Академический Документы
Профессиональный Документы
Культура Документы
Bkf1323-Organic Chemistry 21213 PDF
Загружено:
rushdiИсходное описание:
Оригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Bkf1323-Organic Chemistry 21213 PDF
Загружено:
rushdiАвторское право:
Доступные форматы
Universiti
Malaysia
PAHANG
EnIneeiing
CreatMty
FACULTY OF CHEMICAL & NATURAL RESOURCES ENGINEERING
FINAL EXAMINATION
COURSE
ORGANIC CHEMISTRY
COURSE CODE
BKF1323
LECTURER
ABDUL AZIZ MOHD AZODDEIN
CHE KU MOIIAMMAD FAIZAL BIN CHE
KU YAHYA
DATE
19 JUNE 2013
DURATION
3 HOURS
SESSION/SEMESTER
SESSION 2012/2013 SEMESTER II
PROGRAMME CODE :
BKB/BKC/BKG
INSTRUCTIONS TO CANDIDATE:
1.
2.
3.
4.
This question paper consists of FOUR (4) questions. Answer ALL questions.
All answers to a new question should start on new page.
All the calculations and assumptions must be clearly stated.
Candidates are not allowed to bring any material other than those allowed by
the invigilator into the examination room.
EXAMINATION REQUIREMENTS: -
DO NOT TURN THIS PAGE UNTIL YOU ARE TOLD TO DO SO
This examination paper consists of EIGHT (8) printed pages including front page.
BKBIBKC/BKG/121311JBKF1323
CONFIDENTIAL
QUESTION 1
a) Give the IUPAC names for the following compounds.
i)
(1 Mark)
ii)
(1 Mark)
iii)
8r
Br
(1 Mark)
iv)
Bri
Br
(1 Mark)
V)
(1 Mark)
BKB/BKC/BKG/1213111BKF1323
CONFIDENTIAL
vi)
Br
CH
Me
(1 Mark)
b)
Draw structures corresponding to the following TUPAC names:
i) 5-bromo-3-propylhex- 1 -ene
(1 Mark)
ii) 5-chloro-4-methyl-2-hydroxy-hex-3-ene
(1 Mark)
iii) 4,4,4-trifluorobut- 1 -ene
(1 Mark)
iv) 2-methylbut-3 -enal
(1 Mark)
v) Toluene
(1 Mark)
C)
What is the correct oxidation-level ordering of the following compounds, staring
will lowest oxidation level?
A: CH30 B: (HO)2 C0
C: H2CO D: CH4
(2 Marks)
d)
Arrange the following phenols in order of increasing acidity (least to most).
BKB/BKC/BKG/121311/BKF 1323
CONFIDENTIAL
OH
OH
ct:,'
'NH2
NO2
A
OH
(2 Marks)
e)
Hydrocarbon A (C7H12) was treated with BH3 followed by H2 0 2INaOH to provide
B (C7H14 0) as the only product. Reaction of B with TsC1/pyridine followed by
KOH gives C (isomer with A) in addition to other olefin (s). Treatment of C with
ozone, followed by Zn/AcOH produces only the compound shown:
H3CY'^^^CHO
CHO
i)
What is the correct formula for A?
ii)
What is the correct formula for B?
iii)
What is the correct formula for C?
(6 Marks)
f)
Rank the following in order of stability (lowest to highest):
(4 Marks)
CONFIDENTIAL
BKB/BKC/BKG/121311/BKF1323
QUESTION 2
a) Draw the molecules structure of triethylamine and diisopropylamine.
(4 Marks)
b) Explain the increasing in the boiling point of the organic compounds below.
CH3 CH20CH2CH3
CH3CH2CH2CH2NH2
CH3CH2CH2CH20H
(4 Marks)
c) Using structure molecules, explain why tertiary amines (30) have lower boiling point
than 1 0 and 2 amines of comparable molecular weight.
(4 Marks)
d) If we compare an alkylamine and an arylamine, we must look at the availability of the
nonbonded electron pair on N. With CH3CH2NTTI2 for example, the electron pair is
localized on the N atom. Explain by describing the delocalized electron pair on the
benzene ring of an wylamine, and what is the implication to the chemical properties
of amine.
