Академический Документы
Профессиональный Документы
Культура Документы
Techniques of Tar Removal From Producer Gas - A Review
Загружено:
Hotib PerwiraОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Techniques of Tar Removal From Producer Gas - A Review
Загружено:
Hotib PerwiraАвторское право:
Доступные форматы
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
Techniques of Tar Removal from Producer
Gas A Review
Chiranjeevaraoseela1, VinodbabuCh2, VykuntaRao M3
Assistant professor, Department of Mechanical Engineering, GMRIT Rajam, India 1 2 3
ABSTRACT:Energy utilization is a basic measure of economic growth of any country, Most of the countries are
depending mainly on liquid fuels like petrol, diesel etc. ; Petroleum purely contains HC mixtures and carbon dioxide.
Perfect combustion is not possible with petroleum. It will lead to the generation of CO like emissions. Also at high
temperatures the formation of NOX takes place which may lead to the environmental pollution. Also, these fuels are
supposed to be depleted in course of time. So there is the need to search for alternative fuels. Producer gas is one of the
alternative fuels which can be obtained from the local available wastes by thermo-chemical biomass conversion
processes, especially gasification and pyrolysis.
Gasification is the conversion of biomass into a combustible gas mixture by the partial oxidation of biomass at higher
temperature typically in the range 800o900oC.The low calorific value (CV) gas produced (about 46 MJ/N m3) can
be burnt directly or used as a fuel for gas engines and gas turbines. The main constituents of the producer Gas are CH4,
CO, CO2, and other HCs. During gasification of biomass, however, a large amount of tar is also formed. Tar has all
organic contaminants with a molecular weight larger than benzene. Tar formation is one of the major problems to deal
with because of blocking and fouling process equipment such as pipes and valves in gas engine and turbines. Also the
presence of tar will impose serious limitations in the use of producer gas due to engine wear and high maintenance
costs. This work deals with various Tar removal techniques.
KEYWORDS:Bio-fuel, Producer gas, Tar removal.
I. INTRODUCTION
Biomass gasification is the thermo chemical conversion of biomass materials into a producer gas, which is a mixture of
carbon monoxide, carbon dioxide, methane, hydrogen, nitrogen and water vapour.
Fig 1: The process of gasification
The Fig 1 shows the process of gasificationGasification is a process of converting solid fuels into gaseous fuel. It is not
simply pyrolysis, the pyrolysis is only one of steps in conversion process. The other steps are combustion with air and
reduction of the product of combustion(water vapour and CO2) into combustible gases(carbon
monoxide ,hydrogen ,methane ,some other hydro carbons) and inert (carbon dioxide and nitrogen). The process leads
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 258
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
to the gas with some tine dust and condensable compounds termed tar, both of which must be restricted to low values if
the gas is to be used in internal combustion engines
1.1. BIOMASS RESOURCES:-
The biomass resources include organic materials obtainable from forest, agriculture, aquaculture and organic waste
residue from industrial and social activities.
Forests: Forests are source of fuel wood, charcoal and producer gas. Forest waste and residues from forest processing
plants can be used as biomass. Certain plants produce seeds to yield vegetable oils which can be used in biofuels
Agriculture residues: Straw, rice husk, groundnut shell, coconut shell and sugarcane bagasse are crop residues which
are the main biomass resources. The crop residues are generally gasified to obtain producer gas. The crop residues are
also converted into fuel pellets to be used as solid fuels.
Energy crops: Energy crops are the cultivation which provide raw materials for biofuels. These include sugar
plants to provide bioethanol, starch plants to produce bioethanol and oil producing plants to produce biodiesel.
Urban waste: Urban waste can be garbage or municipal solid waste and sewage or liquid waste. Garbage can be burnt
to obtain biomass energy while sewage has to processed to obtain biogas.
Aquatic plants : Certain aquatic or water plants are capable of growing extremely fast and supply organic raw materials
for producing biogas. The fast growing water plants include water hyacinth, seaweed algae and kelp.
1.2. TYPES OF GASIFIERS:
Conventionally, gasifier can be classified as :Fluidized bed gasifier and Fixed bed gasifier
1.2.1. FIXED BED GASIFIERS:
Fixed bed gasifires are classified depending upon the flow path of feedstock (biomass) and the generated gas (producer
gas) asUpdraft ,Down draft and Cross draft systems
Updraft gasifiers:
Fig2 : Updraught Gasifier
The start-up scheme would be built around a fire-on-grate over which the fuel bed rests; The Fig 2 shows, the
Updraught Gasifier the hottest zone is created over the grate because of the exothermic reaction of air with char and
biomass. This zone extends over a few particle depths through which the oxygen will get consumed. The products of
combustion will percolate through the bed, heating up the biomass till volatiles are expelled from it. These volatiles are
at temperatures of 300oc to 400oc. The gases with a mix of product gases and volatiles will move further in the bed,
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 259
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
transferring the heat to dry the biomass till both temperatures are nearly same. By proper feeding process, this
temperature can be maintained at 150oC to 200oC to ensure that the volatiles do not condense inside the reactor. The
final product gas is composed of significant moisture, volatiles and product gases. The volatiles may condense into a
viscous fluid.
