Академический Документы
Профессиональный Документы
Культура Документы
Sodium Hydroxide
Загружено:
Srinivasan RajenderanИсходное описание:
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Sodium Hydroxide
Загружено:
Srinivasan RajenderanАвторское право:
Доступные форматы
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
Sodiumhydroxide
FromWikipedia,thefreeencyclopedia
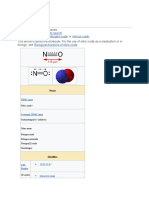
Sodiumhydroxide(NaOH),
Sodiumhydroxide
alsoknownaslyeandcaustic
soda,[1][2]isaninorganic
compound.Itisawhitesolid
and
Names
PreferredIUPACname
Sodiumhydroxide[3]
SystematicIUPACname
Sodiumoxidanide[4]
Othernames
Causticsoda
Lye[1][2]
Ascarite
Whitecaustic
Sodiumhydrate[3]
Identifiers
CASNumber
1310732
ChEBI
CHEBI:32145
ChemSpider
14114
ECNumber
2151855
GmelinReference
68430
Jmol3Dmodel
Interactiveimage
KEGG
D01169
MeSH
Sodium+Hydroxide
PubChem
14798
RTECSnumber
WB4900000
UNII
55X04QC32I
UNnumber
1823
InChI
[show]
SMILES
[show]
Properties
https://en.wikipedia.org/wiki/Sodium_hydroxide
1/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
Chemicalformula
NaOH
Molarmass
39.9971gmol1
Appearance
White,waxy,opaquecrystals
Odor
odorless
Density
2.13g/cm3
Meltingpoint
318C(604F591K)
Boilingpoint
1,388C(2,530F1,661K)
Solubilityinwater
418g/L(0C)
1110g/L(20C)
3370g/L(100C)
Solubility
solubleinglycerol
negligibleinammonia
insolubleinether
Solubilityinmethanol
238g/L
Solubilityinethanol
<<139g/L
Vaporpressure
<2.4kPa(at20C)
Basicity(pKb)
0.2[5]
Refractiveindex(nD)
1.3576
Thermochemistry
Specific
heatcapacity(C)
59.66J/molK
Stdmolar
entropy(So298)
64Jmol1K1[6]
Stdenthalpyof
formation(fHo298)
427kJmol1[6]
Gibbsfree
energy(fG)
380.7kJ/mol
Hazards
Safetydatasheet
ExternalMSDS
GHSpictograms
GHSsignalword
Danger
GHShazard
statements
H314
GHSprecautionary
statements
P280,P305+351+338,P310
EUclassification(DSD)
Rphrases
R35
Sphrases
(S1/2),S26,S37/39,S45
NFPA704
ALK
Lethaldoseorconcentration(LD,LC):
LD50(mediandose) 40mg/kg(mouse,
intraperitoneal)[8]
LDLo(lowest
published)
500mg/kg(rabbit,oral)[9]
UShealthexposurelimits(NIOSH):
PEL(Permissible)
TWA2mg/m3[7]
REL(Recommended) C2mg/m3[7]
https://en.wikipedia.org/wiki/Sodium_hydroxide
3[7]
2/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
IDLH(Immediate
danger)
10mg/m3[7]
Relatedcompounds
Otheranions
Sodiumhydrosulfide
Othercations
Caesiumhydroxide
Lithiumhydroxide
Potassiumhydroxide
Rubidiumhydroxide
Exceptwhereotherwisenoted,dataaregivenfor
materialsintheirstandardstate(at25C[77F],
100kPa).
verify(whatis
?)
Infoboxreferences
highlycausticmetallicbaseandalkaliofsodiumwhichisavailableinpellets,
flakes,granules,andaspreparedsolutionsatanumberofdifferent
concentrations.[10]Sodiumhydroxideformsanapproximately50%(by
weight)saturatedsolutionwithwater.[11]
Sodiumhydroxideissolubleinwater,ethanol,andmethanol.
Thisalkaliisdeliquescentandreadilyabsorbsmoistureandcarbon
dioxideinair.
Sodiumhydroxideisusedinmanyindustries,mostlyasa
strongchemicalbaseinthemanufactureofpulpandpaper,textiles,drinking
water,soapsanddetergentsandasadraincleaner.Worldwideproductionin
2004wasapproximately60milliontonnes,whiledemandwas51million
tonnes.[12]
Contents[hide]
1 Properties
1.1 Physicalproperties
1.2 Chemicalproperties
1.2.1 Reactionwithacids
1.2.2 Reactionwithacidicoxides
1.2.3 Reactionwithamphotericmetalsandoxides
1.2.4 Precipitant
1.2.5 Saponification
2 Production
3 Uses
3.1 Chemicalpulping
3.2 Tissuedigestion
3.3 Dissolvingamphotericmetalsandcompounds
3.4 Esterificationandtransesterificationreagent
3.5 Foodpreparation
3.6 Cleaningagent
3.7 Watertreatment
3.8 Historicaluses
3.9 Experimental
4 Safety
5 Commercialbrands
https://en.wikipedia.org/wiki/Sodium_hydroxide
3/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
6 Seealso
7 References
8 Bibliography
9 Externallinks
Properties
[ edit ]
Physicalproperties
[ edit ]
Puresodiumhydroxideisawhitishsolid,soldinpellets,flakes,andgranular
form,aswellasinsolution.Itishighlysolubleinwater,withalowersolubility
inethanolandmethanol,butisinsolubleinetherandothernonpolarsolvents.
