mouse oocyte membrane permeabilities in the presence of
cryoprotectant. Cryobiology, 34, 122130.
Paynter, S., Cooper, A., O'Neil, L. and Fuller, B. (1997b) Use of amiloride to
reduce osmotic stress injury of mouse oocytes during cryopreservation.
Mol. Hum. Reprod., 3, 457.
Paynter, S.J., Fuller, B.J. and Shaw, R.W. (1999a) Temperature dependence of
KedemKatchalsky membrane transport coefcients for mature mouse
oocytes in the presence of ethylene glycol. Cryobiology, 39, 169176.
Paynter, S.J., Cooper, A., Gregory, L., Fuller, B.J. and Shaw, R.W. (1999b)
Permeability characteristics of human oocytes in the presence of the
cryoprotectant dimethylsulphoxide. Hum. Reprod., 14, 23382342.
Pedro, P.B., Zhu, S.E., Makino, N., Sakurai, T., Edashige, K. and Kasai, M.
(1997) Effects of hypotonic stress on the survival of mouse oocytes and
embryos at various stages. Cryobiology, 35, 150158.
Poledna, J. and Berger, W. (1996) A mathematical model of temperature
distribution in frozen tissue. Gen. Physiol. Biophys., 15, 315.
Pollard, J.W. and Leibo, S.P. (1994) Chilling sensitivity of mammalian
embryos. Theriogenology, 41, 101106.
J.M.Shaw and G.M.Jones
602
Vasuthevan, S., Ng, S.C., Edirisinghe, R., Bongso, A. and Ratnam, S. (1992)
The evaluation of various culture media in combination with
dimethylsulfoxide for ultrarapid freezing of murine embryos. Fertil.
Steril., 58, 12501253.
Wang, J.H. (2000) A comprehensive evaluation of the effects and mechanisms
of antifreeze proteins during low-temperature preservation. Cryobiology,
41, 19.
Williams, T.J. and Johnson, S.E. (1986) A method for one-step freezing of
mouse embryos Theriogenology, 26, 125133.
Wilmut, I. (1972) The effect of cooling rate, warming rate, cryoprotective
agent and stage of development on survival of mouse embryos during
freezing and thawing. Life Sci., 11, 10711079.
Wilson, L. and Quinn, P. (1989) Development of mouse embryos
cryopreserved by an ultra-rapid method of freezing. Hum. Reprod., 4,
8690.
Wowk, B. and Fahy, G.M. (2002) Inhibition of bacterial ice nucleation by
polyglycerol polymers. Cryobiology, 44, 1423.
Wowk, B., Darwin, M., Harris, S.B., Russell, S.R. and Rasch, C.M. (1999)
Effects of solute methoxylation on glass-forming ability and stability of
vitrication solutions Cryobiology, 39, 215227.
Wowk, B., Leitl, E., Rasch, C.M., Mesbah-Karimi, N., Harris, S.B. and Fahy,
G.M. (2000) Vitrication enhancement by synthetic ice blocking agents.
Cryobiology, 40, 228236.
Yeoman, R.R., Gerami-Naini, B., Mitalipov, S., Nusser, K.D., WidmannBrowning, A.A. and Wolf, D.P. (2001) Cryoloop vitrication yields
superior survival of Rhesus monkey blastocysts. Hum. Reprod., 16, 1965
1969.
Zeron, Y., Ocheretny, A., Kedar, O., Borochov, A., Sklan, D. and Arav, A.
(2001) Seasonal changes in bovine fertility: relation to developmental
competence of oocytes, membrane properties and fatty acid composition
of follicles. Reproduction, 121, 447454.
Zeron, Y., Sklan, D. and Arav, A. (2002) Effect of polyunsaturated fatty acid
supplementation on biophysical parameters and chilling sensitivity of ewe
oocytes. Mol. Reprod. Dev., 61, 271278.
Zhu, S.E., Kasai, M., Otoge, H., Sakurai, T. and Machida, T. (1993)
Cryopreservation of expanded mouse blastocysts by vitrication in
ethylene glycol-based solutions. J. Reprod. Fertil., 98, 139145.
