Академический Документы
Профессиональный Документы
Культура Документы
PAT (Process Analytical Technology) For "LIQUID PARENTERAL" Pharmaceuticals I.E. SVP & LVP Manufacturing Line ...
Загружено:
Alfinovita KabanОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
PAT (Process Analytical Technology) For "LIQUID PARENTERAL" Pharmaceuticals I.E. SVP & LVP Manufacturing Line ...
Загружено:
Alfinovita KabanАвторское право:
Доступные форматы
See
discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/255171679
PAT (Process Analytical Technology) For “LIQUID PARENTERAL” Pharmaceuticals i.e. SVP & LVP Manufacturing Line ....
Article in Pharmaceutical Development and Technology · August 2013
CITATIONS READS
0 725
1 author:
Shivang Chaudhary
National Institute of Pharmaceutical Education and Research
59 PUBLICATIONS 74 CITATIONS
SEE PROFILE
Some of the authors of this publication are also working on these related projects:
Formulation R&D Engineer | Lean Quality by Design / Process Analytical Technology (F&D | Lean QbD / PAT) View project
All content following this page was uploaded by Shivang Chaudhary on 05 June 2014.
The user has requested enhancement of the downloaded file.
© Created & Copyrighted by Shivang Chaudhary
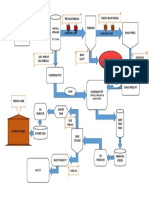
PROCESS ANALYTICAL TECHNOLOGY (PAT) FOR
LIQUID PARENTERAL PHARMACEUTICALS MANUFACTURING LINE
(shivaniper@gmail.com)
© Created & Copyrighted by Shivang Chaudhary
API / EXCIPIENT PURITY by STEAM FOR STERILIZATION WFI FOR CLEAN IN TEMPERATURE & RELATIVE
IN PLACE (SIP) PLACE (CIP) STEAM FOR STERILIZATION HUMIDITY by At Line
In line BRUKER FT-NIR WFI FOR CLEAN IN
STEAM FOR STERILIZATION WFI FOR CLEAN IN IN PLACE (SIP) Thermo-hygrometer
pH METER & PLACE (CIP)
IN PLACE (SIP) PLACE (CIP) DO METER NITROGEN
NITROGEN NITROGEN FLUSHING
API / EXCIPIENT PARTICLE FLUSHING FLUSHING DO METER
VACCUM & PRESSURE
SIZE DISTRIBUTION by DO METER PUM SYSTEM VACCUM & PRESSURE
In line Lasentec FBRM
COMPOUNDING PUM SYSTEM
VACCUM & PRESSURE VESSEL STORAGE
PUM SYSTEM VESSEL
STERILE
RATE OF STIRRING FOR COMPLETE
VACUUM WATER FOR INJECTION
WATER PHASE HOMOGENIZED STATE FOR SOLUTION
TRANSFER MANUFACTURING
HEATING & MIXING & by BRUKER FT-NIR
SYSTEM STORAGE
VESSEL VESSEL
WORKING
PLATFORM
RATE OF STERILIZATION by In RATE OF STERILIZATION by In
Line Chemical Indicator for Line Chemical Indicator for
Parametric Release Parametric Release
INTEGRATED FILTRATION FILTRATION
PYROGENS / ENDOTOXIN
CONTROL PANEL
RATE OF STERILIZATION by At ASSEMBLY & RATE OF CONTROLLED FLOCCULATION ASSEMBLY &
OR EFFECTIVE PRECIPITATION by In DETECTION by In Line
Line Bacteriological Indicator TRANSFER TRANSFER
Line Lasentec FBRM or PVM OR STERLIZATION PROCESS
for Parametric Release PUMPS PUMPS
At Line SVR/SHR Tests FOR PARAMETER S RECORDING
SUSPENSION OR EMULSIONS OR At Line LAL Test
LIQUID PARENTERAL (BFS) MANUFACTURING LINE WITH CONTROL STRATEGY &
PROCESS ANALYTICAL TECHNOLOGY
© Copyrighted by Shivang Chaudhary
View publication stats
Вам также может понравиться
- EC Tech Report High Performance Polyurethanes: PDFОт EverandEC Tech Report High Performance Polyurethanes: PDFVincentz Network GmbH & Co. KGОценок пока нет
- Liquid Parenteral SCДокумент3 страницыLiquid Parenteral SCKamran AlamОценок пока нет
- ETP Block Diagram. (23) PDFДокумент1 страницаETP Block Diagram. (23) PDFnetai biswasОценок пока нет
- Brochure - MBR 20 - 80 - HMIДокумент2 страницыBrochure - MBR 20 - 80 - HMIkoen irawanОценок пока нет