Similartothehydrationofsulfuricacid,dissolutionofsolidsodiumhydroxidein
waterisahighlyexothermicreaction[why?]inwhichalargeamountofheatis
liberated,posingathreattosafetythroughthepossibilityofsplashing.The
resultingsolutionisusuallycolourlessandodorless.Aswithotheralkaline
solutions,itfeelsslipperywhenitcomesincontactwithskin.
Chemicalproperties
[ edit ]
Reactionwithacids [ edit ]
Sodiumhydroxidereactswithproticacidstoproducewaterandthe
correspondingsalts.Forexample,whensodiumhydroxidereacts
withhydrochloricacid,sodiumchlorideisformed:
NaOH(aq)+HCl(aq)NaCl(aq)+H2O(l)
Ingeneral,suchneutralizationreactionsarerepresentedbyonesimplenetionic
equation:
OH (aq)+H+ (aq)H2O(l)
Thistypeofreactionwithastrongacidreleasesheat,andhenceisexothermic.
Suchacidbasereactionscanalsobeusedfortitrations.However,sodium
hydroxideisnotusedasaprimarystandardbecauseitishygroscopicand
absorbscarbondioxidefromair.
Reactionwithacidicoxides [ edit ]
Sodiumhydroxidealsoreactswithacidicoxides,suchassulfurdioxide.Such
reactionsareoftenusedto"scrub"harmfulacidicgases(likeSO2andH2S)
producedintheburningofcoalandthuspreventtheirreleaseintothe
atmosphere.Forexample,
2NaOH+SO2Na2SO3+H2O
Reactionwithamphotericmetalsandoxides [ edit ]
Glassreactsslowlywithaqueoussodiumhydroxidesolutionsatambient
temperaturestoformsolublesilicates.Becauseofthis,glassjoints
andstopcocksexposedtosodiumhydroxidehaveatendencyto
"freeze".Flasksandglasslinedchemicalreactorsaredamagedbylong
exposuretohotsodiumhydroxide,whichalsofroststheglass.Sodium
https://en.wikipedia.org/wiki/Sodium_hydroxide
4/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
hydroxidedoesnotattackironsinceirondoesnothaveamphotericproperties
(i.e.,itonlydissolvesinacid,notbase).Afewtransitionmetals,however,may
reactvigorouslywithsodiumhydroxide.
In1986,analuminiumroadtankerintheUKwasmistakenlyusedtotransport
25%sodiumhydroxidesolution,[13]causingpressurizationofthecontentsand
damagetothetanker.Thepressurizationwasduetothehydrogengaswhichis
producedinthereactionbetweensodiumhydroxideandaluminium:
2Al+2NaOH+6H2O2NaAl(OH)4+3H2
Precipitant [ edit ]
Unlikesodiumhydroxide,thehydroxidesofmosttransitionmetalsareinsoluble,
andthereforesodiumhydroxidecanbeusedtoprecipitatetransitionmetal
hydroxides.Thefollowingcoloursareobserved:bluecopper,greeniron(II),
yellow/browniron(III).Zincandleadsaltsdissolveinexcesssodiumhydroxide
togiveaclearsolutionofNa2ZnO2orNa2PbO2.
Aluminiumhydroxideisusedasagelatinousflocculanttofilteroutparticulate
matterinwatertreatment.Aluminiumhydroxideispreparedatthetreatment
plantfromaluminiumsulfatebyreactingitwithsodiumhydroxideorcarbonate.
Al2(SO4)3+6NaOH2Al(OH)3+3Na2SO4
Al2(SO4)3+6NaHCO32Al(OH)3+3Na2SO4+6CO2
Saponification [ edit ]
Sodiumhydroxidecanbeusedforthebasedrivenhydrolysisofesters(as
insaponification),amidesandalkylhalides.However,thelimitedsolubilityof
sodiumhydroxideinorganicsolventsmeansthatthemoresolublepotassium
hydroxide(KOH)isoftenpreferred.Touchingsodiumhydroxidesolutionwiththe
barehands,whilenotrecommended,producesaslipperyfeeling.Thishappens
becausetheoilsofthehandareconvertedtosoap.
Production
[ edit ]
Sodiumhydroxideisindustriallyproducedasa50%solutionbyvariationsofthe
electrolyticchloralkaliprocess.Chlorinegasisalsoproducedinthisprocess.
Solidsodiumhydroxideisobtainedfromthissolutionbytheevaporationof
water.Solidsodiumhydroxideismostcommonlysoldasflakes,prills,andcast
blocks.[12]
In2004,worldproductionwasestimatedat60milliondrymetrictonnesof
sodiumhydroxide,anddemandwasestimatedat51milliontonnes.[12]In1998,
totalworldproductionwasaround45milliontonnes.NorthAmericaandAsia
eachcontributedaround14milliontonnes,whileEuropeproducedaround
10milliontonnes.IntheUnitedStates,themajorproducerofsodiumhydroxide
istheDowChemicalCompany,whichhasannualproductionaround
3.7milliontonnesfromsitesatFreeport,Texas,andPlaquemine,Louisiana.
