Академический Документы
Профессиональный Документы
Культура Документы
UV-Vis Exercise 1 - Food Dye AnalysisTeacher Resource Pack - ENGLISH PDF
Загружено:
Kizzy Anne Boatswain CarbonОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
UV-Vis Exercise 1 - Food Dye AnalysisTeacher Resource Pack - ENGLISH PDF
Загружено:
Kizzy Anne Boatswain CarbonАвторское право:
Доступные форматы
1
Exercise 1
Food dye analysis
Introduction
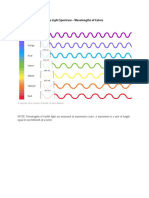
The electromagnetic spectrum ranges from radio waves
with wavelengths the size of buildings down to gamma rays,
the size of atomic nuclei. White light forms a small part of this
spectrum and is composed of a range of different wavelengths
which can be dispersed using a prism into its component
colours. The colour an object, or a solution, appears will depend
on which light is transmitted or reflected in the visible spectrum
and which light is absorbed. Using a UV-visible spectrometer
and a range of food dyes you will test how the absorbance
wavelength value relates to the colour of the solution.
UV-Visible Spectrometer
UV-visible spectrometers can be used to measure the absorbance of ultra violet or visible light by a sample.
The spectrum produced is a plot of absorbance versus wavelength (nm) in the UV and visible section of the
electromagnetic spectrum. Instruments can be used to measure at a single wavelength or perform a scan over
a range in the spectrum. The UV region ranges from 190 to 400 nm and the visible region from 400 to 800 nm.
The technique can be used both quantitatively and qualitatively.
Copyright 2009 Royal Society of Chemistry www.rsc.org
If a substance 450 nm
400 nm
800 nm - Visible Region
absorbs here...
Ultraviolet - Visible Spectroscopy (UV) Exercise 1 - Food dye analysis 2
Violet
Blue Red
Method 495 nm Visible Region Colour Wheel 620 nm
1. Prepare a dilute sample for each colour to be tested using 4. Use the colour wheel to predict absorbance values
a cuvette and distilled water (approximately 1 drop food for each solution and recordGreen
your predictions in the
Orange
colouring to 100 ml distilled water). table provided.
2. For each colour sample fill a plastic cuvette and stopper
Yellow
5. Set up the spectrometer to scan the visible region from ...it appe
with a lid. 350-800 nm and run each sample. Print out the spectrum as this c
and note the wavelength for570each
nm
of the absorbance 590
peaks.
nm
3. Prepare a blank sample cuvette containing distilled water
Compare these with your predictions.
only and stopper with a lid.
Red 620-750 nm
Orange 590-620 nm
UV Region Yellow 570-590 nm
Green 496-570 nm
Blue 450-495 nm
Violet 380-450 nm
190 nm 400 nm
If a substance 450 nm
400 nm
800 nm - Visible Region
absorbs here...
Violet
Blue Red
495 nm Visible Region Colour Wheel 620 nm
Green Orange
Yellow ...it appears
as this colour
570 nm 590 nm
Materials Red 620-750 nm
Orange 590-620 nm
Chemicals Instrument
Yellow 570-590 nm
Food colouring samples - Green 496-570 nmSpectrometer (integral printer and paper)
UV-visible
Red, yellow, green, blue, pink, black Laptop (optional)
De-ionised/distilled water Blue 450-495
Printer nm
(optional)
Apparatus
Violet 380-450 nm cables x 2 (optional)
Connection
Set up for laptop and printer use:
Disposable plastic cuvettes and stoppers
Wash bottles x 4 Connect UV-vis to laptop via left hand front
100 ml beakers x 10 USB port (Com 5)
1 box pasteur pipettes and teats Connect printer to any USB port
(Plastic for younger children) From spectrometer menu Select printer /
Tissues auto print on / Computer USB / OK
Open PVC program, set auto print to on or off depending
on requirements.