II. PROBLEM DEFINITION
Gasification converts biomass into combustible gases, such as H2, CO, CH4 that can be used in boilers and in internal
combustion engine or turbine to produce electricity generation [1]. The table 2.1 shows Gas quality requirements for
power generation [2]
Constituent units IC engine Gas turbine
Particles mg/Nm3 < 50 <30
Particle size m <100 <5
Tar mg/Nm3 <10 n.d.*
Alkali metals mg/Nm n.d.* <0.24
Table 2.1. Gas quality requirements for power generation
During gasification of biomass, however, a large amount of tar is also formed. Tar has all organic contaminants with a
molecular weight larger than benzene [4] .Tar formation is one of the major problems to deal with because of blocking
and fouling process equipment such as pipes and valves in gas engine and turbines [3] [4]. Also the presence of tar will
impose serious limitations in the use of producer gas due to engine wear and high maintenance costs [5].
By far, tar removal is the most problematic. Thus the successful implementation of gasification technology for gas
engine/turbine based power projects or for fuel cell applications depends much on the effective and efficient
removal/conversion of tar from the producer gas.The aim of the research was to reduce tar content directly in the
gasifier and to produce a producer gas with a high caloric value
2.1. CONVERSION METHODOLOGIES:
By far, tar removal is the most problematic. Thus the successful implementation of gasification technology for gas
engine/turbine based power projects or for fuel cell applications depends much on the effective and efficient
removal/conversion of tar from the producer gas. Several approaches are presently being investigated as a solution to
address this problem.
Primary methods:
Tar formation results from the incomplete conversion of the liquid products in the gasification. This is caused by the
limitations on account of the physical or geometrical design of the gasifier coupled with the limitations in the chemical
reactivity [6]. Apart from overcoming the limitations in the gasifier design , various catalysts are being investigated to
enhance the chemical reactivity [7].
Secondary methods
One of the challenges related to primary measures is a particular design would apply to one type of a gasification fuel.
In secondary gas cleaning system, the gas contaminated with tars, particulates and other components acts as the input to
such a system. The gas cleaning systems should meet the requirements of the end-use or the downstream applications.
Removal of tars can be done by two approaches:
a) Physical removal [2]: The gas cleaning is mainly done by means of scrubbers, i.e. by condensing tars.
The aerosols formed are later removed by means of wet ESPs. Newer methods also proposes to recycle the scrubbed
tars in to the gasifier [8].
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 260
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
b) Chemical removal [9]: In this approach a chemical conversion of tars is achieved, i.e. by means of
thermal (by partial oxidation) or catalytic cracking (by dolomite, or nickel catalyst).
To extract thermal energy from the biomass following methods are available. Conversion of biomass to energy is
undertaken using two main process technologies:
A. Thermo-chemical
B. Bio-chemical / biological.
C. Mechanical extraction.
Within thermo-chemical conversion four process options are available:
i. combustion ii. gasification iii. Liquefaction and iv. pyrolysis
Combustion: The burning of biomass in air ,i.e. combustion, is used over a wide range of outputs to convert the
chemical energy stored in biomass into heat , mechanical power ,or electricity using various items of process
equipment ,e.g. stoves, furnaces, boilers, steam turbines, etc. Combustion of biomass produces hot gases at
temperatures around 80010000c. It is possible to burn any type of biomass but in practice combustion is feasible only
for biomass with a moisture content <50%, unless the biomass is pre dried.
Gasification: Gasification is the conversion of biomass into a combustible gas mixture by the partial oxidation of
biomass at higher temperature typically in the range 800o900oC.It is a combination of oxidative pyrolysis and hot-
char caused reduction reactions. The low calorific value (CV) gas produced (about 46 MJ/N m3) can be burnt directly
or used as a fuel for gas engines and gas turbines.
Liquefaction: Liquefaction is the conversion of biomass into a stable liquid hydrocarbon using low temperatures and
high hydrogen pressures. This process is more complex and more expensive than pyrolysis process.
Pyrolysis: Pyrolysis is a process in which the solid fuel will be converted at high temperatures to vapour and gases in
the absence of any oxidant. The output can be condensed to ambient temperature to lead to water and some complex
oxygenated hydrocarbons in the form of liquids and permanent gases like CO, H2, CO2 and CH4.There is also residual
solid in the form of char. The fraction of solids, liquids and gases will depend on the rate of temperature rise and actual
temperature to which it is raised. Typically, volatiles begin to evolve at about 300oc to 500oc depending on the actual
biomass considered. at a low end, drums built to carry petroleum tar and other crude oils are often used to produce char
by burning away the volatiles leading to 20% to 30% char.