OthermajorUSproducersincludeOxychem,PPG,Olin,PioneerCompanies,
Inc.(PIONA,whichwaspurchasedbyOlin),andFormosa.Allofthese
companiesusethechloralkaliprocess.[14]
https://en.wikipedia.org/wiki/Sodium_hydroxide
5/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
Historically,sodiumhydroxidewasproducedbytreatingsodium
carbonatewithcalciumhydroxideinametathesisreaction.(Sodiumhydroxideis
solublewhilecalciumcarbonateisnot.)Thisprocesswascalledcausticizing.[15]
Ca(OH)2(aq)+Na2CO3(s)CaCO3+2NaOH(aq)
ThisprocesswassupersededbytheSolvayprocessinthelate19thcentury,
whichwasinturnsupplantedbythechloralkaliprocesswhichweusetoday.
Sodiumhydroxideisalsoproducedbycombiningpuresodiummetalwithwater.
Thebyproductsarehydrogengasandheat,oftenresultinginaflame,making
thisacommondemonstrationofthereactivityofalkalimetalsinacademic
environmentshowever,itisnotcommerciallyviable,astheisolationofsodium
metalistypicallyperformedbyreductionorelectrolysisofsodiumcompounds
includingsodiumhydroxide.
Forfurtherinformationinhistoricalproduction,seealkalimanufacture.
Uses
[ edit ]
Sodiumhydroxideisapopular
strongbaseusedinindustry.Around
56%ofsodiumhydroxideproducedis
usedbyindustry,25%ofwhichisusedin
thepaperindustry.Sodiumhydroxideis
alsousedinthemanufactureofsodium
saltsanddetergents,pHregulation,and
organicsynthesis.ItisusedintheBayer
processofaluminiumproduction.[12]In
bulk,itismostoftenhandledas
anaqueoussolution,[16]sincesolutions
arecheaperandeasiertohandle.
Canisterofsodiumhydroxide.
Sodiumhydroxideisusedinmany
scenarioswhereitisdesirabletoincreasethealkalinityofamixture,orto
neutralizeacids.
Forexample,inthepetroleumindustry,sodiumhydroxideisusedasanadditive
indrillingmudtoincreasealkalinityinbentonitemudsystems,toincreasethe
mudviscosity,andtoneutralizeanyacidgas(suchashydrogen
sulfideandcarbondioxide)whichmaybeencounteredinthegeological
formationasdrillingprogresses.
Poorqualitycrudeoilcanbetreatedwithsodiumhydroxideto
removesulfurousimpuritiesinaprocessknownascausticwashing.Asabove,
sodiumhydroxidereactswithweakacidssuchashydrogensulfideand
mercaptanstoyieldnonvolatilesodiumsalts,whichcanberemoved.Thewaste
whichisformedistoxicanddifficulttodealwith,andtheprocessisbannedin
manycountriesbecauseofthis.In2006,Trafigurausedtheprocessand
thendumpedthewasteinAfrica.[17][18]
Seealso:Hydrodesulfurization
https://en.wikipedia.org/wiki/Sodium_hydroxide
6/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
Chemicalpulping
[ edit ]
Mainarticle:Pulp(paper)
Sodiumhydroxideisalsowidelyusedinpulpingofwoodformakingpaperor
regeneratedfibers.Alongwithsodiumsulfide,sodiumhydroxideisakey
componentofthewhiteliquorsolutionusedto
separateligninfromcellulosefibersinthekraftprocess.Italsoplaysakeyrole
inseverallaterstagesoftheprocessofbleachingthebrownpulpresultingfrom
thepulpingprocess.Thesestages
includeoxygendelignification,oxidativeextraction,andsimpleextraction,allof
whichrequireastrongalkalineenvironmentwithapH>10.5attheendofthe
stages.
Tissuedigestion
[ edit ]
Inasimilarfashion,sodiumhydroxideisusedtodigesttissues,asinaprocess
thatwasusedwithfarmanimalsatonetime.Thisprocessinvolvedplacinga
carcassintoasealedchamber,thenaddingamixtureofsodiumhydroxideand
water(whichbreaksthechemicalbondsthatkeepthefleshintact).This
eventuallyturnsthebodyintoaliquidwithcoffeelikeappearance,[19][20]andthe
onlysolidthatremainsarebonehulls,whichcouldbecrushedbetweenone's
fingertips.[21]Sodiumhydroxideisfrequentlyusedintheprocessof
decomposingroadkilldumpedinlandfillsbyanimaldisposalcontractors.[20]Due
toitslowcostandavailability,ithasbeenusedtodisposeofcorpsesby
criminals.ItalianserialkillerLeonardaCianciulliusedthischemicaltoturndead
bodiesintosoap.[22]InMexico,amanwhoworkedfordrugcartelsadmitted
disposingofover300bodieswithit.[23]Sodiumhydroxideisadangerous
chemicalduetoitsabilitytohydrolyzeprotein.Ifadilutesolutionisspilledonthe
skin,burnsmayresultiftheareaisnotwashedthoroughlyandforseveral
minuteswithrunningwater.Splashesintheeyecanbemoreseriousandcan
leadtoblindness.[24]Thisdangerisoftenoverlooked.