Copyright 2009 Royal Society of Chemistry www.rsc.org
Ultraviolet - Visible Spectroscopy (UV) Exercise 1 - Food dye analysis 3
Results
Colour Predicted Absorbance Actual Absorbance Notes
Value (nm) Value (nm)
Red 496 - 570 519 & 528 Absorbs Green
Yellow 380 - 450 428 Absorbs Violet
Green 620 - 750 427 & 635 Absorbs Red
Blue 590 - 620 409 & 628 Absorbs Orange
496 - 470 Absorbs possibly
Pink 510
570 - 590 Green/Yellow
Note: This absorbs both
in the Red and Green which
Black ? 519 & 635 are directly opposite, solution
appears black
Higher Lower
Frequency
Visible Spectrum Frequency
VIOLET BLUE GREEN YELLOW ORANGE RED
UV IR
400 500 600 700 800
wavelength (nm)
Copyright 2009 Royal Society of Chemistry www.rsc.org
Ultraviolet - Visible Spectroscopy (UV) Exercise 1 - Food dye analysis 4
Model Spectra
Red: 519 and 528 nm
Yellow: 428 nm
Copyright 2009 Royal Society of Chemistry www.rsc.org
Ultraviolet - Visible Spectroscopy (UV) Exercise 1 - Food dye analysis 5
Green: 635 nm
Blue: 628 nm
Copyright 2009 Royal Society of Chemistry www.rsc.org
Ultraviolet - Visible Spectroscopy (UV) Exercise 1 - Food dye analysis 6
Pink: 510 nm
Black: 519 and 635 nm
Copyright 2009 Royal Society of Chemistry www.rsc.org
Ultraviolet - Visible Spectroscopy (UV) Exercise 1 - Food dye analysis 7
Student Work Sheet
Colour Predicted Absorbance Actual Absorbance Notes
Value (nm) Value (nm)
Red
Yellow
Green
Blue
Pink
Black
Higher Lower
Frequency
Visible Spectrum Frequency
VIOLET BLUE GREEN YELLOW ORANGE RED
UV IR
400 500 600 700 800
wavelength (nm)
Copyright 2009 Royal Society of Chemistry www.rsc.org
Вам также может понравиться
- UV PosterДокумент1 страницаUV PosterYossuara Pitti100% (1)
- CS 2400 - 2500 BrochureДокумент8 страницCS 2400 - 2500 BrochureOo Kenx OoОценок пока нет
- Lambda Max KMnO4Документ9 страницLambda Max KMnO4Rayeha90% (10)
- Brochure Newton 7 0 in Vivo ImagingДокумент6 страницBrochure Newton 7 0 in Vivo ImagingPrasad KulkarniОценок пока нет
- 1993 - Schaffler - Rapid Analysis of SugarДокумент8 страниц1993 - Schaffler - Rapid Analysis of SugarOpal Priya WeningОценок пока нет
- Mass Spec Exercise 1 - Murder Mystery - Student Resource Pack - ENGLISHДокумент7 страницMass Spec Exercise 1 - Murder Mystery - Student Resource Pack - ENGLISHKizzy Anne Boatswain CarbonОценок пока нет
- Wavelengths of ColorsДокумент1 страницаWavelengths of ColorsSwas PrhrywnОценок пока нет
- Infrared LED Lights Product Lineup: Abundant Lineup, Total of 56 Models Available For Various ApplicationsДокумент12 страницInfrared LED Lights Product Lineup: Abundant Lineup, Total of 56 Models Available For Various ApplicationsKaiser MonterreyОценок пока нет
- Uv Vis Spectroscopy QuestionsДокумент6 страницUv Vis Spectroscopy QuestionsFahra Aqilla AzzurahОценок пока нет
- 6.UV Visible LatestДокумент61 страница6.UV Visible Latestdimitra shenoyОценок пока нет
- Application Notes: Application Note #1 Light - Some Physical BasicsДокумент13 страницApplication Notes: Application Note #1 Light - Some Physical BasicsBhagyashri Jog LimayeОценок пока нет
- MATTHEW NAZARRO - Chem 2208 Lab Experiment No. 8-Visible Spectrophotometry of Nickel (II) ChlorideДокумент7 страницMATTHEW NAZARRO - Chem 2208 Lab Experiment No. 8-Visible Spectrophotometry of Nickel (II) ChlorideMATTHEW NAZARROОценок пока нет