Bio-chemical conversion encompasses two process:
i. Digestion (production of biogas, a mixture of mainly CH4 and CO2)
ii. Fermentation (production of ethanol)
Fermentation: It is used commercially on a large scale in various countries to produce ethanol from sugar crop sand
starch crops. The biomass is ground down and the starch converted by enzymes to sugars, with yeast then converting
the sugars to ethanol
Anaerobic digestion: AD is the conversion of organic material directly to a gas, termed biogas, a mixture of mainly
methane and carbon dioxide with small quantities of other gases such as hydrogen sulphide.
Producer gas can either be used in mono or dual fuel mode in reciprocating engines. In case of mono fuel mode of
operation, the gas is fuelled to a SI engine, whereas in the dual fuel mode it is operated along with small quantity of
liquid fuel in a compression ignition (CI)engine. The choice of mode of operation is entirely dictated by the economics
of the operation, and of course on availability of appropriate engines.
III.TAR REMOVAL TECHNIQUES
There are different methods to remove the tars from the producer gas. The following Fig 3 shows the some of them
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 261
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
Fig 3: The techniques of Tar removal
Cooling Towers and Venturi Scrubbers
Cooling/scrubbing towers are usually used after cyclones as the first wet scrubbing units. All heavy tar components
condense there. In venturi scrubber a throat section is built into the duct. As the gas enters the venturi throat, both gas
velocity and turbulence increase. The scrubbing liquid is sprayed into the gas stream before the gas encounters the
venturi throat, or in the throat, or upwards against the gas flow in the throat. The scrubbing liquid is then atomized into
small droplets by the turbulence in the throat and droplet-particle interaction is increased. After the throat section, the
mixture decelerates, and further impacts occur causing the droplets to agglomerate. Once the particles have been
captured by the liquid, the wetted particulate matter and excess liquid droplets are separated from the gas stream by an
entrainment section which usually consists of a cyclonic separator or mist eliminator [ 10 ]
Thermal Cracking of Tars
Thermal processes raise the temperature of the producer gas to the levels that crack the heavy aromatic tar species
into lighter and less problematic species, such as hydrogen, carbon monoxide and methane. For this process, it is
suggested that temperatures exceed 1000oC in order to reduce tars effectively[ 10 ]
Partial Oxidation
When oxygen is added selectively to different stages, such as in secondary zones of a pyrolysis-cracker reactor, tars can
be preferentially oxidized[ 10 ]
Catalytic Cracking of Tars
The research on catalytic, hot-gas cleanup has involved (a) incorporating or mixing the catalyst with the feed biomass
to achieve so-called catalytic gasification or pyrolysis; and (b) treatment of gasifier raw gas in a second bed or beds[10]
Syngas Sampling
The structure of the gas sampling process is shown in Fig 4.
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 262
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
Fig 4:The structure of the gas sampling process
Gas traveled through copper connection tubing to the filter assembly where the particulates are collected. At
temperatures around 250oC, most of the tar contained in the gas passed through the connection tubing to a series of
impinger bottles containing acetone in which it was dissolved.. The vacuum pump generated suction for the process
and the flow regulator controlled and measured the volume of gas sampled.
All condensed tar particles were washed from the transfer lines (e.g. copper tubing) using the high purity acetone and
combined with the tar collected in the impinger bottles. The mass of gravimetric tar was determined through solvent
evaporation. The results were calculated using the following equation:
Ct=
Where: C t = Concentration of tar in syngas (g/m3)
W t = Weight of tar (g)
V g = Volume of sampled gas (m3) [ 10]
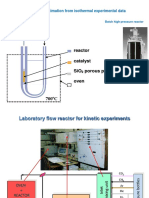
III. BIOMASS PYROLYSIS
A schematic representation of the experimental setup shown in fig 5. the feed stock was prepared from Japanese cedar,
crushed and sieved with a mesh size of 0.71-1.00mm. the feedstock was dried at a temperature of 1050c overnight in
an oven , before it was packed in the feeder, to remove moisture .
Fig 5 :Diagram of the experimental equipment
The proximate and ultimate analysis result of the feedstock are summarized. The feedstock was introduced into the
pyrolyzer by a controlled screw feeder with a Continuous feed rate of 0.6 g/min. the pyrolyzer reactor was made of
stainless steel with an inner diameter of 30 mm and length of 280 mm, and was surrounded by an electric heater . in
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 263
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
each experiment, the pyrolyzer was heated up to a temperature of 6000c and kept at this temperature for 30 min before
starting the feeder, in order to ensure that pyrolyzer tar production should be done under a steady state operation.
When the feedstock was introduced into the pyrolyzer, the feedstock released its volatiles in the form of syngas and
tar aerosols, which was carried out of the pyrolyzer by nitrogen, used as a carrier gas, and entered the gas cleaning
equipment with the flow rate of 1.51/min. the product 0f the pyrolysis process was sampled at the exit of the reformer,
which was kept at 4000c, in order to avoid tar decomposition or condensation.