- Shredder Oil Lubrication and Cooling SystemДокумент1 страницаShredder Oil Lubrication and Cooling SystemFredrick OtienoОценок пока нет
- Spreading SopДокумент3 страницыSpreading SopTANYA TANYAОценок пока нет
- SK FormsДокумент1 страницаSK FormsJholly JumadayОценок пока нет
- QuickMist Automatic Nozzles LeafletДокумент6 страницQuickMist Automatic Nozzles LeafletDiana Carolina Rendon PeñaОценок пока нет
- Rector 2013Документ18 страницRector 2013Sutha Tamil NambeОценок пока нет
- Applications of Vacuum TechniqueДокумент1 страницаApplications of Vacuum Techniquesynayak100% (2)
- A Batch Centrifugal MachineДокумент1 страницаA Batch Centrifugal MachineFredrick OtienoОценок пока нет
- MixRite 2 5 Product Page 280615Документ2 страницыMixRite 2 5 Product Page 280615RicardoArturoPerezОценок пока нет
- 3 - NBC Form No - 77-001-S - Sanitary Plumbing PermitДокумент1 страница3 - NBC Form No - 77-001-S - Sanitary Plumbing Permitm0uth_wash1ngОценок пока нет
- Supplier:Neel Metal Products Ltd. (Faridabad) : Process FmeaДокумент1 страницаSupplier:Neel Metal Products Ltd. (Faridabad) : Process FmearohitОценок пока нет
- Presentation 1Документ6 страницPresentation 1Home Grown Digital AdvertisingОценок пока нет
- Sanitary Plumbing Permit 1Документ2 страницыSanitary Plumbing Permit 1jslumansocОценок пока нет
- Automatic Washer Study CourseДокумент20 страницAutomatic Washer Study CourseRichard WeimerОценок пока нет
- Sigma AutocoaterДокумент4 страницыSigma Autocoaterfad12345Оценок пока нет
- Supplier:Neel Metal Products Ltd. (Faridabad) : Process FmeaДокумент1 страницаSupplier:Neel Metal Products Ltd. (Faridabad) : Process FmearohitОценок пока нет
- Flowchart R-Pome WilmarДокумент1 страницаFlowchart R-Pome WilmarLaboratorium Bea Cukai DumaiОценок пока нет
- WBA Webinar - Dominique Kuehner, Andritz Feed and BiofuelДокумент27 страницWBA Webinar - Dominique Kuehner, Andritz Feed and BiofuelyascheОценок пока нет
- Manual Group 5 Fluid Flow IIДокумент54 страницыManual Group 5 Fluid Flow IISuciderОценок пока нет
- Primary Member Process Flow ChartДокумент3 страницыPrimary Member Process Flow Chartmadhu krishnaОценок пока нет
- Supplier Profile Snap Shot - Rev 2Документ3 страницыSupplier Profile Snap Shot - Rev 2Adnan QayumОценок пока нет
- Renr5846 00Документ2 страницыRenr5846 00Pauk HseinОценок пока нет
- F1 - Process Flow Chart Fabrication - BeamsДокумент2 страницыF1 - Process Flow Chart Fabrication - BeamssurwaneОценок пока нет
- Air Handling Units ListДокумент2 страницыAir Handling Units ListmiracОценок пока нет
- Eng DetailsДокумент2 страницыEng DetailsSchields PedroОценок пока нет
- Bread and Pastry Production Ncii Workshop Layout: Proofing CabinetДокумент1 страницаBread and Pastry Production Ncii Workshop Layout: Proofing CabinetCyvie Fate AntolinОценок пока нет
- Ammonium Sulfate ProductionДокумент2 страницыAmmonium Sulfate Productionsinggih0% (1)
- Sampl Eofweldi Ngprocedurespeci Fi Cati On (WPS) : Manual, Semi - Aut Omat I C, Aut Omat I C, Robot I CДокумент2 страницыSampl Eofweldi Ngprocedurespeci Fi Cati On (WPS) : Manual, Semi - Aut Omat I C, Aut Omat I C, Robot I CAshish PatelОценок пока нет
- Fulton Thermocompressor: 5875 Peachtree Industrial Blvd. Suite 330 Norcross, GA 30092Документ2 страницыFulton Thermocompressor: 5875 Peachtree Industrial Blvd. Suite 330 Norcross, GA 30092hrtoroОценок пока нет