(8 Marks)
e) Give an explanation for Hofmann elimination by showing the steps to synthesis
propene from propylamine.
(5 Marks)
CONFIDENTIAL
BKBIBKCIBKG/1213111BKF1323
QUESTION 3
a) The reaction of an aldehyde or ketone with Grignard reagent, RMgX is a
nucleophilic addition to the carbon-oxygen double bond. Based on your
understanding, answer the following question
i).
Give the definition of nucleophile?
(2 Marks)
ii).
What is the function of Grignard reagent in this reaction?
(2 Marks)
iii).
What product is formed initially and product forms when water is added.
Show the mechanism to produce a both products.
(6 Marks)
b)
By using the suitable example, discuss about concept of inductive and resonance
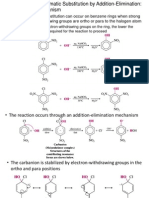
effect that effluent the location and rate of aromatic substitution reactions.
(6 Marks)
C)
Draw a stepwise mechanism for the reaction below
CH3
CH3
H2SO4
CH3
+ 1120
aCH3
(4 Marks)
d)
Write out the steps describing the mechanism of the free-radical bromination of
methane. Classify each step as initiation, propagation or termination.
(5 Marks)
BKB/BKCIBKG/1213111BKF1323
CONFIDENTIAL
QUESTION 4
a)
Draw the organic products formed in each reaction
1).
Cr03
H2SO4/H20
(D-,*,^
(2 Marks)
ii).
H2SO4
? + H20
+ CH30H
(2 Marks)
iii).
Cr03
H2SO41H20
? + ?
(4
Marks)
iv).
[1] cHI (excess)
0-
[2] Ag20
[3] Heat
(2 Marks)
b)
Answer the following questions.
i). Give curved arrow mechanisms that show how both of the products A and
B of the following reaction are formed. Indicate which is the major and
which the minor product.
7
BKB/BKC/BKG/1213111BKF1323
CONFIDENTIAL
Br
Ci+
HBr
(4 Marks)
ii).
Briefly explain why one of the products is major and the other minor.
(4 Marks)
c) Draw a detailed mechanism for the chlorination of benzene using C1 2 and FeCl3.
(4 Marks)
d) Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards
nucleophilic attack.
1CHO
C HO
cyclohexanecarbaldehyde
benzaldehyde
(3 Marks)
END OF QUESTION PAPER
Вам также может понравиться
- Chapter 1 - Introduction To MechanicsДокумент14 страницChapter 1 - Introduction To MechanicsrushdiОценок пока нет
- Friction: Vector Mechanics For Engineers: StaticsДокумент22 страницыFriction: Vector Mechanics For Engineers: StaticsrushdiОценок пока нет
- L4 - Preliminary Treatment Sept 2020 PDFДокумент20 страницL4 - Preliminary Treatment Sept 2020 PDFrushdiОценок пока нет
- Brochure Oilfield Corrosion Inhibitors GlobalДокумент5 страницBrochure Oilfield Corrosion Inhibitors GlobalAlfonso Dominguez Gonzalez100% (1)
- Topic 10 Organic Chemistry SL 2021Документ41 страницаTopic 10 Organic Chemistry SL 2021HotTornado XDОценок пока нет
- L5 - Physical TreatmentДокумент67 страницL5 - Physical Treatmentrushdi100% (1)
- OrganicДокумент3 страницыOrganickaifiiОценок пока нет
- Organic ChemistryДокумент3 страницыOrganic Chemistryawais gujjarОценок пока нет
- Organic ChemistryДокумент5 страницOrganic Chemistryapi-233187566Оценок пока нет
- Enoolate Chemistry ExcerciesДокумент15 страницEnoolate Chemistry ExcerciesClara CarreraОценок пока нет
- Sample Formal Report in Organic ChemistryДокумент10 страницSample Formal Report in Organic ChemistryAudrey CobankiatОценок пока нет