4.1 TAR DECOMPOSITION:
Pyrolysis tar decomposition experiments under different reforming agents were carried out in an externally heated
reactor. The reactor called as the reformer was made of stainless steel with an inner diameter of 25mm and a length of
1300mm. The entire pyrolysis product in the form of gas was supplied directly in to the reformer in the tar
decomposition experiment. The effect of each reforming condition was investigated on the basis of the changing
gravimetric tar mass and concentration of tar compounds ,and their changes were compared with the result of the
pyrolysis condition. All experimental conditions of tar decomposition are summarized. In the studies of steam and air
reforming, water and fresh air were heated up to the temperature of reforming, before being supplied in to the reformer
for supporting the tar decomposition reaction. The parameters of steam and air reforming used to observe the effect on
tar decomposition are given by the steam to biomass(S/B)ratio and the equivalent ratio (ER),respectively, where the
S/B ratio is dened as the steam mass owrate divided by the feedstock mass owrate on the dry basis.while the the
ER is dened as the actual amount of air supplied to the reformer divided by the stoichiometric amount of air for
complete combustion of the feed.
4.2. TAR ADSORPTION:
Adsorption studies were carried out using a xed-bed type adsorbed, which was installed downstream of the reformer.
The entire reformer product was supplied directly into the adsorption bed through a high temperature resistance tube
connector. The reformed gas was cooled down to the temperature of 2530C by natural convection without any heat
exchanger device. Before starting the tar adsorption experiments there formed gas was by- passed from the adsorption
bed for 30min in order to ensure that the upstream reactions were under a steady-state condition.Wood chip, as the
same type of feedstock and synthetic porous cordierite were selected to study the adsorption ability of tar compound to
compare them with the adsorption ability of activated carbon, which has a good adsorption performance to adsorb
hydrocarbons. In case of wood chip, it was dried in a controlled oven at a temperature of 1050c Over night before using
as an adsorbent .The characteristics of each adsorbent are summarized.
Adsorbent characteristics
Adsorbent Activated carbon Wood chip Synthetic porous
Shape mean particle Granular 4 Chip 1.0- cordierite
Pellet
diameter(mm)
Volume of adsorbent (cm3) 200 1.5
200 200
Weight of adsorbent (g) 85.1 37.9 109.4
Advantage Good adsorption Low cost Re-usable
performance
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 264
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
Fig 5 : Tar removal
In the experiments activated carbon was applied to reduce tar components in producer gas. In experiments, the total
amount of tar in producer gas was reduced six fold when activated carbon was used in the upper reactor of the
gasifire( 11 )
The plant has two reactors made of STS316 are in series, which were heated indirectly with electricity and. The
bottom reactor was filled with silica sand and the upper reactor was filled with activated carbon for tarcracking. The
two reactors were separated by a distributor. Air, a fluidizing medium, was heated by a preheater to reduce heat loss
of bottom reactor and then entered in the bottom reactor. Immediately after the gasification, producer gas passes
through a char separation system consisting of a cyclone and a hot filter. After that liquid was collected condensers.
Finally, electrostatic precipitator (EP) captured particles and aerosols in producer gas.
IV. CONCLUSION
As the reactor temperature increased, H2 concentration strongly increased up to 16.6 vol.%. With respect to the
influence on equivalence ratio, it was found that increase in equivalence ratio led to the higher production of CO2,
Activated carbon played a significant role in tarcracking and H2 production. Total tar content consisting of tar on
apparatus and in condensate liquid reduced from 24 g to 4 g, when activated carbon is used. [11]
Liquid fuels like petrol, diesel etc, which are supposed to be depleted in course of time. So there is the need to search
for alternative fuels. biomass are cheap compared to the other alternative fuels and these are abundantly available.
Tar elimination efficiencies of the removal of tar decomposition combined with physical tar adsorption were
investigated by using a reformer as the first step and a xed bed absorber as the second step. Thermal tar
decomposition was carried out at a temperature of 8000c. High temperature had a strong inuence on tar decomposition.
removal of tar aerosols in producer gas may be block the engine.
The non-condensable tar could be combusted in the downstream combustion equipment, three different adsorbents
were conducted to study their performance of light tar removal. activated carbon showed the best adsorption
performance for both light aromatic hydrocarbon tar and light tar among other adsorbents.
As inert co2 we have to remove co2 from the producer gas from the mentioned techniques..
REFERENCES
[1] M. Dogru, A. Midilli, C. R. Howard, Fuel processing Technology, 2002, 75, 55-82.
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 265
ISSN(Online): 2319 - 8753
ISSN (Print) :2347 - 6710
International Journal of Innovative Research in Science,
Engineering and Technology
(An ISO 3297: 2007 Certified Organization)
Vol. 4, Issue 2, February 2015
[2] Hasler P., Nussbaumer Th., Gas cleaning requirements for IC engine applications fixed bed biomass gasification Biomass and Bioenergy,
16(1999) 385-395.
[3] J. J. Manya, J. L. Sanchez, J. Abrego, A. Gonzalo, J. Arauzo, Fuel ,2006, 85, 2027-2033.
[4] L. Devi, K. J, Ptasinski, F. J .J. G. Janssen, Biomass and Bioenergy, 2003, 24,125-140.