Zhu, S.E., Sakurai, T., Edashige, K., Machida, T. and Kasai, M. (1996)
Cryopreservation of zona-hatched mouse blastocysts. J. Reprod. Fertil.,
107, 3742.
Zhu, S.E., Zeng, S.M., Yu, W.L., Li, S.J., Zhang, Z.C. and Chen, Y.F. (2001)
Vitrication of in vivo and in vitro produced ovine blastocysts. Anim.
Biotechnol., 12, 193203.
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
glycerolsucrose solutions on day-3 mouse embryos. J. Reprod. Fertil.,
80, 309316.
Takeda, T., Elsden, R.P. and Seidel, G.E. Jr (1984) cryopreservation of mouse
embryos by direct plunging into liquid nitrogen Theriogenology, 21, 266.
Tedder, R.S., Zuckerman, M.A., Goldstone, A.H., Hawkins, A.E., Fielding,
A., Briggs, E.M., Irwin, D., Blair, S., Gorman, A.M., Patterson, K.G. et al.
(1995) Hepatitis B transmission from contaminated cryopreservation
tank. Lancet, 346 (8968), 137140.
Tomlinson, M. and Sakkas, D. (2000) Is a review of standard procedures for
cryopreservation needed?: safe and effective cryopreservationshould
sperm banks and fertility centres move toward storage in nitrogen vapour?
Hum. Reprod., 15, 24602463.
Toner, M., Cravalho, E.G., Karel, M. and Armant, D.R. (1991)
Cryomicroscopic analysis of intracellular ice formation during freezing
of mouse oocytes without cryoadditives. Cryobiology, 28, 5571.
Trad, F.S., Toner, M. and Biggers, J.D. (1999) Effects of cryoprotectants and
ice-seeding temperature on intracellular freezing and survival of human
oocytes. Hum. Reprod., 14, 15691577.
Trounson, A. (1986) Preservation of human eggs and embryos. Fertil. Steril.,
46, 112.
Trounson, A., Peura, A., Freemann, L. and Kirby, C. (1988) Ultrarapid
freezing of early cleavage stage human embryos and eight-cell mouse
embryos. Fertil. Steril., 49, 822826.
Ushijima, H., Yamakawa, H. and Nagashima, H. (1999) Cryopreservation of
bovine pre-morula-stage in vitro matured/in vitro fertilized embryos after
delipidation and before use in nucleus transfer. Biol. Reprod., 60, 534
539.
Vajta, G. (2000) Vitrication of the oocytes and embryos of domestic animals.
Anim. Reprod. Sci., 6061, 357364.
Vajta, G., Holm, P., Kuwayama, M., Booth, P.J., Jacobsen, H., Greve, T. and
Callesen, H. (1998) Open pulled straw (OPS) vitrication: a new way to
avoid cryoinjuries of mammalian ova and embryos. Mol. Reprod. Dev.,
51, 5358.
VanBlerkom, J. (1989) Maturation at high frequency of germinal-vesicle-stage
mouse oocytes after cryopreservation: alterations in cytoplasmic, nuclear,
nucleolar and chromosomal structure and organization associated with
vitrication. J. Hum. Reprod., 4, 883898.
Vandervorst, M., Vanderzwalmen, P., Standaart, V. et al. (2001) Blastocyst
transfer after vitrication in a hemi-straw (HS) system. Hum. Reprod., 16
(Abstract book 1) 153154, Abstract P-133.
Vanderzwalmen, P., Bertin, G., Debauche, Ch., Standaert, V., van Roosendaal,
E.,Vandervorst, M., Bollen, N., Zech, H., Mukaida, T., Takahashi, K. and
Schoysman, R. (2002) Births after vitrication at morula and blastocyst
stages: effect of articial reduction of the blastocoelic cavity before
vitrication. Hum. Reprod., 17, 744751.