- Organic Chemistry PDFДокумент11 страницOrganic Chemistry PDFAli AyanОценок пока нет
- Organic Chemistry: Prepared By: Goce, Ivan Rei LДокумент35 страницOrganic Chemistry: Prepared By: Goce, Ivan Rei LReiVanОценок пока нет
- 15 - DNA Affinity Screening of Plants, BJC. v.27, n.2, 2010Документ4 страницы15 - DNA Affinity Screening of Plants, BJC. v.27, n.2, 2010Bolivian Journal of ChemistryОценок пока нет
- Execises - Organic Chemistry Chapter 2 Part 1Документ2 страницыExecises - Organic Chemistry Chapter 2 Part 1Michelle ChicaizaОценок пока нет
- Current+Organic+Chemistry +2008, 12,+1116-1183Документ96 страницCurrent+Organic+Chemistry +2008, 12,+1116-1183Murali Venkat NagОценок пока нет
- Anic Chemistry PDFДокумент30 страницAnic Chemistry PDFHakim Abbas Ali PhalasiyaОценок пока нет
- College of Arts and Sciences: CHM 215 Organic Chemistry IДокумент3 страницыCollege of Arts and Sciences: CHM 215 Organic Chemistry INajmul Puda PappadamОценок пока нет
- Worksheet: CaramelizationДокумент3 страницыWorksheet: CaramelizationLoreto T. Porcari JrОценок пока нет
- Substitution ProcessДокумент5 страницSubstitution ProcesselabagsОценок пока нет
- Recognizing Endo and Exo - Master Organic ChemistryДокумент9 страницRecognizing Endo and Exo - Master Organic ChemistryashishОценок пока нет
- Journal-1-Molecular Modeling in Organic ChemistryДокумент8 страницJournal-1-Molecular Modeling in Organic ChemistryRochelle Joyce Olmilla BersaminОценок пока нет
- Organic Chemistry - Some Basic Principles and TechniquesДокумент16 страницOrganic Chemistry - Some Basic Principles and TechniquesAbhayОценок пока нет
- Intro To Organic Chemistry PDFДокумент64 страницыIntro To Organic Chemistry PDFYuen Kim100% (1)
- 2018 01 Organic Chemistry Introductory AnalysisДокумент1 страница2018 01 Organic Chemistry Introductory Analysisapi-222503660Оценок пока нет
- General Organic Chemistry (GOC)Документ33 страницыGeneral Organic Chemistry (GOC)Jitendra Verma100% (1)
- Chemistry 6310 Advanced Topics in Organic Chemistry: The Organic ChemistryДокумент5 страницChemistry 6310 Advanced Topics in Organic Chemistry: The Organic Chemistryapi-20179616Оценок пока нет
- Organic ChemistryДокумент40 страницOrganic ChemistryGlenda ResultayОценок пока нет
- 07 Introduction To Organic ChemistryДокумент28 страниц07 Introduction To Organic ChemistryM BОценок пока нет
- Organic NomenclatureДокумент11 страницOrganic NomenclatureAmalia SillerОценок пока нет
- Organic Chemistry: Alkene NotesДокумент11 страницOrganic Chemistry: Alkene NotesDommie FranklinОценок пока нет
- Important Order and Facts of Organic ChemistryДокумент6 страницImportant Order and Facts of Organic ChemistryDEEPAK KUMAR MALLICKОценок пока нет
- Organic Chemistry LecturesДокумент32 страницыOrganic Chemistry LecturesAbdulHameedОценок пока нет
- ASSIGNMENT of Organic ChemistryДокумент8 страницASSIGNMENT of Organic ChemistryWania AliОценок пока нет
- Chapter 6 Reactions of Haloalkanes: S 2Документ8 страницChapter 6 Reactions of Haloalkanes: S 2Roberto SIlvaОценок пока нет
- Organic Chemistry - Pertemuan KeduapptДокумент60 страницOrganic Chemistry - Pertemuan Keduapptnadhilah shabrinaОценок пока нет