[5] Milne T.A., Evans R.J., Biomass Gasification tars: their nature, formation and conversion NREL, Golden, Colorado, USA. Report No.
NREL/TP-570-25357, 1998.
[6] Kersten S.R.A., Biomass gasification in circulating fluidized beds PhD Thesis, University of Twente, The Netherlands, 2002.
[7] Devi L., Ptasinski K.J., Janssen F.J.J.G., A review of the primary measures for tar elimination in biomass gasification process Biomass and
Bioenergy, 24(2003),125- 140.
[8] Bergman P.C.A., van Paasen S.V.B., Boerrigter H., The novel OLGA technology for complete tar removal from biomass producer gas
Paper presented at Pyrolysis and Gasification of Biomass and Waste, Expert Meeting, 30 September - 1 October 2002, Strasbourg, France.
[9] Simell P. Catalytic hot gas cleaning of gasification gas PhD Thesis, Helsinki University of technology, Finland, 1997.
[10] Christopher OsitaAkudo quantification of tars and particulates from a pilot scale, downdraft biomass gasifier A Thesis Submitted to
the Graduate Faculty of the Louisiana State University and Agricultural and Mechanical College in partial fulfillment of the requirements for the
degree of Master of Science in Biological and Agricultural Engineering in The Department of Biological and Agricultural Engineering May 2008
[11] Joo-Sik Kim*, Tae-Young Mun, Bo-Sung Kang Production of clean producer gas with high Heating from biomass by air gasification Using
two-stage gasifire The 5th ISFR (October 11-14, 2009, Chengdu, China)
[12] J. P. A. Neeft, H. A. M. Knoef, P. Onaji, Behaviour of tar in biomass gasification systems. Tar related problems and their solutions.
Novem. Report No.9919. Energy from Waste and Biomass(EWAB), The Netherlands, 1999..
Copyright to IJIRSET DOI: 10.15680/IJIRSET.2015.0402055 266
Вам также может понравиться
- Experiment Evolution For Downdraft Gasifier: With Using Various Biomass Wood, Bagasse and Coconut ShellДокумент5 страницExperiment Evolution For Downdraft Gasifier: With Using Various Biomass Wood, Bagasse and Coconut ShellMounicaRasagyaPallaОценок пока нет
- Use of Biomass Gas in Running Internal Combustion Engine To Generate Electricity-A ReviewДокумент7 страницUse of Biomass Gas in Running Internal Combustion Engine To Generate Electricity-A Reviewkashipuranataraj69Оценок пока нет
- Carbon Dioxide Capture, Sequestration, Compressor and Power CycleДокумент10 страницCarbon Dioxide Capture, Sequestration, Compressor and Power CycleIJRASETPublicationsОценок пока нет
- A Review and Discussion of Waste Tyre Pyrolysis AnДокумент8 страницA Review and Discussion of Waste Tyre Pyrolysis AnMarwan BОценок пока нет
- How To Improve Boiler EfficiencyДокумент6 страницHow To Improve Boiler EfficiencyPawan GoyalОценок пока нет
- A Review On Continuous Carbon Capturing Methods and UtilisationДокумент8 страницA Review On Continuous Carbon Capturing Methods and UtilisationBandiDevendraReddyОценок пока нет
- Plant Design and Economics ProjectДокумент43 страницыPlant Design and Economics Projectetayhailu100% (5)
- 64 AnalysisДокумент5 страниц64 AnalysisSaran CityОценок пока нет
- 1 s2.0 S0960852422018958 MainДокумент14 страниц1 s2.0 S0960852422018958 MainJuan Pablo Arteaga RamosОценок пока нет
- Waste To EnergyДокумент7 страницWaste To EnergyJohn Brown100% (1)
- Ncineration: Electricity Heat Waste Source Energy Recovery Methane Methanol EthanolДокумент8 страницNcineration: Electricity Heat Waste Source Energy Recovery Methane Methanol Ethanolanon_225411759Оценок пока нет
- Environmental Impacts: GasificationДокумент14 страницEnvironmental Impacts: GasificationSana RasheedОценок пока нет
- 45 ThermalДокумент18 страниц45 Thermalfreddy kurniawanОценок пока нет
- Industrial Emission Treatment Technologies: Manh Hoang and Anita J. HillДокумент22 страницыIndustrial Emission Treatment Technologies: Manh Hoang and Anita J. HillLaxit PanaraОценок пока нет
- Paper V2BI 2Документ5 страницPaper V2BI 2Jung Kyung WooОценок пока нет
- 2010 - Energy - Paper With Cover Page v2Документ8 страниц2010 - Energy - Paper With Cover Page v2Elhissin ElhissinnОценок пока нет
- Utilization of Co2 For EtoДокумент8 страницUtilization of Co2 For EtoMuhammad Dzikri AjauhariОценок пока нет