Dissolvingamphotericmetalsandcompounds
[ edit ]
Strongbasesattackaluminium.Sodiumhydroxidereactswithaluminiumand
watertoreleasehydrogengas.Thealuminiumtakestheoxygenatomfrom
sodiumhydroxide,whichinturntakestheoxygenatomfromthewater,and
releasesthetwohydrogenatoms,Thereactionthusproduceshydrogengas
andsodiumaluminate.Inthisreaction,sodiumhydroxideactsasanagentto
makethesolutionalkaline,whichaluminiumcandissolvein.Thisreactioncan
beusefulinetching,removinganodizing,orconvertingapolishedsurfacetoa
satinlikefinish,butwithoutfurtherpassivationsuchasanodizingoralodiningthe
surfacemaybecomedegraded,eitherundernormaluseorinsevere
atmosphericconditions.
IntheBayerprocess,sodiumhydroxideisusedintherefining
ofaluminacontainingores(bauxite)toproducealumina(aluminiumoxide)
whichistherawmaterialusedtoproducealuminiummetalvia
theelectrolyticHallHroultprocess.Sincethealuminaisamphoteric,itdissolves
https://en.wikipedia.org/wiki/Sodium_hydroxide
7/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
inthesodiumhydroxide,leavingimpuritieslesssolubleathighpHsuchasiron
oxidesbehindintheformofahighlyalkalineredmud.
Seealso:Ajkaaluminaplantaccident
Otheramphotericmetalsarezincandleadwhichdissolveinconcentrated
sodiumhydroxidesolutionstogivesodiumzincateandsodiumplumbate
respectively.
Esterificationandtransesterificationreagent
[ edit ]
Sodiumhydroxideistraditionallyusedinsoapmaking(cold
processsoap,saponification).[25]Itwasmadeinthenineteenthcenturyfora
hardsurfaceratherthanliquidproductbecauseitwaseasiertostoreand
transport.
Forthemanufactureofbiodiesel,sodiumhydroxideisusedasacatalystfor
thetransesterificationofmethanolandtriglycerides.Thisonlyworks
withanhydroussodiumhydroxide,becausecombinedwithwaterthefatwould
turnintosoap,whichwouldbetaintedwithmethanol.NaOHisusedmoreoften
thanpotassiumhydroxidebecauseitischeaperandasmallerquantityis
needed.
Sodiumhydroxideisalsobeingusedexperimentallyinanewtechnologyto
createsyntheticgasoline.[26]
Foodpreparation
[ edit ]
Foodusesofsodiumhydroxideincludewashingorchemicalpeeling
offruitsandvegetables,chocolateandcocoaprocessing,caramel
coloringproduction,poultryscalding,softdrinkprocessing,andthickeningice
cream.[27]Olivesareoftensoakedinsodiumhydroxidefor
softeningPretzelsandGermanlyerollsareglazedwithasodiumhydroxide
solutionbeforebakingtomakethemcrisp.Owingtothedifficultyinobtaining
foodgradesodiumhydroxideinsmallquantitiesforhomeuse,sodium
carbonateisoftenusedinplaceofsodiumhydroxide.[28]
Specificfoodsprocessedwithsodiumhydroxideinclude:
TheScandinaviandelicacyknownaslutefisk(fromlutfisk,"lyefish").
Hominyisdriedmaize(corn)kernelsreconstitutedbysoakinginlyewater.
Theseexpandconsiderablyinsizeandmaybefurtherprocessedbyfrying
tomakecornnutsorbydryingandgrindingtomakegrits.Nixtamalis
similar,butusescalciumhydroxideinsteadofsodiumhydroxide.
Sodiumhydroxideisalsothechemicalthatcausesgellingofeggwhitesin
theproductionofCenturyeggs.
Germanpretzelsarepoachedinaboilingsodiumcarbonatesolutionorcold
sodiumhydroxidesolutionbeforebaking,whichcontributestotheirunique
crust.
Lyewaterisanessentialingredientinthecrustofthetraditionalbaked
Chinesemooncakes.
MostyellowcolouredChinesenoodlesaremadewithlyewaterbutare
https://en.wikipedia.org/wiki/Sodium_hydroxide
8/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
commonlymistakenforcontainingegg.
Somemethodsofpreparingolivesinvolvesubjectingthemtoalyebased
brine.[29]
TheFilipinodessert(kakanin)calledkutsintausesasmallquantityoflye
watertohelpgivethericeflourbatterajellylikeconsistency.Asimilar
processisalsousedinthekakaninknownaspitsipitsiorpichipichiexcept
thatthemixtureusesgratedcassavainsteadofriceflour.
Cleaningagent
[ edit ]
Mainarticle:Cleaningagent
Sodiumhydroxideisfrequentlyusedasanindustrialcleaningagentwhereitis
oftencalled"caustic".Itisaddedtowater,heated,andthenusedtoclean
processequipment,storagetanks,etc.Itcan
dissolvegrease,oils,fatsandproteinbaseddeposits.Itisalsousedforcleaning
wastedischargepipesundersinksanddrainsindomestic
properties.Surfactantscanbeaddedtothesodiumhydroxidesolutioninorder
tostabilizedissolvedsubstancesandthuspreventredeposition.Asodium
hydroxidesoaksolutionisusedasapowerfuldegreaseronstainlesssteeland
glassbakeware.Itisalsoacommoningredientinovencleaners.