- Course Type Course Code Name of Course L T P Credit: Aromaticity: Introduction To Aromaticity, Anti-AromaticДокумент1 страницаCourse Type Course Code Name of Course L T P Credit: Aromaticity: Introduction To Aromaticity, Anti-AromaticOmkar KurlekarОценок пока нет
- Alkanes and Alkenes, Introduction To ORGANIC CHEMISTRYДокумент2 страницыAlkanes and Alkenes, Introduction To ORGANIC CHEMISTRYNayeemAhmedОценок пока нет
- Organic Chemistry: An Indian JournalДокумент5 страницOrganic Chemistry: An Indian Journalsnigdha shromaОценок пока нет
- s5 Organic Chemistry 30-03-20Документ208 страницs5 Organic Chemistry 30-03-20ONAP PATRICK JOSEPHОценок пока нет
- Organic ChemistryДокумент17 страницOrganic ChemistryMichael lIuОценок пока нет
- M.sc. - II, Organic ChemistryДокумент15 страницM.sc. - II, Organic ChemistryDeepak50% (2)
- Organic Chemistry 2 - Syllabus - USTHДокумент3 страницыOrganic Chemistry 2 - Syllabus - USTHMinh MinhОценок пока нет
- EAMCET QR Chemistry SR Chem 17.organic Chemistry Carbonyl CompoundsДокумент11 страницEAMCET QR Chemistry SR Chem 17.organic Chemistry Carbonyl CompoundsJagadeesh GoliОценок пока нет
- Organic Chemistry I-Edit PDFДокумент132 страницыOrganic Chemistry I-Edit PDFJeevitha SivamОценок пока нет
- Organic Chemistry Experiment 2Документ17 страницOrganic Chemistry Experiment 2Goh Chun KitОценок пока нет
- Summary Notes Organic ChemistryДокумент1 страницаSummary Notes Organic ChemistryVernonОценок пока нет
- CHEM1201-Lecture - 4A - Organic Chemistry Lecture NotesДокумент4 страницыCHEM1201-Lecture - 4A - Organic Chemistry Lecture NotesYoussef LatashОценок пока нет
- Organic Chemistry - Lesson 2Документ17 страницOrganic Chemistry - Lesson 2knlsinhaОценок пока нет
- Organic Chemistry TestДокумент1 страницаOrganic Chemistry Testron971Оценок пока нет
- Organic Chemistry 2Документ6 страницOrganic Chemistry 2mydreamcometrueОценок пока нет
- Organic Chemistry ExperimentДокумент7 страницOrganic Chemistry ExperimentKayla RhodesОценок пока нет
- Chemistry Assignment: Name: Asniza Binti Zul'AzmanДокумент5 страницChemistry Assignment: Name: Asniza Binti Zul'AzmanAsОценок пока нет
- Anatomy QuesДокумент7 страницAnatomy QuesShabab AliОценок пока нет
- Organic Chemistry Nucleophilic SubstitutДокумент1 страницаOrganic Chemistry Nucleophilic Substitut027 กัญญาภรณ์ ตันกลางОценок пока нет
- Organic Chemistry QuestionsДокумент2 страницыOrganic Chemistry QuestionsKevin Dacre100% (1)
- Practices Exam - Organic Chemistry To 2nd PartialДокумент10 страницPractices Exam - Organic Chemistry To 2nd PartialShary MosqueraОценок пока нет
- Compendium On Problems in Physical-Organic ChemistryДокумент27 страницCompendium On Problems in Physical-Organic ChemistrychemptnkОценок пока нет
- Organic ChemistryДокумент21 страницаOrganic ChemistryCHRISTINE JOY PASTURANОценок пока нет
- Definition of Organic ChemistryДокумент5 страницDefinition of Organic ChemistryBlinded ShipОценок пока нет
- Metalated Hetero Cycles and Their Applications in Synthetic Organic ChemistryДокумент56 страницMetalated Hetero Cycles and Their Applications in Synthetic Organic Chemistrygokay05Оценок пока нет
- CCH Anatomy SyllabusДокумент6 страницCCH Anatomy SyllabusNikhil SinghОценок пока нет