- Clean Heat and Power From Solid Fuels - Modern Approaches: CGPL, Indian Institute of Science, Bangalore 560 012, IndiaДокумент13 страницClean Heat and Power From Solid Fuels - Modern Approaches: CGPL, Indian Institute of Science, Bangalore 560 012, IndiaDhopesor hazarikaОценок пока нет
- Z-Tar 2Документ44 страницыZ-Tar 2tamil thendralОценок пока нет
- Exhaust Gas Recirculation System: October 2015Документ7 страницExhaust Gas Recirculation System: October 2015Luyen NguyenОценок пока нет
- 08 Chapter 2Документ37 страниц08 Chapter 2RomanОценок пока нет
- 240 Experimental NДокумент6 страниц240 Experimental NdhruvОценок пока нет
- Upgrade BiogasДокумент5 страницUpgrade BiogasHarsh GutgutiaОценок пока нет
- EnergyReports 1951 2021Документ15 страницEnergyReports 1951 2021ฉัตรประภา แวววงศ์Оценок пока нет
- WSN 492 2016 321 334 PDFДокумент14 страницWSN 492 2016 321 334 PDFSandeep DalviОценок пока нет
- Process Description Alphakat Update Dec 2010Документ20 страницProcess Description Alphakat Update Dec 2010Son Timur100% (2)
- Gasification: (Turnw2E™ Gasification Enhanced Gasification Performance)Документ2 страницыGasification: (Turnw2E™ Gasification Enhanced Gasification Performance)Arini Rizal100% (1)
- Overview of EnvironmentaДокумент6 страницOverview of EnvironmentaInternational Journal of Innovative Science and Research TechnologyОценок пока нет
- Nadeem - Performance Evaluation of An Industrial BoilerДокумент44 страницыNadeem - Performance Evaluation of An Industrial Boilervazzoleralex6884Оценок пока нет
- Unit 2 - Power Plant Engineering - WWW - Rgpvnotes.inДокумент19 страницUnit 2 - Power Plant Engineering - WWW - Rgpvnotes.inSarvjeet ThakreОценок пока нет
- Iarjset 2022 9440Документ10 страницIarjset 2022 9440RaviОценок пока нет
- 6 275 InfluenceДокумент7 страниц6 275 Influenceahamdanadam2413Оценок пока нет
- Thermochemical Biomass GasificationДокумент40 страницThermochemical Biomass Gasificationjambo98Оценок пока нет
- Clean Coal TechnologiesДокумент8 страницClean Coal TechnologiesArunChandraBabuОценок пока нет
- Gasification and IncinerationДокумент13 страницGasification and IncinerationPriyanshu ShekharОценок пока нет
- Circulating Fluidized Bed Reactor Design and TheoryДокумент28 страницCirculating Fluidized Bed Reactor Design and TheoryMehran Rasheed Goraya100% (3)
- Pers Cct2003Документ8 страницPers Cct2003VIJAYPORОценок пока нет
- Combustion and Environmental Performance of Clean Coal End ProductsДокумент14 страницCombustion and Environmental Performance of Clean Coal End ProductsΘΕΜΗΣ ΦΩΤΙΑΔΗΣОценок пока нет
- Environmental Impact of Sawdust Briquettes Use - Experimental ApproachДокумент6 страницEnvironmental Impact of Sawdust Briquettes Use - Experimental ApproachFood CatОценок пока нет
- Thermal Conversion Technologies For Solid WastesДокумент19 страницThermal Conversion Technologies For Solid Wastesraowaleedahmad100% (1)
- Vipppp4 BicakovaДокумент14 страницVipppp4 Bicakovahafeez khanОценок пока нет
- Lecture 3 Industrial Gases+ Ammonia 2019Документ49 страницLecture 3 Industrial Gases+ Ammonia 2019Mohamed AbdelaalОценок пока нет
- Coal Gasification Using AspenДокумент6 страницCoal Gasification Using AspenTaniadi Suria100% (1)
- 23-03-2021-1616495206-6-.-1. Engg - Production of A Producer Gas From Woody Biomass Using Co2 Capturing ChemicalsДокумент12 страниц23-03-2021-1616495206-6-.-1. Engg - Production of A Producer Gas From Woody Biomass Using Co2 Capturing ChemicalsImpact JournalsОценок пока нет
- Increasing IC engine efficiency using electrolysisДокумент5 страницIncreasing IC engine efficiency using electrolysisrassОценок пока нет
- Increasing Efficiency of Ic Engine by Electrolysis ProcessДокумент5 страницIncreasing Efficiency of Ic Engine by Electrolysis ProcessjishnuОценок пока нет
- Exhaust Gas Recirculation SystemДокумент7 страницExhaust Gas Recirculation Systemmalik visheshОценок пока нет
- Dry Ice Acetylene Power Plant (Dia Power Plant)Документ10 страницDry Ice Acetylene Power Plant (Dia Power Plant)IJRASETPublicationsОценок пока нет
- Industrial IncineratorДокумент16 страницIndustrial IncineratorAnthony FelixОценок пока нет
- Carbon Capture and Sequestration An OverviewДокумент7 страницCarbon Capture and Sequestration An OverviewIJRASETPublicationsОценок пока нет