Terminology for cryopreservation techniques
Appendix 1
Table X. Terminology for equipment/hardware which can be used for cryopreservation
`Cryotop'
Ampoule
Box
Cane
Canister
Closed pulled straw
Cryoloop, nylon loop,
loop
Dewar
Flexipet
Forceps
Freezing machine(s)/
biological
freezer
Grid
Heat sealer
Impulse sealer
Insulation
Inventory
Liquid nitrogen
Liquid nitrogen slush
603
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Capillary
A very ne strip onto which oocytes or embryos can be loaded. The strip is then immersed into liquid nitrogen or nitrogen
vapour to give a very fast cooling rate (Hochi et al., 2003)
Ampoules made of glass were used by many early cryopreservation studies. They were usually ame-sealed, or sealed with
plastic caps. They have usually been replaced by plastic vials, or other freezing containers, in part because of the risk of
explosion with attendant spread of glass shards. Mainly used now for slow cooling
Cardboard or plastic boxes are commonly used as storage devices to hold straws or vials within a tank. Not as widely used
in assisted reproduction as goblets or canes. Mainly used in slow cooling of cell lines
Commonly used storage device. Usually a ~30 cm long aluminium strip with four to six paired projections designed to grip
straws and goblets. A code (usually the patient ID) written on the cane facilitates retrieval of particular specimens in a tank.
Alternative storage devices are boxes and goblets
Storage device for canes and/or goblets that ts inside a storage vessel. Liquid nitrogen tanks commonly have six to 10 canisters
each one with a diameter that matches the opening (neck). The canisters are usually `parked' inside the tank along the wall, but
have an attached handle which allows them to be moved. Specimens (usually canes or goblets) can only be placed in a canister
when it is moved to the position directly under the tank opening. The canister can then be raised out of the tank via the tank
opening.
Glass capillaries are brittle when used for cryopreservation but were used by Hochi et al. (2001) to show interactions between
cooling and warming rates. They found for 7.2 mol/l ethylene glycol and 1.0 mol/l sucrose that `capillaries of 2000, 1400,
1000, 630 and 440 mm diameters provided cooling rates of 2000, 3000, 5000, 8000 and 12 000 C/min and warming rates of
5000, 8000, 17 000, 33 000 and 62 000C/min, respectively.' They observed that a moderate warming rate (3000C/min) gave
the highest post-thaw survival of bovine oocytes irrespective of the warming rate
Describes a new way of loading embryos into an open pulled straw (OPS, see below) for rapid cooling which prevents liquid
nitrogen from coming into direct contact with the cryoprotectant bead containing the oocyte/embryo. Cooling rate would be
approximately the same as for OPS, i.e. >20 000C/min
A thin nylon loop. Oocytes and embryos can be suspended in a cryoprotectant lm within the loop of nylon. Specialized vials
have been developed to handle and store the loops. They achieve a very high cooling rate (Lane et al., 1999; Yeoman et al.,
2001). Device initially developed for crystallography
Originally a double-walled silvered glass vessel in which the space between the walls is evacuated, and used for storage of
liquids at low temperature. Invented by the Scottish chemist Sir James Dewar. Now used more generally for any similar container
in which liquid nitrogen can be held. Can refer to large storage tanks or small (e.g. 1 l) containers for short-term use, e.g. for
bench-top work and to transport samples between rooms. Most commonly made from stainless steel. `Thermos'-type domestic
Dewars with glass walls should never be used, even for short-term work as they can explode because the caps have no pressure
vent, and the walls are not sufciently thick to cope with materials as cold as liquid nitrogen
A pipette tip which can be used as the container in which oocytes or embryos are cryopreserved. Rapid cooling only
(Liebermann et al., 2002)
Paired metal blades for holding specimens deep within the vapour phase or retrieving specimens from liquid nitrogen. Long
handles or blades, or specialized tools such as the `cryoclaw' are less likely to result in frost bite. Also used in
ice-nucleation/seeding. May damage straws if not equilibrated to their temperature or if used with too much pressure
For controlled rate cooling and warming of specimens. Different types include: (i) platform cooling devices and equivalent
where specimens are gradually being lowered into nitrogen vapour (vapour cooling); (ii) gradient freezers which gradually
move the specimen along an increasingly cold surface; (iii) chamber freezers in which controlled amounts of nitrogen vapour
are injected into the specimen chamber; (iv) uncontrolled or passive cooling devices which are placed for example in a 80C
freezer or on dry ice; (v) chamber freezers in which the outside of the chamber is at 196C and the specimen's temperature
is regulated by controlled warming of the chamber wall. Machines differ in: size, portability, durability, exibility
(range of options),
repeatability and controllability (e.g. over rates/times, number of programs etc.), purchase price, maintenance costs, number
and size of specimens, nitrogen consumption, lowest controllable temperature, seeding procedure, ease of loading/unloading,
ease of use, permanent record output and dependence on a reliable power supply and decibel output
Very thin 5 mm diameter copper or gold mesh upon which oocytes and embryos can be placed; the uid is then blotted away
from below, and the grids rapidly cooled. They can achieve a very high cooling rate (Martino et al., 1996a,b). Initially developed
for electron microscopy. Cooling rate; in nitrogen ~ 9000C/min and up to 30000C/min in nitrogen slush.