- Loudon Organic Chemistry Chapter 14Документ32 страницыLoudon Organic Chemistry Chapter 14JohnОценок пока нет
- Organic Chemistry of Biomolecules PDFДокумент3 страницыOrganic Chemistry of Biomolecules PDFMeghna KumarОценок пока нет
- CHM301 Apr 08Документ12 страницCHM301 Apr 08anon_35712225Оценок пока нет
- Organic Chem (With Solution) 2Документ75 страницOrganic Chem (With Solution) 2vlОценок пока нет
- Canadia EhcoДокумент18 страницCanadia EhcorushdiОценок пока нет
- Ehco Present and FutureДокумент5 страницEhco Present and FuturerushdiОценок пока нет
- Ehco Short Distance CanadianДокумент8 страницEhco Short Distance CanadianrushdiОценок пока нет
- Sludge DisposalДокумент53 страницыSludge DisposalrushdiОценок пока нет
- Competitive Dynamics: Dr. Satirenjit K JohlДокумент42 страницыCompetitive Dynamics: Dr. Satirenjit K JohlrushdiОценок пока нет
- A Review of Technologies For Transporting Heavy CRДокумент11 страницA Review of Technologies For Transporting Heavy CRrushdiОценок пока нет
- Lect6 - Total Site Composite Curves-SLIDESДокумент21 страницаLect6 - Total Site Composite Curves-SLIDESrushdiОценок пока нет
- Strategic Management Analysis: Case of Erzeni LTD: Epoka University, Albania, Akruja@epoka - Edu.alДокумент14 страницStrategic Management Analysis: Case of Erzeni LTD: Epoka University, Albania, Akruja@epoka - Edu.alrushdiОценок пока нет
- Lect5 - Steam Power BalancesДокумент27 страницLect5 - Steam Power Balancesrushdi100% (1)
- Lect8 - Optimising Steam levels-SLIDESДокумент30 страницLect8 - Optimising Steam levels-SLIDESrushdiОценок пока нет
- Lect7 - Cogeneration targets-SLIDESДокумент24 страницыLect7 - Cogeneration targets-SLIDESrushdiОценок пока нет
- L1-Introduction Environmental EmissionsДокумент30 страницL1-Introduction Environmental EmissionsrushdiОценок пока нет
- Research Philosophy and Overview - AP DR KamilДокумент85 страницResearch Philosophy and Overview - AP DR KamilrushdiОценок пока нет
- Lecture 2 - Steam Systems (Sep 2020) PDFДокумент8 страницLecture 2 - Steam Systems (Sep 2020) PDFrushdiОценок пока нет
- Lecture 1-Furnaces (Sep 2020) PDFДокумент12 страницLecture 1-Furnaces (Sep 2020) PDFrushdiОценок пока нет
- Lecture 4 - Gas Turbines (Sept 2020) PDFДокумент6 страницLecture 4 - Gas Turbines (Sept 2020) PDFrushdiОценок пока нет
- Bank Questions (CRE Chapter 2-3)Документ9 страницBank Questions (CRE Chapter 2-3)rushdiОценок пока нет
- Lecture 3 - Steam Turbines (Sept 2020) PDFДокумент9 страницLecture 3 - Steam Turbines (Sept 2020) PDFrushdiОценок пока нет
- Research Management DR Mohamed Hasnain IsaДокумент62 страницыResearch Management DR Mohamed Hasnain IsarushdiОценок пока нет
- Bank Questions Chapter 3-4Документ3 страницыBank Questions Chapter 3-4rushdiОценок пока нет
- Bank Questions Cre Chapter 7)Документ5 страницBank Questions Cre Chapter 7)rushdiОценок пока нет
- H2 Chemistry: Nitrogen CompoundsДокумент31 страницаH2 Chemistry: Nitrogen CompoundsPeng Jia XinОценок пока нет
- 56 TH Ipc AbstractДокумент2 страницы56 TH Ipc Abstractdinesh111180Оценок пока нет
- Vecom 0503 RustremoverДокумент1 страницаVecom 0503 RustremoverSPYROОценок пока нет
- Mock Test: About Disha PublicationДокумент23 страницыMock Test: About Disha PublicationkeerthyОценок пока нет