Acommonuseofsodiumhydroxideisintheproductionofparts
washerdetergents.Partswasherdetergentsbasedonsodiumhydroxideare
someofthemostaggressivepartswashercleaningchemicals.Thesodium
hydroxidebaseddetergentsincludesurfactants,rustinhibitorsanddefoamers.
Apartswasherheatswaterandthedetergentinaclosedcabinetandthen
spraystheheatedsodiumhydroxideandhotwateratpressureagainstdirty
partsfordegreasingapplications.Sodiumhydroxideusedinthismanner
replacedmanysolventbasedsystemsintheearly
1990s[citationneeded]whentrichloroethanewasoutlawedbytheMontrealProtocol.
Waterandsodiumhydroxidedetergentbasedpartswashersareconsideredto
beanenvironmentalimprovementoverthesolventbasedcleaningmethods.
Sodium
hydroxideis
usedinthe
homeasatype
ofdrain
openerto
Hardwarestoresgrade
sodiumhydroxidetobeused
asatypeofdraincleaner.
unblockclogged
drains,usuallyin
theformofadry
crystalorasa
thickliquidgel.
Thealkalidissolvesgreasestoproducewater
solubleproducts.It
alsohydrolyzestheproteinssuchasthosefound
Paintstrippingwithcaustic
soda
inhairwhichmayblockwaterpipes.These
reactionsarespedbytheheatgeneratedwhen
https://en.wikipedia.org/wiki/Sodium_hydroxide
9/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
sodiumhydroxideandtheotherchemicalcomponentsofthecleanerdissolvein
water.Suchalkalinedraincleanersandtheiracidicversionsare
highlycorrosiveandshouldbehandledwithgreatcaution.
Sodiumhydroxideisusedinsomerelaxerstostraightenhair.However,because
ofthehighincidenceandintensityofchemicalburns,manufacturersofchemical
relaxersuseotheralkalinechemicalsinpreparationsavailabletoaverage
consumers.Sodiumhydroxiderelaxersarestillavailable,buttheyareused
mostlybyprofessionals.
Asolutionofsodiumhydroxideinwaterwastraditionallyusedasthemost
commonpaintstripperonwoodenobjects.Itsusehasbecomelesscommon,
becauseitcandamagethewoodsurface,raisingthegrainandstainingthe
colour.
Watertreatment
[ edit ]
SodiumhydroxideissometimesusedduringwaterpurificationtoraisethepHof
watersupplies.IncreasedpHmakesthewaterlesscorrosivetoplumbingand
reducestheamountoflead,copperandothertoxicmetalsthatcandissolveinto
drinkingwater.[30][31]
Historicaluses
[ edit ]
Sodiumhydroxidehasbeenusedfordetectionofcarbonmonoxidepoisoning,
withbloodsamplesofsuchpatientsturningtoavermilioncoloruponthe
additionofafewdropsofsodiumhydroxide.[32]Today,carbonmonoxide
poisoningcanbedetectedbyCOoximetry.
Experimental
[ edit ]
Sodiumhydroxidetestforflavonoids
Safety
[ edit ]
Likeothercorrosiveacidsandalkalis,
dropsofsodiumhydroxidesolutionscan
readily
decomposeproteinsandlipidsinliving
tissuesviaamidehydrolysisandester
hydrolysis,whichconsequently
causechemicalburnsandmayinduce
permanentblindnessuponcontactwith
eyes.[1][2]Solidalkalicanalsoexpressits
corrosivenatureifthereiswater,such
aswatervapor.Thus,protective
equipment,likerubbergloves,safety
clothingandeyeprotection,should
alwaysbeusedwhenhandlingthis
chemicaloritssolutions.Thestandard
firstaidmeasuresforalkalispillsonthe
https://en.wikipedia.org/wiki/Sodium_hydroxide
Chemicalburnscausedbysodium
hydroxidesolutionphotographed44
hoursafterexposure.
10/11
7/26/2016
SodiumhydroxideWikipedia,thefreeencyclopedia
skinis,asforothercorrosives,irrigationwithlargequantitiesofwater.Washing
iscontinuedforatleasttentofifteenminutes.
Moreover,dissolutionofsodiumhydroxideishighlyexothermic,andtheresulting
heatmaycauseheatburnsorigniteflammables.Italsoproducesheatwhen
reactedwithacids.
Sodiumhydroxideiscorrosivetoseveralmetals,likealuminiumwhichreacts
withthealkalitoproduceflammablehydrogengasoncontact:[33]
2Al+6NaOH+3H2+2Na3AlO3
2Al+2NaOH+2H2O3H2+2NaAlO2
2Al+2NaOH+6H2O3H2+2NaAl(OH)4
Sodiumhydroxideisalsomildlycorrosivetoglass,whichcancausedamage
toglazingorfreezingofgroundglassjoints.Carefulstorageisneeded.