Piece of equipment (e.g. autoclave bag sealer or plastic bag sealer) used to close and seal plastic straws
Piece of equipment used to close and seal CBS straws (Cryo Bio System by IMV Technologies, France)
Material surrounding a storage tank to minimize heat transfer through the walls.
Sometimes used to refer to the hardware placed within storage vessels to hold the specimens
Nitrogen below its boiling point (196C). Often abbreviated LN2 or LN2
When the liquid nitrogen is at a temperature lower than its boiling point it becomes more viscous. The most common strategy
by which nitrogen slush is made is by applying a vacuum to liquid nitrogen. This increases the evaporation rate, which in turn
lowers the temperature due to evaporative cooling
J.M.Shaw and G.M.Jones
Table X Continued
Loading
Minimum drop size
Nitrogen
Nitrogen vapour
Opening size
Passive cooling
device
Seal
Sintered disk
Static holding time
Static nitrogen
consumption
Straw
Tank
Vapour shipper
Vial
Volume
Waterproof pens
604
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Open pulled straw
(OPS)
A term describing the process of transferring oocytes and embryos to the container in which they are to be cryopreserved (e.g.
oocytes are loaded into straws). To reduce the risk of contamination during storage, the loading procedure should avoid all
contact between the cryoprotectant solution (with the cells) and all surfaces that come in direct contact with liquid nitrogen or
other specimens. Failure to avoid this contact could result in contaminationfor example viruses have been transferred
between samples carried in the liquid nitrogen
A vitrication method in which nanolitre-sized drops can be cooled at up to 130 000C/min (Arav et al., 2002)
Air contains almost 80% nitrogen gas. It is liquid below its boiling point of 196C. Note the dangers associated with
asphyxiation by nitrogen gas, and frost burns from the liquid
Usually referring to the cold nitrogen gas that accumulates above the surface of liquid nitrogen. Specimens can be held deep
within the vapour phase as part of a cooling protocol. This gives a less abrupt cooling rate than plunging directly into liquid
nitrogen (see vapour cooling) due to the higher temperatures and the lower thermal transfer between nitrogen vapour
compared to transfer from LN2. This reduces the likelihood of physical damage (cracks and stress fractures) and may give as
good or better results than plunging into liquid nitrogen. However, some cell types (e.g. early pig embryos) appear to
benet from the highest possible cooling rates. Specimens are also commonly held in the vapour phase as the rst event in the
thawing step, to reduce the risk of specimens exploding due to trapped nitrogen, and to reduce this likelihood of stress
fractures (see stress fractures) (at very low temperatures (e.g. <196C) most material is brittle and is easily damaged by
non-uniform sudden temperature changes. Cooling and warming is best performed deep within a stable layer of vapour
where the temperature is <130C. To establish such a stable, cold, vapour layer the liquid nitrogen level must be well below
the rim of the container (>15 cm) and left to stand for >10 min without being disturbed by air currents
A type of container used to rapidly cool oocytes and embryos. Basically a plastic straw which has been narrowed and thinned
by stretching, and then cut at its thinnest point to give two `OPS' straws. These are usually loaded by capillary action. A straw
which is pulled to narrow its diameter so that it will take up ~2 ml of cryoprotectant. When immersed deeply into the vapour
phase or just let to touch the surface of liquid nitrogen the OPS gives a cooling rates of >20 000C/min (Vajta et al., 1998).
