Академический Документы
Профессиональный Документы
Культура Документы
51LC S13 Elimination Background PDF
Загружено:
ButterlesstoastОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
51LC S13 Elimination Background PDF
Загружено:
ButterlesstoastАвторское право:
Доступные форматы
51LC EXP #2 SPRING 2013
DEHYDROBROMINATION OF 1- AND 2-BROMOBUTANE AND
DEHYDRATION OF 1- AND 2-BUTANOL:
ANALYSIS OF GASEOUS PRODUCTS BY GAS CHROMATOGRAPHY 1
The acid-catalyzed dehydration of 1-butanol and 2-butanol and the base-induced
dehydrobromination of 1-bromobutane and 2-bromobutane with strong base both lead to the
formation of a mixture of butene isomers. The composition of this mixture varies, however, as a
result of mechanistic differences in these two pathways.
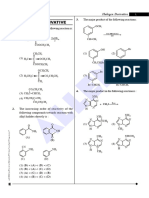
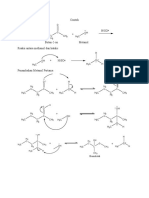
A1. Acid-catalyzed dehydration of a secondary alcohol:
Dehydration of a secondary alcohol proceeds readily in the presence of strong acid such as
sulfuric or phosphoric acid, and proceeds via an E1 mechanism whose intermediates, but not
arrows, are shown in Scheme 1. In step 1, protonation of the hydroxyl group of the alcohol with
the acid catalyst converts the poor leaving group -OH to a much better leaving group, H2O. The
loss of a water molecule from the oxonium ion intermediate results in a carbocation intermediate
that undergoes E1 elimination to form an alkene. If the elimination occurs with either one of the
protons from the terminal methyl (CH3) group, the resulting product is the terminal alkene – 1-
butene. Elimination from the protons on the methylene (CH2) group leads to an internal alkene.
Depending on which hydrogen is deprotonated, either the cis- or trans-alkene can be formed. The
least substituted alkene product is also known as the Hoffman product, and the more-substituted,
alkenes are known as the Saytzeff products. Of the two possible Saytzeff products, the trans-
alkene is the most stable. Since the E1 reaction is under thermodynamic control, the relative
stability of the resulting alkenes should determine the product distribution ratio. This is also known
as Saytzeff’s rule.
Scheme 1:
H3C
CH3 CH3
H2SO4
CH3 H3C CH2
H3C
O
H H3C H3C +
HO H ∆ H
H H H3C CH3
Hoffman
product
Saytzeff
products
1
A part of this procedure is adopted from an article published by H.M. Gilow in the Journal of Chemical Education,
CHEM 51LC Rev 3/12/13
According to LeChatelier's principle, removing a product from a chemical system at
equilibrium shifts the equilibrium in the direction favoring the formation of the products. You will
carry out the dehydration reaction in a reaction tube connected to a gas collector so that the product
will continuously escape out of the reaction mixture as it is formed. Removal of the product will
shift the equilibrium to the right and thus complete the reaction. The collected gaseous product
will be analyzed by gas chromatography, which will show peaks with fair resolution. From the
relative area of peaks, you can calculate the percentage composition of the product mixture.
A2. Acid-catalyzed dehydration of a primary alcohol:
Dehydration of a primary alcohol proceeds analogously to that of a secondary one. The
major difference is that instead of forming a secondary carbocation, the loss of a water molecule
results in the formation of the much more unstable primary carbocation (Scheme 2). Elimination of
that intermediate should result solely in the formation of the terminal alkene 1-butene. The high
activation energy required to form the carbocation intermediate, however, should be so high that
no elimination reaction occurs.
Scheme 2:
H
H2SO4
H
H3C OH
∆
H3C O
H
X H3C H H3C
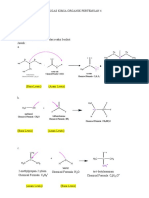
B1. Base-induced dehydrobromination of a secondary bromide:
Base-induced dehydrohalogenation of an alkyl halide is another alternative for synthesizing
alkenes, and proceeds via an E2 mechanism. The use of strong and bulky base promotes the
elimination reaction and disfavors the competing SN2 reaction. The E2 reaction requires an anti-
periplanar arrangement of the beta-H and leaving group (the beta-H and leaving group must be in
the same plane and anti to each other). Because 2-bromobutane has two different types of beta-
H’s, elimination on either side can lead to both internal and terminal alkenes (Scheme 3).