- Puteri Calorifice GrasimiДокумент8 страницPuteri Calorifice GrasimieugenvitanОценок пока нет
- Emissions and Energy - Exergy Efficiency in An Industrial Boiler With Biodiesel and Other FuelsДокумент15 страницEmissions and Energy - Exergy Efficiency in An Industrial Boiler With Biodiesel and Other FuelsDaris BaihaqiОценок пока нет
- An Overview of Hydrogen Production From Biomass: Meng Ni, Dennis Y.C. Leung, Michael K.H. Leung, K. SumathyДокумент12 страницAn Overview of Hydrogen Production From Biomass: Meng Ni, Dennis Y.C. Leung, Michael K.H. Leung, K. SumathyNancy Eloisa RodriguezОценок пока нет
- Capture and Future of CO2 EmissionsДокумент63 страницыCapture and Future of CO2 EmissionsJlkm BnjlkmОценок пока нет
- Carbon Black ProcessДокумент4 страницыCarbon Black ProcessAnonymous azD9vQD100% (1)
- Syngas: 2.1 Steam Methane ReformingДокумент4 страницыSyngas: 2.1 Steam Methane ReformingthetreeoutthewindowОценок пока нет
- Clean Ironmaking and Steelmaking Processes: Efficient Technologies for Greenhouse Emissions AbatementОт EverandClean Ironmaking and Steelmaking Processes: Efficient Technologies for Greenhouse Emissions AbatementОценок пока нет
- Water Gas Shift Reaction: Research Developments and ApplicationsОт EverandWater Gas Shift Reaction: Research Developments and ApplicationsОценок пока нет
- Chapter 8Документ80 страницChapter 8Hotib PerwiraОценок пока нет
- Solubility and Complexation EquilibriaДокумент14 страницSolubility and Complexation EquilibriaHotib PerwiraОценок пока нет
- Energy balance on chemical reactorsДокумент41 страницаEnergy balance on chemical reactorsHotib PerwiraОценок пока нет
- Chapter 5Документ11 страницChapter 5Hotib PerwiraОценок пока нет
- Chapter 1Документ30 страницChapter 1Hotib PerwiraОценок пока нет
- Chapter 1Документ30 страницChapter 1Hotib PerwiraОценок пока нет
- Chapter 4Документ27 страницChapter 4Hotib PerwiraОценок пока нет
- Balances On Plug Flow Packed Bed Reactor (PFPBR)Документ11 страницBalances On Plug Flow Packed Bed Reactor (PFPBR)Hotib PerwiraОценок пока нет
- How To Approach A Meta-Analysis: DR Julie Cox, Consultant Radiologist, University Hospital of North DurhamДокумент3 страницыHow To Approach A Meta-Analysis: DR Julie Cox, Consultant Radiologist, University Hospital of North DurhamHotib PerwiraОценок пока нет
- Chapter 6.1Документ10 страницChapter 6.1Hotib PerwiraОценок пока нет
- Chapter 3Документ16 страницChapter 3Hotib PerwiraОценок пока нет
- 429518Документ11 страниц429518Hotib PerwiraОценок пока нет
- English VersionДокумент193 страницыEnglish VersionHotib PerwiraОценок пока нет
- GP6 - Stiegel060905Документ59 страницGP6 - Stiegel060905Hotib PerwiraОценок пока нет
- Biomass Gasification For Power Generation: A Comparison With Steam Boilers in The Brazilian ScenarioДокумент10 страницBiomass Gasification For Power Generation: A Comparison With Steam Boilers in The Brazilian ScenarioHotib PerwiraОценок пока нет
- Energy balance on chemical reactorsДокумент41 страницаEnergy balance on chemical reactorsHotib PerwiraОценок пока нет
- Air Treatment WaterleauДокумент28 страницAir Treatment WaterleauSankar CdmОценок пока нет
- Properties of Coal Classification and AnalysisДокумент7 страницProperties of Coal Classification and AnalysisJaco KotzeОценок пока нет
- Explorative Studies Performed in The Initial Stage of Research WorkДокумент7 страницExplorative Studies Performed in The Initial Stage of Research WorkHotib PerwiraОценок пока нет
- P450Документ102 страницыP450Hotib PerwiraОценок пока нет
- Im SWP L 002fДокумент42 страницыIm SWP L 002fHotib PerwiraОценок пока нет
- Dust Collection OverviewДокумент27 страницDust Collection Overviewmarn-in2501Оценок пока нет
- Ppact121 1Документ39 страницPpact121 1Hotib PerwiraОценок пока нет
- Power PlantДокумент108 страницPower PlantAmrita ChakrabortyОценок пока нет
- Permeance enДокумент12 страницPermeance enHotib PerwiraОценок пока нет
- Ipo 215061Документ8 страницIpo 215061Hotib PerwiraОценок пока нет
- 8098 pt1Документ385 страниц8098 pt1Hotib PerwiraОценок пока нет
- Teh Palm Residues FinalДокумент39 страницTeh Palm Residues FinalPutri Indra NovianiОценок пока нет