- Performance Task 5 - SPECTROPHOTOMETRYДокумент2 страницыPerformance Task 5 - SPECTROPHOTOMETRYKeith BagangОценок пока нет
- CC SpectrosДокумент1 страницаCC SpectrosAlexander PanodОценок пока нет
- 7 Spectrophotometry Loops - ManualДокумент5 страниц7 Spectrophotometry Loops - ManualJerry WangОценок пока нет
- Features: 1. The Highest Level of The High-Sensitivity in The Class: S/N 1200 (RMS) (S/N 360 Peak To Peak)Документ2 страницыFeatures: 1. The Highest Level of The High-Sensitivity in The Class: S/N 1200 (RMS) (S/N 360 Peak To Peak)PrianurraufikachmadОценок пока нет
- Alternate Light Source Imaging Forensic Photography TechniquesДокумент99 страницAlternate Light Source Imaging Forensic Photography Techniquesjadrio1556Оценок пока нет
- 3 CC1Документ6 страниц3 CC1marianОценок пока нет
- Fotometer: Foto: Cahaya Meter: PengukuranДокумент38 страницFotometer: Foto: Cahaya Meter: PengukuranRahmatika IntianiОценок пока нет
- CHM260 Lab Report SubmissionДокумент38 страницCHM260 Lab Report Submissionasta yuno100% (1)
- John Kunes - DL - Spectral LinesДокумент3 страницыJohn Kunes - DL - Spectral Linesapi-549570937Оценок пока нет
- 3 SpectrophotometryДокумент3 страницы3 SpectrophotometryRonald Angelo CironОценок пока нет
- Fotometer: Foto: Cahaya Meter: PengukuranДокумент38 страницFotometer: Foto: Cahaya Meter: PengukuranDiduunОценок пока нет
- Molecular SpectroДокумент11 страницMolecular SpectroNikita SharmaОценок пока нет
- 2019 NatureElec M. Holler Et Al. - Three-Dimensional Imaging of Integrated CircuitsДокумент7 страниц2019 NatureElec M. Holler Et Al. - Three-Dimensional Imaging of Integrated CircuitsMaximeОценок пока нет
- Pharmaceutical Standards Guide For UV-Vis SpectrophotometersДокумент8 страницPharmaceutical Standards Guide For UV-Vis SpectrophotometersGeorgescu MugurelОценок пока нет
- RA1611004010Документ27 страницRA1611004010Aarthi ShivaОценок пока нет
- Fundamentals of Optics Ebook FinalДокумент38 страницFundamentals of Optics Ebook FinalSuman RachaОценок пока нет
- Spectrophotometry: Calderon, Edna J. Britanico, Gyle Lynette P. Peralta, Angelo M. Sente, Thiesli Marc Y.Документ11 страницSpectrophotometry: Calderon, Edna J. Britanico, Gyle Lynette P. Peralta, Angelo M. Sente, Thiesli Marc Y.Lyn BritanicoОценок пока нет
- Siskomoptik: Ref: Berbagai SumberДокумент23 страницыSiskomoptik: Ref: Berbagai SumberFariОценок пока нет
- Blue Modern Company Profile PresentationДокумент24 страницыBlue Modern Company Profile PresentationPrasya Mauliya Anbar KhairaniОценок пока нет
- FP 8000E Presentation ABLE SmallДокумент99 страницFP 8000E Presentation ABLE SmallAliОценок пока нет
- Em Spectrum Drawing and WebquestДокумент8 страницEm Spectrum Drawing and Webquestgenaro.dapugОценок пока нет
- Publication 10 11891 250 UV-Vis2Документ12 страницPublication 10 11891 250 UV-Vis2abhinavОценок пока нет
- Clinical Chemistry Laboratory - SpectrophotometryДокумент2 страницыClinical Chemistry Laboratory - SpectrophotometrynikkiОценок пока нет
- Avantes ApplicationsДокумент24 страницыAvantes ApplicationsphmartОценок пока нет
- Schott Puravis Datasheet enДокумент6 страницSchott Puravis Datasheet enNicholas TanОценок пока нет
- vis-uvvis-spectroscopy-solutions-industrial-labs-FL52993 Genesys 30Документ2 страницыvis-uvvis-spectroscopy-solutions-industrial-labs-FL52993 Genesys 30ReneОценок пока нет