CHEM 51LC Rev 3/12/13
Traditionally, E2
Scheme 3:
eliminations also follow
Saytzeff’s rule. However, Base
H
H
the use of a very bulky base H H3C
H H3C
can change the outcome of H3C
Br
H
Hoffman H H
product
the reaction. When there
Br
are steric problems
approaching one of the
H
beta-hydrogens, the base H3C
Base H
will preferentially H
CH3
H3C CH3
deprotonate the hydrogen H CH3 H CH3
Br
that is least sterically Br
congested by adjacent alkyl
groups. In the case of 2-
H
bromobutane, this most- Base
H3C H
accessible hydrogen is the H
H
CH3
H3C CH3
CH3
hydrogen on the terminal Br H3C H
methyl (CH3) group Saytzeff
Br
products
resulting in the formation
of the least substituted alkene.
B2. Base-induced dehydrobromination of a primary bromide:
The base-induced dehydrohalogenation of 1-bromobutane is analogous to that of 2-
bromobutane. Unlike 2-bromobutane, however, the halide resides on the terminal methyl group
and therefore there is only one possible elimination (Scheme 4).
Scheme 4:
Base
H
H CH3
H CH3
H
Br
CHEM 51LC Rev 3/12/13
C. A note about unexpected results:
Before running a reaction, chemists apply concepts from similar reactions to try and predict
the results. Oftentimes, this works and the outcome is predicted correctly. Sometimes, this does
not work and it is left to the scientist (you) to come up with a rationale for the results observed.
Bear in mind that an unexpected outcome is not necessarily because the concepts you applied were
wrong, but perhaps because something else is at work.
One or more reactions in this set of experiments might give unexpected results. Your job is
to identify which reactions gave the expected results, which give unexpected results, and explain
the unexpected results.
CHEM 51LC Rev 3/12/13
Вам также может понравиться
- Drug Abuse Ag8114en MKДокумент56 страницDrug Abuse Ag8114en MKA VegaОценок пока нет
- Organic compounds structures and reactionsДокумент2 страницыOrganic compounds structures and reactionsNicolas MartinezОценок пока нет
- Important reactions of aromatic compounds summarizedДокумент1 страницаImportant reactions of aromatic compounds summarizedRoronoa ZoroОценок пока нет
- Mini Presentation of IBu-For Prof - SankararamanДокумент16 страницMini Presentation of IBu-For Prof - SankararamanCreative ThinkerОценок пока нет
- Aldehydes and KetonesДокумент58 страницAldehydes and KetonesAniruddha KawadeОценок пока нет
- Neutral pH Flow BatteryДокумент1 страницаNeutral pH Flow BatteryMonika Bartocha WróblewskaОценок пока нет
- Chem SketchДокумент17 страницChem SketchUsman GhaniОценок пока нет
- BiosyntheseДокумент1 страницаBiosyntheseBITOMBOОценок пока нет
- CHEMSKETCHДокумент17 страницCHEMSKETCHUsman GhaniОценок пока нет
- Reductions PPT 29-08-2020Документ12 страницReductions PPT 29-08-2020jkc collegeОценок пока нет
- An Overview of Alkaloids: Their Classification, Extraction, Isolation, and Role in PlantsДокумент49 страницAn Overview of Alkaloids: Their Classification, Extraction, Isolation, and Role in PlantsmanishaОценок пока нет
- S20 Ac Quim 10 Viellard EstebanДокумент1 страницаS20 Ac Quim 10 Viellard Estebanesteban viellardОценок пока нет
- S22 - URAIAN Dan KONTROL PROSES PEMBUATANДокумент7 страницS22 - URAIAN Dan KONTROL PROSES PEMBUATANTrisnawati AmaliaОценок пока нет
- Halogen DerivativeДокумент6 страницHalogen DerivativeSantanu DasОценок пока нет
- Practice Problems On Alkane Nomenclature: CH CHДокумент2 страницыPractice Problems On Alkane Nomenclature: CH CHRishav Sasmal100% (1)