Slower rates for OPS straws have, however, been measured (5300C/min in nitrogen and 10 300C/min in slush)
(http://www.agri.gov.il/AnimalScience/Reproduction/Rep-Arav1.html)
The diameter of the opening in a storage tank. A larger opening allows bigger canisters/inventory to be used, but also
compromises the insulation rating, reducing the static holding time
A container which, when placed at a specic temperature (e.g. 80C), cools at a known rate, e.g. 1C/min. A non-controlled
cooling method used for slow cooling exemplied by the isopropyl alcohol lled `Mr frosty' (Nalgene) for cooling vials
Usually used in the context of straws. To prevent liquid nitrogen entering or the contents leaking out, straws can be sealed
with heat (thermosoldering), PVA or PVP powder, identication plugs, ball bearings, or ultrasonically with an impulse sealer
A porous disk, for example made of brass, which can be applied to the end of nitrogen hoses to control the ow and reduce
the likelihood of personal injury
The length of time taken for a liquid nitrogen storage vessel which is not used or re-lled to go from full to empty. Commonly
measured in weeks. Old or damaged tanks may have a greatly reduced static holding time (can be as little as hours)
The amount of nitrogen which boils off from a storage vessel which is not in use. Commonly 0.11 l a day. The rate of loss
can be
much higher in vessels which are opened frequently
Traditionally supplied as 0.25 or 0.5 ml plastic straws. Now available in a range of materials and congurations. Materials
include polyvinyl chloride (PVC), polyethylene terephthalate glycol (PETG) and high-security ionomeric resin (IR). Straws
may or may not be sealed after inserting specimens (see seal). The likelihood of leakage and breakage in liquid nitrogen is
lowest for soft straws (type `CBS', Cryo Bio System by IMV Technologies, France) sealed with an `impulse sealer', but we are not
aware of any instance where these have been used for rapid cooling procedures. Straws which have been stretched and then cut at the
narrowest point have a smaller diameter and thinner walls and are widely used for rapid cooling (see open pulled straws and closed
pulled straws). Cooling rates of ~200C/min (in vapour), >2000C/min (plunging in nitrogen) (Kasai et al., 1996) and 4000 C/min
for plunging in nitrogen slush.
Warming rate is ~2500 C/min (Rall and Fahy, 1985).
Usually refers to the container into which specimens are placed for long-term storage. Three main categories: liquid nitrogen
tanks, nitrogen vapour tanks and ultralow temperature (<130C) electrical freezers. Can also be referred to as a vessel or a Dewar
A specialized container to maintain specimens at low subzero temperatures during transport. They can be shipped in cars and
aeroplanes. They are pre-cooled with liquid nitrogen, but the nitrogen is then emptied out fully and specimens inserted. Their
well-insulated design allows a low subzero temperature to be maintained for days after the nitrogen has been emptied out.
Must be kept upright at all times during transport
Container, usually of 15 ml capacity. Used mainly for slow cooling of cells or gonadal tissue. Only vials which are made to
withstand cryopreservation should be used to reduce the risk of explosions. The size should be chosen to t the canes,
freezing machine and specimen. Vials differ in volume, the conguration of the base, the shape inside (U- or V-shape), internal
or external thread, the writing area, the type of O ring, and whether the lids can be colour-coded. They are bulky but easy to
load, and have a large surface on which to write ID details. Some manufacturers make vials with bar-coding printed on them.