- Absorbance Rather Than Transmittance Is Most Useful In: SpectrophotometryДокумент3 страницыAbsorbance Rather Than Transmittance Is Most Useful In: SpectrophotometryDimple May Gianne DumaguitОценок пока нет
- Hellma BrochureДокумент6 страницHellma BrochureNeelima CherukupalliОценок пока нет
- Fluorescence Spectrophotometer LFS A10 and LFSДокумент4 страницыFluorescence Spectrophotometer LFS A10 and LFSlabtrondigitalОценок пока нет
- Paper For Upload PDFДокумент6 страницPaper For Upload PDFAnonymous uJpBMLqHОценок пока нет
- UV VIS SpectrosДокумент37 страницUV VIS SpectrosDiana JeronimoОценок пока нет
- Qc2 m1 Spectrometric Method of Analysis of Drug TransesДокумент7 страницQc2 m1 Spectrometric Method of Analysis of Drug TransesKrisel AguilarОценок пока нет
- Uv-V ApplicationДокумент42 страницыUv-V ApplicationTare ye TesfuОценок пока нет
- Calibration Spectrophotometer PDFДокумент29 страницCalibration Spectrophotometer PDFpiagiopersempreОценок пока нет
- Iub Pha404 Autumn 2022 AasДокумент40 страницIub Pha404 Autumn 2022 AasTanvir FahimОценок пока нет
- CHM260 Lab Report SubmissionДокумент37 страницCHM260 Lab Report Submissionasta yuno100% (1)
- 6 Dan 7. Bahan Kuliah GLP, SpektroДокумент30 страниц6 Dan 7. Bahan Kuliah GLP, SpektroFildzah HusnaОценок пока нет
- Tech Note 14Документ2 страницыTech Note 14meteohrОценок пока нет
- Validacion Uv ShimadzuДокумент2 страницыValidacion Uv Shimadzuchegue head hunterОценок пока нет
- DEOXYRIBOSE, RIBOSEPHOSPHATE, ATP AND DNA BY DIRECT (150-190 NM) AND FAR-UV (190-260 NM) REGIONS USING SYNCHROTRON RADIATION AS A LIGHT SOURCEДокумент4 страницыDEOXYRIBOSE, RIBOSEPHOSPHATE, ATP AND DNA BY DIRECT (150-190 NM) AND FAR-UV (190-260 NM) REGIONS USING SYNCHROTRON RADIATION AS A LIGHT SOURCE石子Оценок пока нет
- OM 19 Brochure STD 2009Документ2 страницыOM 19 Brochure STD 2009Borza DorinОценок пока нет
- INDEX FinalДокумент13 страницINDEX FinalGazi Mehedi HasanОценок пока нет
- F. UUVis - Colours in Organic CompoundsДокумент8 страницF. UUVis - Colours in Organic Compoundsharsheen kaurОценок пока нет
- WTW TurbidityДокумент6 страницWTW TurbidityIndra AditamaОценок пока нет
- Myco 2Документ19 страницMyco 2pecqueur.thomasОценок пока нет
- High-Performance Systems: Lambda UV/Vis/NIR and UV/Vis Spectrophotometers - 950, 850, and 650Документ8 страницHigh-Performance Systems: Lambda UV/Vis/NIR and UV/Vis Spectrophotometers - 950, 850, and 650primadanaОценок пока нет
- Quantization of Energy Lab Report: TitleДокумент5 страницQuantization of Energy Lab Report: TitleKaylee LambertОценок пока нет
- Application of Spectral Studies in Pharmaceutical Product development: (Basic Approach with Illustrated Examples) First Revised EditionОт EverandApplication of Spectral Studies in Pharmaceutical Product development: (Basic Approach with Illustrated Examples) First Revised EditionОценок пока нет
- Gemmological Instruments: Their Use and Principles of OperationОт EverandGemmological Instruments: Their Use and Principles of OperationРейтинг: 5 из 5 звезд5/5 (4)
- Form 1 Scheme of Work 2015-2016Документ7 страницForm 1 Scheme of Work 2015-2016Kizzy Anne Boatswain CarbonОценок пока нет
- BIO 11 Lab 02 2004 Enz. Copy - DoДокумент10 страницBIO 11 Lab 02 2004 Enz. Copy - DoKizzy Anne Boatswain CarbonОценок пока нет