- Estructura de Compuestos Biologicos-Parte 2: F.-EsteroidesДокумент7 страницEstructura de Compuestos Biologicos-Parte 2: F.-EsteroidesffhkeОценок пока нет
- OrganicChemistryChapter7 PDFДокумент30 страницOrganicChemistryChapter7 PDFSeanne CruzОценок пока нет
- Program Chem Check-Up 2.0: Predicting structures, naming compounds and identifying reactionsДокумент2 страницыProgram Chem Check-Up 2.0: Predicting structures, naming compounds and identifying reactionsanis fazilaОценок пока нет
- Problem Set 3, Question 2 InitiationДокумент1 страницаProblem Set 3, Question 2 InitiationRudra Shankha NandyОценок пока нет
- Journal Pre-ProofДокумент53 страницыJournal Pre-ProofAndres Fernando Silvestre SuarezОценок пока нет
- Alcohol, Phenol - EthersДокумент1 страницаAlcohol, Phenol - Etherssarthakyedlawar04Оценок пока нет
- N-Acylation and O-AcylationДокумент2 страницыN-Acylation and O-AcylationBritany DyerОценок пока нет
- Functional Group Interconversion Scheme PDFДокумент1 страницаFunctional Group Interconversion Scheme PDFBilal AhmadОценок пока нет
- Chem 115 Myers: Stereoselective, Directed Aldol ReactionДокумент24 страницыChem 115 Myers: Stereoselective, Directed Aldol ReactionChemical MoleculeОценок пока нет
- A Single-Event Microkinetic Model For Xylene Isomerisation and Ethylbenzene DealkylationДокумент2 страницыA Single-Event Microkinetic Model For Xylene Isomerisation and Ethylbenzene DealkylationAor PimpornОценок пока нет
- Summary of All Reactions For Organic ChemistryДокумент4 страницыSummary of All Reactions For Organic Chemistryfoodytang91% (23)
- Module-3 (Part-I)Документ218 страницModule-3 (Part-I)Prajay GОценок пока нет
- Ejercicios de Nomenclatura de Alcoholes.Документ3 страницыEjercicios de Nomenclatura de Alcoholes.Diego Fernando Ardila ArizaОценок пока нет
- FROM MULTIPLE METHODS: PREPARATION OF PHENOL FROM ALKENES, DIAZONIUM SALTS, AND CUMENEДокумент1 страницаFROM MULTIPLE METHODS: PREPARATION OF PHENOL FROM ALKENES, DIAZONIUM SALTS, AND CUMENERonak kadamОценок пока нет
- FitoquimiaДокумент2 страницыFitoquimialuisgerardo000000Оценок пока нет
- Chemfig enДокумент82 страницыChemfig enDũng Nguyễn NhoОценок пока нет
- IIT JEE Screen 2004 PDFДокумент17 страницIIT JEE Screen 2004 PDFharshОценок пока нет
- MechanismДокумент2 страницыMechanismRyan BoodramlallОценок пока нет
- Chemistry Common Name.Документ1 страницаChemistry Common Name.jangidlokeshkumar11Оценок пока нет
- F H C B F F F Chemical Formula: CH BF OДокумент3 страницыF H C B F F F Chemical Formula: CH BF OFadilla AzhariОценок пока нет
- Grignard Reagent C C Bond Mind MapДокумент1 страницаGrignard Reagent C C Bond Mind MapSii SheikhОценок пока нет
- Cope EliminationДокумент2 страницыCope EliminationArt Julius D. HallazgoОценок пока нет
- Local sales representatives for pricing and availability of monomers listДокумент36 страницLocal sales representatives for pricing and availability of monomers listandersonquimicaОценок пока нет
- Reaksi antara Metanol dan Butan-2-on menghasilkan Ketal 2,2-dimetoksibutanaДокумент2 страницыReaksi antara Metanol dan Butan-2-on menghasilkan Ketal 2,2-dimetoksibutanaElis TianiОценок пока нет
- Reaksi antara Metanol dan Butan-2-on menghasilkan Ketal 2,2-dimetoksibutanaДокумент2 страницыReaksi antara Metanol dan Butan-2-on menghasilkan Ketal 2,2-dimetoksibutanaElis TianiОценок пока нет
- Amino Acid Sidechain ChargesДокумент2 страницыAmino Acid Sidechain ChargesAlisaClarkОценок пока нет
- SEM 8 Chemsketch 2022Документ12 страницSEM 8 Chemsketch 2022Leyre FunciaОценок пока нет