Commercialbrands
[ edit ]
RedDevilLye
LewisLye
Seealso
[ edit ]
HAZMATClass8CorrosiveSubstances
Commonchemicals
Listofcleaningagents
AcidandBase
References
[ edit ]
1.^ a b c
https://en.wikipedia.org/wiki/Sodium_hydroxide
11/11
Вам также может понравиться
- Sodium BicarbonateДокумент63 страницыSodium BicarbonateSridhar RaparthiОценок пока нет
- Hydrochloric AcidДокумент9 страницHydrochloric Acidvaradjoshi41Оценок пока нет
- Technical Report PDFДокумент184 страницыTechnical Report PDFSrinivasan RajenderanОценок пока нет
- FNCP, Rnking and Scaling, Home Visit PlanДокумент6 страницFNCP, Rnking and Scaling, Home Visit Planmarie100% (10)
- Introduction To Environmental Health 2012Документ66 страницIntroduction To Environmental Health 2012Mark Lopez100% (1)
- Sodium SilicateДокумент12 страницSodium Silicatemoses_cОценок пока нет
- Giz2011 en Exploring Biogas Market Opportunities VietnamДокумент92 страницыGiz2011 en Exploring Biogas Market Opportunities Vietnamlhphong021191Оценок пока нет
- 7.scrap Metal ShavingsДокумент1 страница7.scrap Metal ShavingsRakesh Ranjan MishraОценок пока нет
- Omron Industrial Automation Guide 2015Документ696 страницOmron Industrial Automation Guide 2015Omron Industrial Automation100% (2)
- Ba 88aДокумент77 страницBa 88aAnonymous GhWU5YK8Оценок пока нет
- Master's Thesis Evgeny Pribytkov 2013 PDFДокумент83 страницыMaster's Thesis Evgeny Pribytkov 2013 PDFSrinivasan RajenderanОценок пока нет
- Nortec 600Документ300 страницNortec 600JohnYuill100% (1)
- Ecological Profile 2014 PDFДокумент229 страницEcological Profile 2014 PDFTineОценок пока нет
- Sodium BicarbonateДокумент3 страницыSodium BicarbonateMecОценок пока нет
- 1Документ19 страниц1Rayadi Sinaga100% (1)
- Methanesulfonic AcidДокумент7 страницMethanesulfonic AcidJoseph Steven Evesa LlenaОценок пока нет
- Sodium Hydroxide - NaOH - PubChemДокумент57 страницSodium Hydroxide - NaOH - PubChemGemechis AsfawОценок пока нет
- From Wikipedia, The Free Encyclopedia Sodium HydroxideДокумент4 страницыFrom Wikipedia, The Free Encyclopedia Sodium Hydroxideanon_769688211Оценок пока нет
- Lithium Chloride: Jump To Navigation Jump To SearchДокумент11 страницLithium Chloride: Jump To Navigation Jump To SearchChaeyoung SonОценок пока нет
- Potassium Hydroxide - Wikipedia PDFДокумент42 страницыPotassium Hydroxide - Wikipedia PDFAmit KumarОценок пока нет
- Chemical NameДокумент49 страницChemical Namemanmadharathi342436Оценок пока нет
- Common & Chemical Names: Dr. Gaurav GargДокумент12 страницCommon & Chemical Names: Dr. Gaurav GargATUL SINGHОценок пока нет
- New Microsoft Word DocumentДокумент42 страницыNew Microsoft Word DocumentSalamat AliОценок пока нет
- Sulfuric AcidДокумент5 страницSulfuric AcidAnu ShanthanОценок пока нет
- Sodium Borohydride: Jump To Navigation Jump To SearchДокумент27 страницSodium Borohydride: Jump To Navigation Jump To SearchChaeyoung SonОценок пока нет
- Hydrochloric AcidДокумент9 страницHydrochloric AcidWaleed El-azabОценок пока нет
- Hypochlorous AcidДокумент99 страницHypochlorous Acidmahmoud IbrahimОценок пока нет
- Sodium Carbonate FAO PDFДокумент18 страницSodium Carbonate FAO PDFAnonymous ZON7qMОценок пока нет
- Borax: Jump To Navigation Jump To SearchДокумент4 страницыBorax: Jump To Navigation Jump To Searchelika.alfonsoОценок пока нет
- Reactions: Maleic Anhydride (Cis-Butenedioic Anhydride, Toxilic Anhydride, 2,5-Dioxofuran) Is AnДокумент3 страницыReactions: Maleic Anhydride (Cis-Butenedioic Anhydride, Toxilic Anhydride, 2,5-Dioxofuran) Is Anurukiora04Оценок пока нет
- Bahan NOДокумент18 страницBahan NOjujuОценок пока нет
- Nitric Oxide: Jump To Navigation Jump To SearchДокумент86 страницNitric Oxide: Jump To Navigation Jump To SearchChaeyoung SonОценок пока нет
- Common and Chemicals Names of Some Compounds - PDFДокумент3 страницыCommon and Chemicals Names of Some Compounds - PDFkvinodan73Оценок пока нет
- 10 Chemistry ABS 6Документ2 страницы10 Chemistry ABS 6Aryan GuptaОценок пока нет
- Aluminium Hydroxide OccurrenceДокумент8 страницAluminium Hydroxide OccurrenceFahad KhalidОценок пока нет