- Bonding Structure and Periodicity Assessed HWДокумент17 страницBonding Structure and Periodicity Assessed HWKizzy Anne Boatswain CarbonОценок пока нет
- Notes On The MoleДокумент22 страницыNotes On The MoleKizzy Anne Boatswain CarbonОценок пока нет
- Bonding Structure and Periodicity Assessed HW MsДокумент10 страницBonding Structure and Periodicity Assessed HW MsKizzy Anne Boatswain CarbonОценок пока нет
- SHAPES OF MOLECULES AND IONS (Single Bonds Only) : Chemguide - QuestionsДокумент1 страницаSHAPES OF MOLECULES AND IONS (Single Bonds Only) : Chemguide - QuestionsDaneilla BanksОценок пока нет
- The Superheroes of The Atomic Model Activity Teacher InstructionsДокумент7 страницThe Superheroes of The Atomic Model Activity Teacher InstructionsKizzy Anne Boatswain CarbonОценок пока нет
- Organic Chemistry Delivery GuideДокумент43 страницыOrganic Chemistry Delivery GuideKizzy Anne Boatswain Carbon100% (1)
- Balancing Equations Teacher InstructionsДокумент7 страницBalancing Equations Teacher InstructionsKizzy Anne Boatswain CarbonОценок пока нет
- Mechanism FlashcardsДокумент8 страницMechanism FlashcardsKizzy Anne Boatswain CarbonОценок пока нет
- 5.4 NotesДокумент24 страницы5.4 Notesmuhajireen0% (1)
- Balancing Equations Teacher InstructionsДокумент7 страницBalancing Equations Teacher InstructionsKizzy Anne Boatswain CarbonОценок пока нет
- BIOL2161 Lec2 RE 2017Документ23 страницыBIOL2161 Lec2 RE 2017Kizzy Anne Boatswain CarbonОценок пока нет
- What Is This MoleculeДокумент2 страницыWhat Is This MoleculeKizzy Anne Boatswain CarbonОценок пока нет
- Exam Report 2015 To The Nation. UPDATED. 1Документ20 страницExam Report 2015 To The Nation. UPDATED. 1alixОценок пока нет
- Ecological Pyramids WorksheetДокумент4 страницыEcological Pyramids WorksheetTrilok SawantОценок пока нет
- BIOL 2163 Assignment 2Документ5 страницBIOL 2163 Assignment 2Kizzy Anne Boatswain CarbonОценок пока нет
- Balancing Chemical EquationsДокумент13 страницBalancing Chemical EquationsKizzy Anne Boatswain CarbonОценок пока нет
- Action Plan 1Документ2 страницыAction Plan 1Kizzy Anne Boatswain CarbonОценок пока нет
- 4.9 NotesДокумент17 страниц4.9 NotesKizzy Anne Boatswain CarbonОценок пока нет
- 4.2 TeacherДокумент8 страниц4.2 TeacherHardianty HamzahОценок пока нет
- 4.11 NotesДокумент24 страницы4.11 NotesKeri Gobin SamarooОценок пока нет
- CSEC Chemistry June 2009 P1Документ14 страницCSEC Chemistry June 2009 P1Kizzy Anne Boatswain Carbon83% (18)
- Balancing Equations Using Periodic Table Puzzle PiecesДокумент5 страницBalancing Equations Using Periodic Table Puzzle PiecesKizzy Anne Boatswain CarbonОценок пока нет
- 4.11 NotesДокумент24 страницы4.11 NotesKeri Gobin SamarooОценок пока нет
- Topic 4.10 Organic Synthesis and AnalysisДокумент12 страницTopic 4.10 Organic Synthesis and AnalysisPawat SilawattakunОценок пока нет
- Topic 1 NotesДокумент25 страницTopic 1 NotesKizzy Anne Boatswain CarbonОценок пока нет
- Topic 4.10 Organic Synthesis and AnalysisДокумент12 страницTopic 4.10 Organic Synthesis and AnalysisPawat SilawattakunОценок пока нет
- 1.4 NotesДокумент9 страниц1.4 NotesUmer SalmanОценок пока нет
- Mesoionic Compounds: An Unconventional Class of Aromatic HeterocyclesДокумент9 страницMesoionic Compounds: An Unconventional Class of Aromatic HeterocyclesEliton S. MedeirosОценок пока нет