- Grignard Reagent C-C Bond: R D O OДокумент1 страницаGrignard Reagent C-C Bond: R D O OKeshav PargeeОценок пока нет
- Organic Compounds StructuresДокумент7 страницOrganic Compounds StructuresDewei LohОценок пока нет
- Enols and Enolate Ions: Key Intermediates in Carbonyl ChemistryДокумент1 страницаEnols and Enolate Ions: Key Intermediates in Carbonyl ChemistryshinexblazerОценок пока нет
- Exp 7 Preparation of AlkenesДокумент14 страницExp 7 Preparation of AlkenesGeorge PiliposyanОценок пока нет
- e8ffa69f-b114-4cc9-a141-f659e484fd68-1712932543188-4102600920398609Документ20 страницe8ffa69f-b114-4cc9-a141-f659e484fd68-1712932543188-4102600920398609rafiyashaik943Оценок пока нет
- Dia orДокумент8 страницDia orNaman MahawarОценок пока нет
- Alcohol Phenol Ether (1) 6Документ9 страницAlcohol Phenol Ether (1) 6sdnishacОценок пока нет
- Nomenclature of Organic CompoundsДокумент80 страницNomenclature of Organic CompoundsSajjad MiraniОценок пока нет
- Parte 4Документ1 страницаParte 4Esteban Mauricio Viellard CorralesОценок пока нет
- Trabajo Quimica Superior NAVIDADДокумент13 страницTrabajo Quimica Superior NAVIDADSebastian GuerraОценок пока нет
- Reaksi Esterifikasi Asam Sederhana Menggunakan NaOHДокумент1 страницаReaksi Esterifikasi Asam Sederhana Menggunakan NaOHSoy Viranda KusumaОценок пока нет
- 314 Stereochem ProbsДокумент14 страниц314 Stereochem ProbsAtul SinghОценок пока нет
- 1,3 CycloДокумент1 страница1,3 CycloIsmail ZitouniОценок пока нет
- DM pp61-80Документ20 страницDM pp61-80MLUNGISI MkhwanaziОценок пока нет
- L1 - S2 - PW1 - Bromocresol Green in Aqueous SolutionДокумент4 страницыL1 - S2 - PW1 - Bromocresol Green in Aqueous SolutionZirəddin TağıyevОценок пока нет
- Alkenes 1 QPДокумент7 страницAlkenes 1 QPemanОценок пока нет
- Clicker Quiz 3 AnswersДокумент10 страницClicker Quiz 3 AnswersButterlesstoastОценок пока нет
- Dehydration of An AlcoholДокумент13 страницDehydration of An AlcoholButterlesstoastОценок пока нет
- Homework 5 Chapt 13, 14, 15, 16, 17 AnswersДокумент11 страницHomework 5 Chapt 13, 14, 15, 16, 17 AnswersButterlesstoastОценок пока нет
- Calculating Percent Recovery and YieldДокумент2 страницыCalculating Percent Recovery and YieldNur AishaОценок пока нет
- Syllabus Calc3 271Документ4 страницыSyllabus Calc3 271ButterlesstoastОценок пока нет
- Exam 2 Chem350 - F16 (Key)Документ8 страницExam 2 Chem350 - F16 (Key)ButterlesstoastОценок пока нет
- Chem220 SpectrophotometryДокумент46 страницChem220 SpectrophotometryButterlesstoastОценок пока нет
- Chem350 Exam 3 - F16 - KeyДокумент9 страницChem350 Exam 3 - F16 - KeyButterlesstoastОценок пока нет
- OC307 Solving NMRДокумент11 страницOC307 Solving NMRNur Farhanah ZulkifliОценок пока нет
- Purify Acetanilide CrystalsДокумент6 страницPurify Acetanilide CrystalsButterlesstoastОценок пока нет
- Glycolysis Handout Payoff AnswersДокумент2 страницыGlycolysis Handout Payoff AnswersButterlesstoastОценок пока нет
- Atomic TheoryДокумент3 страницыAtomic TheoryButterlesstoastОценок пока нет
- Chem 31.1 FR1 SantosДокумент5 страницChem 31.1 FR1 SantosClaire SantosОценок пока нет
- New process for ethylene production from propaneДокумент4 страницыNew process for ethylene production from propaneBuat MainОценок пока нет
- 05-Chem Eng Tools 2016Документ11 страниц05-Chem Eng Tools 2016arif thoha bariklanaОценок пока нет
- Optimum Transportation Method For Development of Extra Heavy Crude OilДокумент21 страницаOptimum Transportation Method For Development of Extra Heavy Crude OilYudi SuwandaОценок пока нет