Vials have been used for some mouse rapid cooling protocols but fast cooling rates can only be achieved if specimens are
dripped into vials precooled to a low subzero temperature. Vials take a long time to warm which means that they are not
suitable for all cryoprotectant solutions. Their thermal inertia makes accidental thawing unlikely
In relation to liquid nitrogen storage vessels, the amount of liquid nitrogen that they will hold (commonly between 20 and 120 l)
Water vapour condenses on cold surfaces, so it is very important that all information on surfaces which are going to become
cold is written with pencil or indelible waterproof pens or using specialized labelling devices. As most waterproof ink is
soluble in alcohol it is important that all information is copied or noted before the surface is wiped with alcohol (for example
if the surfaces are wiped with alcohol to sterilize them before opening them)
Terminology for cryopreservation techniques
Appendix 2
Table XI. Safety issues associated with the use of liquid nitrogen/low temperature preservation (issues are equally relevant to rapid and slow cooling
procedures)
Asphyxia
605
Downloaded from http://humupd.oxfordjournals.org/ by guest on August 18, 2015
Death from asphyxia has occurred in several laboratories around the world where liquid nitrogen is in routine use. Nitrogen gas is
odourless and colourless. It forms 80% of the air we breathe; the remaining 20% is mainly the essential oxygen. Nitrogen gas is
released from liquid nitrogen as it evaporates, and if ventilation is inadequate, it can displace the air, creating a hypoxic or anoxic
environment where there is a high risk of asphyxia. The risk is highest in enclosed, poorly ventilated rooms. A person can become
unconscious after just a few breaths of 100% nitrogen gas. A litre of liquid nitrogen produces ~1600 l of nitrogen gas (at room
temperature and pressure). Liquid nitrogen storage vessels should therefore be stored in well-ventilated rooms. Ideally that room should
have a regularly serviced oxygen meter/alarm. Travelling in a lift (elevator) together with a large nitrogen tank is not recommended.
Liquid nitrogen should not be transported in cars where nitrogen vapour can access the passenger compartment and cannot (legally) be
shipped by air. Specialized vessels (see vapour shippers/dry shippers) which keep specimens at low subzero temperatures without
giving off nitrogen gas are suitable for transport by car and by air
Burns
Tissue damage similar to heat-burns can be caused by exposure to extremely low temperatures, for example contact with liquid
nitrogen at a temperature of <196C. Skin exposed to this temperature will blister in the same way as a burn. A full face visor,
goggles or glasses should be worn when handling/dispensing liquid nitrogen because a splash to the eye could burn the cornea;
appropriate personal protective equipment should be worn to protect the rest of the body. See also `Frostbite'
Cold tolerant
Most containers become brittle at cryogenic temperatures. Only use those specically designed for cryopreservation. Containers such
containers
as Eppendorf tubes are not designed to tolerate low subzero temperatures and easily shatter/explode (see below)
Explosions
Explosions easily happen if great care is not taken when using liquid nitrogen. The most common cause is entry of liquid nitrogen
into a container (such as an unsuitable sample tube). This easily happens due to the reduction of pressure inside the vessel when
the air inside is cooled by placing in liquid nitrogen. When the vessel is rewarmed, the nitrogen boils and the resulting gas pressure
causes the container to explode. This is particularly common with containers not designed for use with liquid nitrogen. Flasks must
never be of the `thermos' type (silvered glass walls with a vacuum) as these can explode, lling whole laboratories with glass shards;
use instead stainless steel asks or polystyrene boxes with appropriate venting to prevent build up of internal pressure
Frostbite
Skin exposed to liquid nitrogen or any other surface cold enough to cause the cells in the skin to freeze will hurt, or if the injury is bad
enough it will blister. Injury is most commonly caused by cold metal surfaces, or ice, due to their high thermal conductivity. See
also `Burns'
Gloves
Used to protect the hands/ngers from frost bite/burns. The best sorts extend up the forearms, e.g. leather welder's gloves, or special
gloves (e.g. Sigma `cryogloves'). Gloves are not designed to allow the user to insert their hands into liquid nitrogen. The user should
use long-handled forceps or tongs to hold specimens within the vapour phase or retrieve specimens from within the liquid nitrogen
Goggles, visors
Used to protect the eyes/face in case of explosions or liquid nitrogen splashes.
Nitrogen level
For tanks as a back-up to provide a warning about low nitrogen levels. Sophisticated versions can be linked to a phone dial-up facility.
monitor
These devices are not failsafe and should be tested according to the manufacturer's recommendations, and routine physical checks of
nitrogen levels made at appropriate intervals
Oxygen meter
Used in liquid nitrogen storage facilities to provide warning of lowered oxygen levels in the air. Note that oxygen meters may need
regular servicing and routine testing to remain functional
Safety valves
Storage vessels for nitrogen (not the specimen storage tanks) hold and dispense nitrogen under pressure. Safety valves are required to
ensure that the pressure stays within safe limits. Pressurized vessels should be safety-tested (pressure-tested) at least every 10 years
Spills
A small spill of nitrogen is generally harmless unless it is to a particularly vulnerable part of the body (e.g. the eye). Larger spills can
cause serious injury. Clothes that become soaked by liquid nitrogen should be removed or at least held away from the body.