- This Study Resource Was: Steam Nomo GraphДокумент2 страницыThis Study Resource Was: Steam Nomo GraphAdelaida CruzОценок пока нет
- Proline Promass 80: Description of Device FunctionsДокумент70 страницProline Promass 80: Description of Device FunctionsLuis Miguel Fernandez BejaranoОценок пока нет
- BONAVERA CHEM 200 FinalДокумент4 страницыBONAVERA CHEM 200 FinalAnurag VermaОценок пока нет
- Plumbing Permit: Office of The Building OfficialДокумент2 страницыPlumbing Permit: Office of The Building Officialhje421Оценок пока нет
- Balara - Mechanical Drawings - IFC PDFДокумент6 страницBalara - Mechanical Drawings - IFC PDFjomer john estoneloОценок пока нет
- E200b HidraulicДокумент2 страницыE200b HidraulicPaul Barraza67% (3)
- Hercuvan Mini CatalogДокумент11 страницHercuvan Mini Cataloglehieua8Оценок пока нет
- Piping & Instrument Diagram Symbols & Legends (Piping)Документ1 страницаPiping & Instrument Diagram Symbols & Legends (Piping)zalabiОценок пока нет
- U-Visc Kinematic Viscometer: ASTM D445 / D446, ISO 3104 / 3105, IP 71Документ2 страницыU-Visc Kinematic Viscometer: ASTM D445 / D446, ISO 3104 / 3105, IP 71moh_ichwanuddinОценок пока нет
- AD 22 FuelOilSystemsДокумент4 страницыAD 22 FuelOilSystemsCire Lop CastroОценок пока нет
- Complete Offerings: Thermal Processing Agglomeration Material HandlingДокумент8 страницComplete Offerings: Thermal Processing Agglomeration Material HandlingjuanalbertohaОценок пока нет
- Scematic Cooling 777E KYDДокумент2 страницыScematic Cooling 777E KYDAgung WicaksonoОценок пока нет
- Senr1678senr1678 Sis PDFДокумент2 страницыSenr1678senr1678 Sis PDFDennis OlayaОценок пока нет
- Training Manual For MerchandisersДокумент15 страницTraining Manual For MerchandisersDhanwaria67% (3)
- BAND 675 North RD - Mechanical IFC - 2023-11-15Документ162 страницыBAND 675 North RD - Mechanical IFC - 2023-11-15Hakar Qadir GardiОценок пока нет
- Ache Manufacturing Process - Pembangunan Mini LPG Plant 2.2 MMSCFDДокумент1 страницаAche Manufacturing Process - Pembangunan Mini LPG Plant 2.2 MMSCFDbayu kusumaОценок пока нет
- Maintenance Prescription: Analysis Integrated With Accuracy and MeaningДокумент1 страницаMaintenance Prescription: Analysis Integrated With Accuracy and MeaningVic AnaboОценок пока нет
- Seal & Seat Matrix-Floating-LatestДокумент5 страницSeal & Seat Matrix-Floating-LatestdennykvgОценок пока нет
- Inspection Request Roughing InsДокумент1 страницаInspection Request Roughing InsJhuzel colisОценок пока нет
- W2P Product RangeДокумент1 страницаW2P Product RangeAngirishОценок пока нет
- Engineering & Installation ManualДокумент61 страницаEngineering & Installation ManualThameem AnsariОценок пока нет
- Bonavera Chem 200 MachineДокумент4 страницыBonavera Chem 200 MachineAjish joОценок пока нет
- Accurate Feeding and Conveying. Vital Ingredients To Any RecipeДокумент20 страницAccurate Feeding and Conveying. Vital Ingredients To Any RecipeMubeen NavazОценок пока нет
- Balancing Kit-HVAC4Документ8 страницBalancing Kit-HVAC4Chaminda KumaraОценок пока нет
- Seacom Skills University B.Tech Course Laboratory Details (CIVIL Dept.)Документ3 страницыSeacom Skills University B.Tech Course Laboratory Details (CIVIL Dept.)RAVI BARTIAОценок пока нет
- Production of Pharmaceutical Syrups: Solid Ingredients Dosing Systems Production TanksДокумент2 страницыProduction of Pharmaceutical Syrups: Solid Ingredients Dosing Systems Production Tankshenrykayode4Оценок пока нет