- Combustion Theory PPT OriginalДокумент30 страницCombustion Theory PPT Originalsameer betalОценок пока нет
- As Fulfillment of The Equirements For TheДокумент58 страницAs Fulfillment of The Equirements For TheTechnicus Proyectos ElectromecánicosОценок пока нет
- Isocratic Gradient HPLCДокумент52 страницыIsocratic Gradient HPLCfarisaОценок пока нет
- Description of Ammonia Manufacturing ProcessesДокумент4 страницыDescription of Ammonia Manufacturing ProcessesSameer PandeyОценок пока нет
- Downhole Water Sink/Loop: Supervisor: Dr. Hashemi Zadeh Student: Mahdi Tohidi Mobark AabadДокумент15 страницDownhole Water Sink/Loop: Supervisor: Dr. Hashemi Zadeh Student: Mahdi Tohidi Mobark AabadmehdiОценок пока нет
- Boiler Chiller ChecklistДокумент5 страницBoiler Chiller ChecklistSyed Haider Abbas KazmiОценок пока нет
- Gas Processing Packages Details InformationДокумент46 страницGas Processing Packages Details InformationmgkvprОценок пока нет
- Selected List of Iranian-Linked Ships Marked by US Treasury DeptДокумент3 страницыSelected List of Iranian-Linked Ships Marked by US Treasury DeptShurat HaDin - Israel Law CenterОценок пока нет
- Ox0 Alcohols: Report No. 21Документ9 страницOx0 Alcohols: Report No. 21kaimanwatsoNОценок пока нет
- Lead Commissioning Engineer - Process Bala ResumeДокумент5 страницLead Commissioning Engineer - Process Bala ResumeMidha Neer50% (2)
- Presentation Lecture Slides Petroleum Refinery EngineeringДокумент49 страницPresentation Lecture Slides Petroleum Refinery EngineeringMUHAMMADTAHA SARWAR100% (1)
- PT Perkasa Heavyndo Engineering Welder Qualification RecordsДокумент91 страницаPT Perkasa Heavyndo Engineering Welder Qualification RecordstskmechОценок пока нет
- Measuring Boiler Efficiency - ASMEДокумент8 страницMeasuring Boiler Efficiency - ASMEyvesdepermentier4622Оценок пока нет
- Gas y Petroleo Enero 2017Документ96 страницGas y Petroleo Enero 2017Jose Santos100% (1)
- Reduce Hazards-Vaccum SystemДокумент7 страницReduce Hazards-Vaccum SystemChem.EnggОценок пока нет
- Organic Chemistry sn2 sn1 E2 E1 PDFДокумент3 страницыOrganic Chemistry sn2 sn1 E2 E1 PDFMCHENLOLОценок пока нет
- Absorption Fundamentals and ApplicationsДокумент57 страницAbsorption Fundamentals and ApplicationsAini PrameswariОценок пока нет
- Fluidized Bed CombustionДокумент2 страницыFluidized Bed Combustionlaila salahОценок пока нет
- Rasching RingsДокумент10 страницRasching RingsMisael SierraОценок пока нет
- Chemical Reaction Rate of ReactionДокумент12 страницChemical Reaction Rate of ReactionDon Amaru SarmaОценок пока нет
- Prospects For Alkaline Zero Gap Water Electrolysers For Hydrogen ProductionДокумент16 страницProspects For Alkaline Zero Gap Water Electrolysers For Hydrogen Productionkhan47pkОценок пока нет
- Malampaya Case StudyДокумент15 страницMalampaya Case StudyMark Kenneth ValerioОценок пока нет
- 87 - Case Study On Multicomponent Distillation and Distillation Column SequencingДокумент15 страниц87 - Case Study On Multicomponent Distillation and Distillation Column SequencingFranklin Santiago Suclla Podesta50% (2)
- Selective Hydrogenation Processes: Beth Mcculloch, Charles Luebke, and Jill MeisterДокумент6 страницSelective Hydrogenation Processes: Beth Mcculloch, Charles Luebke, and Jill MeisterBharavi K SОценок пока нет
- Refining Processes Handbook 2006Документ222 страницыRefining Processes Handbook 2006Neagu Mihaela100% (1)
- 6645646Документ2 страницы6645646honeylet tayactacОценок пока нет