Alternatively the affected clothing can be ushed/soaked with water (e.g. using a safety shower) until the trapped LN2 is all vaporized
Transport/shipment For safety reasons, liquid nitrogen should not be transported by car or air. Vapour shippers can be transported by both cars and aeroplanes
Ventilation
When liquid nitrogen is used/held in enclosed spaces, the ventilation must be sufcient to prevent nitrogen gas accumulation
(see `Asphyxia')
Weight
Liquid nitrogen weighs approximately the same as water. Large storage vessels can be too heavy to move, in particular for small
persons. Canisters, or racks, within the storage vessels, particularly those of large capacity, can be very heavy. Tall tanks
exacerbate the problem. Use of appropriate lifting techniques, trolleys and other equipment must be considered
Вам также может понравиться
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)От EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Рейтинг: 4.5 из 5 звезд4.5/5 (121)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryОт EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryРейтинг: 3.5 из 5 звезд3.5/5 (231)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaОт EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaРейтинг: 4.5 из 5 звезд4.5/5 (266)
- Never Split the Difference: Negotiating As If Your Life Depended On ItОт EverandNever Split the Difference: Negotiating As If Your Life Depended On ItРейтинг: 4.5 из 5 звезд4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerОт EverandThe Emperor of All Maladies: A Biography of CancerРейтинг: 4.5 из 5 звезд4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingОт EverandThe Little Book of Hygge: Danish Secrets to Happy LivingРейтинг: 3.5 из 5 звезд3.5/5 (400)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeОт EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeРейтинг: 4 из 5 звезд4/5 (5794)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyОт EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyРейтинг: 3.5 из 5 звезд3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreОт EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreРейтинг: 4 из 5 звезд4/5 (1090)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersОт EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersРейтинг: 4.5 из 5 звезд4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnОт EverandTeam of Rivals: The Political Genius of Abraham LincolnРейтинг: 4.5 из 5 звезд4.5/5 (234)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceОт EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceРейтинг: 4 из 5 звезд4/5 (895)
- The Unwinding: An Inner History of the New AmericaОт EverandThe Unwinding: An Inner History of the New AmericaРейтинг: 4 из 5 звезд4/5 (45)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureОт EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureРейтинг: 4.5 из 5 звезд4.5/5 (474)
- Astm D3212.380331 1Документ3 страницыAstm D3212.380331 1anish_am2005Оценок пока нет
- Nauticus 3D BeamДокумент114 страницNauticus 3D BeamMinca AndreiОценок пока нет
- The Yellow House: A Memoir (2019 National Book Award Winner)От EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Рейтинг: 4 из 5 звезд4/5 (98)
- Schlumberger JET Manual 23 Fracturing Pump UnitsДокумент68 страницSchlumberger JET Manual 23 Fracturing Pump UnitsVladyslav67% (3)
- Gear Trains ProblemsДокумент9 страницGear Trains Problemsa c s Kumar100% (1)
- Engineering Data Analysis (Mod2)Документ7 страницEngineering Data Analysis (Mod2)Mark Peru100% (1)
- 38 Meter Wind Turbine Blade Design PDFДокумент47 страниц38 Meter Wind Turbine Blade Design PDFWalid MohammedОценок пока нет