- Gen Chem PDFДокумент34 страницыGen Chem PDFPaul VillasencioОценок пока нет
- Reversible Reactions and Dynamic EquilibriumДокумент6 страницReversible Reactions and Dynamic EquilibriumAhmet SofiОценок пока нет
- Sk-2.0-Fizik Topikal Answers F5 C2Документ4 страницыSk-2.0-Fizik Topikal Answers F5 C2Abang ZulhilmyОценок пока нет
- An SCD 374 Crystal Face Indexing For Numerical Absorption Correction DOC-A86-EXS001 WebДокумент2 страницыAn SCD 374 Crystal Face Indexing For Numerical Absorption Correction DOC-A86-EXS001 WebNinik Triayu SОценок пока нет
- Answer KEY of Nature of Light Online TestДокумент12 страницAnswer KEY of Nature of Light Online TestKhanОценок пока нет
- Detailed Lesson PlanДокумент5 страницDetailed Lesson PlanGspr BoJoyОценок пока нет
- Sist Iso 7150 1 1996Документ9 страницSist Iso 7150 1 1996y jasminОценок пока нет
- ACS Review 10 Conjugation in Alkadienes and Allylic SystemsДокумент16 страницACS Review 10 Conjugation in Alkadienes and Allylic SystemsRima MghamesОценок пока нет
- Release Coats Presentation PSTCДокумент26 страницRelease Coats Presentation PSTCShawn ZhengОценок пока нет
- E - Pfaudler Glass Linings 614 4EДокумент18 страницE - Pfaudler Glass Linings 614 4Edevang asherОценок пока нет
- 5990 4547enДокумент16 страниц5990 4547enHoanghanh LequangОценок пока нет
- Internship Report For 5th SemesterДокумент5 страницInternship Report For 5th SemesterBANANI DASОценок пока нет
- TPH and TOGДокумент14 страницTPH and TOGFriday Veronica FlorenciaОценок пока нет
- F2 IS Exam1 (04-05)Документ9 страницF2 IS Exam1 (04-05)羅天佑Оценок пока нет
- Chemical EquationДокумент3 страницыChemical EquationCHRISTIAN F. MAYUGAОценок пока нет
- SPOTIFYДокумент6 страницSPOTIFYAj AntonioОценок пока нет
- Advance Operation of SMR Using Gain Schedulede Model Predictive ControlДокумент17 страницAdvance Operation of SMR Using Gain Schedulede Model Predictive ControlQian Jun AngОценок пока нет
- DLL Science (Week 6)Документ5 страницDLL Science (Week 6)Dianne S. GarciaОценок пока нет
- Silt Density IndexДокумент11 страницSilt Density IndexWilfredo Suarez Torres100% (1)
- Sheldon 2017Документ27 страницSheldon 2017bluedolphin7Оценок пока нет
- WorkshopSolarRoofTop DonBoscoSumbaДокумент75 страницWorkshopSolarRoofTop DonBoscoSumbaDominggus TelupunОценок пока нет
- NSTC 13 SamplePaper 2014-16 XIДокумент24 страницыNSTC 13 SamplePaper 2014-16 XIAqeel Ali0% (1)
- Astronomy Chapter 1 NotesДокумент4 страницыAstronomy Chapter 1 NoteskrmooshОценок пока нет
- Scan MAP Tank Floors: MagazineДокумент52 страницыScan MAP Tank Floors: MagazineRabeh BàtenОценок пока нет
- Gate 2005 CyДокумент14 страницGate 2005 CySumanta- 14Оценок пока нет
- Article in Press: Journal of The European Ceramic SocietyДокумент8 страницArticle in Press: Journal of The European Ceramic SocietyAnca NegrilaОценок пока нет
- Biology: Osmosis and Diffusion Lab Using Potato Cores Class: 3B Mr. Boyer Name: Simon HanДокумент10 страницBiology: Osmosis and Diffusion Lab Using Potato Cores Class: 3B Mr. Boyer Name: Simon Han서연김Оценок пока нет
- OsmosisДокумент4 страницыOsmosisWalica DeokinananОценок пока нет
- Zero Hour Classes 2022 - 23 Odd SemДокумент6 страницZero Hour Classes 2022 - 23 Odd SemRAtnaОценок пока нет