- Sodium BicarbonateДокумент11 страницSodium BicarbonatekgaviolaОценок пока нет
- Sắt (III) oxide-hydroxideДокумент5 страницSắt (III) oxide-hydroxidechithuan08052593Оценок пока нет
- General Chemistry 1Документ14 страницGeneral Chemistry 1killher gazeОценок пока нет
- Aerosil Cab-O-Sil Cab-O-Sil M-5P Wacker HDK: 1. Nonproprietary NamesДокумент6 страницAerosil Cab-O-Sil Cab-O-Sil M-5P Wacker HDK: 1. Nonproprietary NamesUma MaheswararaoОценок пока нет
- What Is A RadicalДокумент2 страницыWhat Is A RadicalDerrick Maatla MoadiОценок пока нет
- List of Common FormulasДокумент3 страницыList of Common FormulasJun YoutubeОценок пока нет
- Chloro AlkaliДокумент9 страницChloro AlkaliSk jahidul IslamОценок пока нет
- Sodium Chloride - WikipediaДокумент53 страницыSodium Chloride - WikipediadaribeefaОценок пока нет
- Chemical Name Handout Microteach 2 - RevisedДокумент2 страницыChemical Name Handout Microteach 2 - Revisedapi-287503448Оценок пока нет
- Try This!Документ5 страницTry This!Almudi TallingОценок пока нет
- Not To Be Confused With .: Sulfuric AcidДокумент7 страницNot To Be Confused With .: Sulfuric AcidAnu ShanthanОценок пока нет
- Calcium HypochloriteДокумент4 страницыCalcium HypochloriteStanleyEzeОценок пока нет
- ACIDS BASES SALTS TheoryДокумент19 страницACIDS BASES SALTS TheoryMr.UnknownОценок пока нет
- Water 1Документ7 страницWater 1Rey Jr. Cobel CababasadaОценок пока нет
- Acid Bases and Salts Previous Year Questiosn Class 10 Science (8 Files Merged)Документ47 страницAcid Bases and Salts Previous Year Questiosn Class 10 Science (8 Files Merged)satyabrata sahooОценок пока нет
- Properties of Potash AlumДокумент8 страницProperties of Potash AlumAminu TeslimОценок пока нет
- Acid, Base & Salts Level ++Документ4 страницыAcid, Base & Salts Level ++ayush.me.27Оценок пока нет
- Acid, Base & Salt - PPT OkДокумент30 страницAcid, Base & Salt - PPT OkLaksmita Zhafira DisaОценок пока нет
- Acid Bases and Salts Previous Year Questiosn Class 10 ScienceДокумент5 страницAcid Bases and Salts Previous Year Questiosn Class 10 Sciencesatyabrata sahooОценок пока нет
- US7405322Документ9 страницUS7405322Galang NinotОценок пока нет
- Maleinanhydrid ENG WIKIДокумент7 страницMaleinanhydrid ENG WIKIKrushnang JoshiОценок пока нет
- Third Term Chemistry SS1Документ75 страницThird Term Chemistry SS1Sunday Ozovehe100% (1)
- Oxalic Acid (Wiki)Документ5 страницOxalic Acid (Wiki)xcvОценок пока нет
- Worksheet On Is Matter Around Us PureДокумент3 страницыWorksheet On Is Matter Around Us PureSMKОценок пока нет
- Water: Jump To Navigation Jump To SearchДокумент6 страницWater: Jump To Navigation Jump To Searchkate trishaОценок пока нет
- Water: Jump To Navigation Jump To SearchДокумент6 страницWater: Jump To Navigation Jump To Searchkate trishaОценок пока нет
- Acids Bases & Salts: Short Answer Type Questions (L) (2 Marks) - Year 2015Документ14 страницAcids Bases & Salts: Short Answer Type Questions (L) (2 Marks) - Year 2015rajamohanОценок пока нет
- 5 AcidsДокумент8 страниц5 AcidsSARANGMUMBAIОценок пока нет
- Wikipedia Benzoic AcidДокумент11 страницWikipedia Benzoic Acidanon_822377657Оценок пока нет
- Chemistry Chapter 2 AssignmentДокумент2 страницыChemistry Chapter 2 Assignmentvihaang809Оценок пока нет
- Bty187 FINAL DRAFT - Compressed PDFДокумент23 страницыBty187 FINAL DRAFT - Compressed PDFGarima AnandОценок пока нет
- 10th Acid Base and Salt Notes 2011Документ5 страниц10th Acid Base and Salt Notes 2011Ashraf Husain100% (4)
- Iridium(III) in Optoelectronic and Photonics ApplicationsОт EverandIridium(III) in Optoelectronic and Photonics ApplicationsEli Zysman-ColmanОценок пока нет
- Omron Ladder Programming PDFДокумент16 страницOmron Ladder Programming PDFSrinivasan RajenderanОценок пока нет
- What Is A Solid State Relay?: Mechanical RelaysДокумент29 страницWhat Is A Solid State Relay?: Mechanical RelaysSrinivasan RajenderanОценок пока нет
- Dis13628 4ewithAnnexLДокумент104 страницыDis13628 4ewithAnnexLSrinivasan RajenderanОценок пока нет
- 20160711125732-Hochfeste Bleche Fuer Jackups e PDFДокумент4 страницы20160711125732-Hochfeste Bleche Fuer Jackups e PDFSrinivasan RajenderanОценок пока нет