- Wet Electroscrubbers For State of The Art Gas Cleaning: Critical ReviewДокумент11 страницWet Electroscrubbers For State of The Art Gas Cleaning: Critical ReviewHotib PerwiraОценок пока нет
- International Handbook of Evidence-Based Coaching Theory, Research and Practice (Siegfried Greif, Heidi Möller, Wolfgang Scholl Etc.) (Z-Library)Документ964 страницыInternational Handbook of Evidence-Based Coaching Theory, Research and Practice (Siegfried Greif, Heidi Möller, Wolfgang Scholl Etc.) (Z-Library)Hoang100% (1)
- Faculity of Busines and Economics Department of Managment: The Role of Effective Communication in Resolving ConflictДокумент41 страницаFaculity of Busines and Economics Department of Managment: The Role of Effective Communication in Resolving ConflictMerry HiyabОценок пока нет
- A Record of Continental Collision and Regional SedДокумент15 страницA Record of Continental Collision and Regional SedAhmad SaepullahОценок пока нет
- Differences and Similarities Between Business Letters and MemorandumДокумент2 страницыDifferences and Similarities Between Business Letters and MemorandumSabeeh Pervaiz100% (1)
- Learning Guide-12: Masonry Level-IIIДокумент17 страницLearning Guide-12: Masonry Level-IIITewodros GeremewОценок пока нет
- Applying Social Cognitive Theory in Coaching Athletes: The Power of Positive Role ModelsДокумент8 страницApplying Social Cognitive Theory in Coaching Athletes: The Power of Positive Role ModelsSuppanadОценок пока нет
- Maxwell's Equations ExplainedДокумент33 страницыMaxwell's Equations ExplainedMekashaw AndargeОценок пока нет
- 2022ecology NeotropicalFishesDatapaperДокумент11 страниц2022ecology NeotropicalFishesDatapaperHelena ReisОценок пока нет
- Bruner TheoryДокумент4 страницыBruner TheoryTulika BansalОценок пока нет
- 5781-01 - Applus Canada MechanicalДокумент3 страницы5781-01 - Applus Canada MechanicalAdrewhassTechnicaОценок пока нет
- Reflection Journal Entry in ESPДокумент12 страницReflection Journal Entry in ESPDick Carlo Anzaldo TorresОценок пока нет
- Nodal's and Maxwell's Theorem Anore, James IvanДокумент16 страницNodal's and Maxwell's Theorem Anore, James IvanMeiko AbrinaОценок пока нет
- Strategies To Improve ListeningДокумент2 страницыStrategies To Improve ListeningCristine G. LagangОценок пока нет
- Analog CommunicationsДокумент2 страницыAnalog CommunicationsShanks1994Оценок пока нет
- 1953-07 IfДокумент126 страниц1953-07 Ifmoenobody100% (1)
- Tomography: Tomography Is Imaging by Sections or Sectioning Through The Use of AnyДокумент6 страницTomography: Tomography Is Imaging by Sections or Sectioning Through The Use of AnyJames FranklinОценок пока нет
- Cdha Literature Review GuidelinesДокумент5 страницCdha Literature Review Guidelinesaflsrxfoe100% (1)
- Daily Safety ReportДокумент51 страницаDaily Safety ReportFarhat SetharОценок пока нет
- MAN-125-0003-C (Pegasus Quick Installation Guide (With Internal Transducers) )Документ30 страницMAN-125-0003-C (Pegasus Quick Installation Guide (With Internal Transducers) )Jorge Caicedo Frv LfuОценок пока нет
- JSW Energy Limited - PestelДокумент23 страницыJSW Energy Limited - PestelDrupad KachiОценок пока нет
- Test Bank For Psychology of Gender 4th Edition HelgesonДокумент8 страницTest Bank For Psychology of Gender 4th Edition HelgesonchaferargeanjxfdОценок пока нет
- Greek MathemaДокумент6 страницGreek MathemaSebastian GhermanОценок пока нет
- DepEdClub English lessonsДокумент11 страницDepEdClub English lessonsElaine Joyce GarciaОценок пока нет
- Selection and Speciation NotesДокумент7 страницSelection and Speciation NotesLIM ZHI SHUENОценок пока нет
- 3rd GradeДокумент1 страница3rd Gradeapi-466941384Оценок пока нет
- EthaneДокумент116 страницEthaneNishanth GaddamОценок пока нет
- Pato Insomnia PDFДокумент14 страницPato Insomnia PDFdilaОценок пока нет
- Recap W5 L10: - Identify Various Event TypesДокумент28 страницRecap W5 L10: - Identify Various Event TypesSu FiОценок пока нет
- Critical Reanalysis of Strachey Prose WorksДокумент3 страницыCritical Reanalysis of Strachey Prose WorksPaz BalmacedaОценок пока нет
- Correlation and Regression: by Tushar BhattДокумент66 страницCorrelation and Regression: by Tushar Bhattchirag sabhaya100% (1)