- Ultrasonic Atomizing Nozzle SystemsДокумент40 страницUltrasonic Atomizing Nozzle SystemsAnonymous H8hysGxA100% (1)
- ReadmeДокумент11 страницReadmerandyprayogoОценок пока нет
- btw6 5 1 2300releasenotesДокумент8 страницbtw6 5 1 2300releasenotesLathamagan ParamananthamОценок пока нет
- Ipss PDFДокумент2 страницыIpss PDFFrida AnggaОценок пока нет
- Hum. Reprod. 2011 Mar 26 (3) 594-603, Figure 1Документ1 страницаHum. Reprod. 2011 Mar 26 (3) 594-603, Figure 1randyprayogoОценок пока нет
- 10128ART Simplified Embryo Rev5 WEBДокумент2 страницы10128ART Simplified Embryo Rev5 WEBrandyprayogoОценок пока нет
- Pi Is 1472648311006535Документ7 страницPi Is 1472648311006535randyprayogoОценок пока нет
- Ebm 2Документ7 страницEbm 2randyprayogoОценок пока нет
- Abdominal Myomectomy Increases Fertility Outcome 2161 0932.1000144 1Документ4 страницыAbdominal Myomectomy Increases Fertility Outcome 2161 0932.1000144 1randyprayogoОценок пока нет
- Bavaria Reverse RdsДокумент15 страницBavaria Reverse RdsDušan JovanovićОценок пока нет
- STK412 150 Sanyo PDFДокумент4 страницыSTK412 150 Sanyo PDFroger.Оценок пока нет
- Symbolic TBДокумент512 страницSymbolic TBJanet LeongОценок пока нет
- Material Characterization of Sugarcane Bagasseepoxy Composites For - 2022Документ5 страницMaterial Characterization of Sugarcane Bagasseepoxy Composites For - 2022bakhrul ilmiОценок пока нет
- Medium Voltage MZДокумент86 страницMedium Voltage MZsujiОценок пока нет
- ABSORPTION AND SP GRAVITY fine agg. QC 467,468- 19-11-2020 العلمين PDFДокумент1 страницаABSORPTION AND SP GRAVITY fine agg. QC 467,468- 19-11-2020 العلمين PDFjajajajОценок пока нет
- Single Aisle Technical Training Manual T1 (CFM 56) (LVL 2&3) LightsДокумент44 страницыSingle Aisle Technical Training Manual T1 (CFM 56) (LVL 2&3) LightsarunОценок пока нет
- ACI 517-2R-87 Standard Specification For Tolerances For Accelerated Curing at Atmospheric PressureДокумент17 страницACI 517-2R-87 Standard Specification For Tolerances For Accelerated Curing at Atmospheric PressureKiramat ShahОценок пока нет
- A Interview QuestionsДокумент363 страницыA Interview QuestionsJaishankar RenganathanОценок пока нет
- lm230wf1 Tla6Документ32 страницыlm230wf1 Tla6Charlie SierraОценок пока нет
- ICE Annex A Methodologies For RecyclingДокумент17 страницICE Annex A Methodologies For RecyclingChai Lin NyokОценок пока нет
- A340-Elec Emer ConfigДокумент13 страницA340-Elec Emer ConfigGerhard StorbeckОценок пока нет
- Elmer TutorialsДокумент65 страницElmer TutorialsmariomatoОценок пока нет
- Comparative Study of P&O and Inc MPPT AlgorithmsДокумент7 страницComparative Study of P&O and Inc MPPT AlgorithmsVinod KohliОценок пока нет
- Un 2 Terrazzo FlooringДокумент6 страницUn 2 Terrazzo FlooringRОценок пока нет
- Hydraulic Home ElevatorДокумент38 страницHydraulic Home ElevatorPatrisha SantosОценок пока нет
- Unit 16Документ35 страницUnit 16api-334267616Оценок пока нет
- Torsion MEДокумент24 страницыTorsion MEmohamed.hassan031Оценок пока нет
- Grdds ReviewДокумент33 страницыGrdds ReviewvaibhavbpatelОценок пока нет
- California Bearing Ratio, Evaluation and Estimation: A Study On ComparisonsДокумент4 страницыCalifornia Bearing Ratio, Evaluation and Estimation: A Study On ComparisonsAmyra MiaОценок пока нет
- Physics I Problems PDFДокумент1 страницаPhysics I Problems PDFbosschellenОценок пока нет
- Architecture Concerns of TST and NATO TST ToolДокумент9 страницArchitecture Concerns of TST and NATO TST Tooldorupara718747Оценок пока нет
- D Praveen Kumar ChaubeyДокумент2 страницыD Praveen Kumar ChaubeyyouvsyouОценок пока нет