- Schiff Bases - A PrimerДокумент1 страницаSchiff Bases - A PrimerEric GrandallОценок пока нет
- Reaction of TryptophanДокумент2 страницыReaction of TryptophansasmithaОценок пока нет
- ALL Lesson 3m and 5 Marks 10m KALVISOLAIДокумент21 страницаALL Lesson 3m and 5 Marks 10m KALVISOLAISivaRajiОценок пока нет
- Trial Exam DCS Che 12 2022 Final (MA)Документ19 страницTrial Exam DCS Che 12 2022 Final (MA)navin chhetriОценок пока нет
- Photo Chemistry of Drugs An Overview and Practice Drugs Photo Chemistry and Photo Stability 1998-1-73 DOI 10 1039 9781847550712 00001Документ73 страницыPhoto Chemistry of Drugs An Overview and Practice Drugs Photo Chemistry and Photo Stability 1998-1-73 DOI 10 1039 9781847550712 00001mariaantoanetaОценок пока нет
- Determination of Plants Proteins Via The Kjeldahl Method and Amino Acid Analysis A Comparative StudyДокумент5 страницDetermination of Plants Proteins Via The Kjeldahl Method and Amino Acid Analysis A Comparative StudySisQha LuCiiajjaОценок пока нет
- Organic 2 Chapter 1&2Документ94 страницыOrganic 2 Chapter 1&2bahru demekeОценок пока нет
- The Systematic Identification of Organic CompoundsДокумент6 страницThe Systematic Identification of Organic CompoundsDwivelia Aftika0% (1)
- MBD Sure-Shot Chem MTPs Solved PDFДокумент34 страницыMBD Sure-Shot Chem MTPs Solved PDFjeet joshiОценок пока нет
- MnSb2O6 Chitosan Nanocomposite An Efficient Catalyst ForДокумент9 страницMnSb2O6 Chitosan Nanocomposite An Efficient Catalyst ForjorgeОценок пока нет
- Schiff Basiss and Their Derivatives PaperДокумент17 страницSchiff Basiss and Their Derivatives PaperSocietalОценок пока нет
- Determination of Protein Content SpectrophotometricallyДокумент10 страницDetermination of Protein Content SpectrophotometricallyTsabit AlbananiОценок пока нет
- Forensic: InternihnalДокумент16 страницForensic: InternihnalCorred Taz'ae100% (1)
- Bacterial Decolorization and Degradation of Azo DyesДокумент12 страницBacterial Decolorization and Degradation of Azo DyesFernanda Stuani PereiraОценок пока нет
- United States Patent (19) : Primary Examiner-Paul F. ShaverДокумент4 страницыUnited States Patent (19) : Primary Examiner-Paul F. ShaverTARIK VОценок пока нет
- Conchem ReviewerДокумент13 страницConchem ReviewerMelissa PasoquinОценок пока нет
- Coatosil Silquest Phy Prop InddДокумент4 страницыCoatosil Silquest Phy Prop InddTarkan AKÇAYОценок пока нет
- On The Origin of Free and Bound Staling Aldehydes in BeerДокумент24 страницыOn The Origin of Free and Bound Staling Aldehydes in BeerVal SelОценок пока нет
- Name Reactions Organic Chemistry NEET Chemistry Nitesh DevnaniДокумент88 страницName Reactions Organic Chemistry NEET Chemistry Nitesh DevnaniSNOB亥SHREDDERОценок пока нет
- Nucleophilic Aromatic SubstitutionДокумент18 страницNucleophilic Aromatic SubstitutionJolaine ValloОценок пока нет
- Chemistry HSSC-II - (3rd Set)Документ8 страницChemistry HSSC-II - (3rd Set)Isha KhanОценок пока нет
- Biochem ProteinДокумент40 страницBiochem ProteinCharlene SibugОценок пока нет
- Chapter 13 AminesДокумент32 страницыChapter 13 AminesStreamer XОценок пока нет
- Chapter 4 SUBSTITUTION REACTIONДокумент35 страницChapter 4 SUBSTITUTION REACTIONHalimatun MustafaОценок пока нет