- Erich Segal Doctors PDFДокумент2 страницыErich Segal Doctors PDFAlicia13% (8)
- Intermed Products: International CatalogueДокумент12 страницIntermed Products: International CatalogueRicardo Bonetti TadenОценок пока нет
- BS9266 2013 Adaptable HousingДокумент46 страницBS9266 2013 Adaptable HousingGrant MastersonОценок пока нет
- Daftar Isian 3 Number Plate, Danger Plate, Anti Climbing DeviceДокумент2 страницыDaftar Isian 3 Number Plate, Danger Plate, Anti Climbing DeviceMochammad Fauzian RafsyanzaniОценок пока нет
- Legislation Statutory Inspection ChecklistДокумент2 страницыLegislation Statutory Inspection ChecklistAry PutraОценок пока нет
- SuperboltДокумент32 страницыSuperboltRajeev Chandel100% (1)
- Komatsu Wheel Loaders Wa250pz 5 Shop ManualДокумент20 страницKomatsu Wheel Loaders Wa250pz 5 Shop Manualmarcia100% (48)
- Manual of Avionics PDFДокумент300 страницManual of Avionics PDFJhony BhatОценок пока нет
- MP65557-1 Manual Técnico - 4900Документ371 страницаMP65557-1 Manual Técnico - 4900Tecnico IncoderОценок пока нет
- CCR Control System Interface HandbookДокумент17 страницCCR Control System Interface Handbooklcastrey23Оценок пока нет
- Finance and RiskДокумент34 страницыFinance and RiskBlen tesfayeОценок пока нет
- Synchronous Motor AC DrivesДокумент40 страницSynchronous Motor AC DrivesHarshal Thakur100% (1)
- Pre Delivery Inspection Checklist For Home Oxygen ConcentratorДокумент2 страницыPre Delivery Inspection Checklist For Home Oxygen ConcentratorPranavKaisthaОценок пока нет
- A Guide To Become A Succesful Marine EngineerДокумент99 страницA Guide To Become A Succesful Marine EngineerTammam Hassan100% (1)
- Parts Catalog MAXSYM400iДокумент70 страницParts Catalog MAXSYM400iAntonio CoelhoОценок пока нет
- The Child and Adolescent Learners and Learning PrinciplesДокумент8 страницThe Child and Adolescent Learners and Learning PrinciplesAragon KhailОценок пока нет
- A Practical Guide To HL7 Interface DevelopmentДокумент5 страницA Practical Guide To HL7 Interface DevelopmentmjohnstnОценок пока нет
- Extenso MeterДокумент8 страницExtenso MeterVijayanandh Raja100% (1)
- Shallow FoundationsДокумент44 страницыShallow FoundationsAjay SinghОценок пока нет
- BITS Pilani: Scale Up and Post Approval ChangesДокумент56 страницBITS Pilani: Scale Up and Post Approval ChangesshrikantmsdОценок пока нет
- Land SurveyingДокумент174 страницыLand SurveyingArifulmakrif Hailee100% (2)
- Module 4 - Community Health AssessmentДокумент8 страницModule 4 - Community Health AssessmentSteffi100% (1)
- ReflectionДокумент1 страницаReflectionHeaven GuzmanОценок пока нет
- Aesa Vs PesaДокумент30 страницAesa Vs Pesakab11512100% (1)
- Fragility Curves For Mixed Concrete-Steel Frames Subjected To SeismicДокумент5 страницFragility Curves For Mixed Concrete-Steel Frames Subjected To SeismicJulián PovedaОценок пока нет
- Development of Mmlps in India: Western DFC Eastern DFCДокумент2 страницыDevelopment of Mmlps in India: Western DFC Eastern DFCsdfg100% (2)
- Word Formation ListДокумент8 страницWord Formation ListpaticiaОценок пока нет
- ISO IEC 11801-5 - IT - Generic Cabling For Data CentreДокумент63 страницыISO IEC 11801-5 - IT - Generic Cabling For Data Centreshazwanshaiful1Оценок пока нет
- Rociadores - FT - GFS-100B - GL SeriesДокумент2 страницыRociadores - FT - GFS-100B - GL SeriesJimmy FernándezОценок пока нет
- Acr On Clean-Up DriveДокумент7 страницAcr On Clean-Up DriveRichard ToliaoОценок пока нет