- Another Development in Marine: Loading Arm TechnologyДокумент2 страницыAnother Development in Marine: Loading Arm TechnologySrinivasan RajenderanОценок пока нет
- Future Net Shape Manufacture Technology For Subsea ApplicationsДокумент26 страницFuture Net Shape Manufacture Technology For Subsea ApplicationsSrinivasan RajenderanОценок пока нет
- 2008 - Reference - Document Technip PDFДокумент244 страницы2008 - Reference - Document Technip PDFSrinivasan RajenderanОценок пока нет
- Design Data For Top Loading Arms: Info@kanon - NLДокумент1 страницаDesign Data For Top Loading Arms: Info@kanon - NLSrinivasan RajenderanОценок пока нет
- 062612DrillRig Posterads PDFДокумент1 страница062612DrillRig Posterads PDFSrinivasan RajenderanОценок пока нет
- Offshore Engineering Kavin Engneering Fpso Corporate Profile 2012Документ1 страницаOffshore Engineering Kavin Engneering Fpso Corporate Profile 2012Srinivasan RajenderanОценок пока нет
- Jean-Martial Carpena - LinkedInДокумент5 страницJean-Martial Carpena - LinkedInSrinivasan RajenderanОценок пока нет
- Mil PRF 24712aДокумент30 страницMil PRF 24712aAnonymous 5pURWfОценок пока нет
- Chapter 6 Research Methodology - Report WrittingДокумент69 страницChapter 6 Research Methodology - Report WrittingYared WendaleОценок пока нет
- Site Equipment List For TAG GokweДокумент8 страницSite Equipment List For TAG GokwetariroОценок пока нет
- Designing Business Solutions For Plastic Waste Management To Enhance Circular Transitions in KenyaДокумент20 страницDesigning Business Solutions For Plastic Waste Management To Enhance Circular Transitions in KenyaSadia IjazОценок пока нет
- Treadmill Plastic ShredderДокумент2 страницыTreadmill Plastic ShredderKyroОценок пока нет
- 051 Health and Safety Good Housekeeping 1Документ6 страниц051 Health and Safety Good Housekeeping 1api-415182300Оценок пока нет
- CV Sergio Duque Short VersionДокумент1 страницаCV Sergio Duque Short VersionratigerОценок пока нет
- Artline Clix WhiteboardДокумент5 страницArtline Clix WhiteboardMahesh Kumar SinghОценок пока нет
- Community Action PlanДокумент17 страницCommunity Action PlanAiko GerongОценок пока нет
- Serba Dinamik Capabilities in Power PlantДокумент11 страницSerba Dinamik Capabilities in Power PlantThiruvengadamОценок пока нет
- Esim264 ManualДокумент52 страницыEsim264 ManualScarlat MadalinОценок пока нет
- Internship Report Management Information SystemsДокумент112 страницInternship Report Management Information Systemshusnain_afzal8697700% (1)
- Cyrus Construction Development Group: Mr. & Mrs. RivasДокумент1 страницаCyrus Construction Development Group: Mr. & Mrs. RivasCyrus RivasОценок пока нет
- 3com AP 9552 - Guia de Acesso RápidoДокумент78 страниц3com AP 9552 - Guia de Acesso Rápidoivanilson_galdinoОценок пока нет
- 1310369102.14 Msds TdsДокумент5 страниц1310369102.14 Msds Tdsheru santosoОценок пока нет
- PX Pumps FolletoДокумент4 страницыPX Pumps FolletoBenicio Joaquín Ferrero BrebesОценок пока нет
- Eco Tourism Plan, Pench Tiger Reserve, MPДокумент26 страницEco Tourism Plan, Pench Tiger Reserve, MPRashi KothariОценок пока нет
- Indonesia Law On Water and Waster Water Quality No 82-2001Документ10 страницIndonesia Law On Water and Waster Water Quality No 82-2001Anonymous 6cQRWqОценок пока нет
- Please Note That This Is An Example of Our Rating Only. Please Contact An Austin Energy Green Building Representative To Find OutДокумент6 страницPlease Note That This Is An Example of Our Rating Only. Please Contact An Austin Energy Green Building Representative To Find OutM-NCPPCОценок пока нет
- Afety Data Sheet - EOS Fixer Part BДокумент5 страницAfety Data Sheet - EOS Fixer Part BMinamii NadiaОценок пока нет
- Virat Singh 7 C: St. Mary's Convent Inter College Manak Nagar, LucknowДокумент21 страницаVirat Singh 7 C: St. Mary's Convent Inter College Manak Nagar, LucknowShakilОценок пока нет
- NHDC Visit Report by Amit KumarДокумент49 страницNHDC Visit Report by Amit Kumaramit kumar67% (3)
- A2 3 11calculatingpropertydrainageanskeyДокумент8 страницA2 3 11calculatingpropertydrainageanskeyapi-247436935Оценок пока нет