Академический Документы
Профессиональный Документы
Культура Документы
Panunumpa NG Propesyonal
Загружено:
Justin DiazИсходное описание:
Оригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Panunumpa NG Propesyonal
Загружено:
Justin DiazАвторское право:
Доступные форматы
IMPORTANT: PLEASE READ
PART III: CONSUMER INFORMATION When given with an appropriate dose of estrogen hormone,
PROVERA significantly reduces the risk of developing
PR
PROVERA* endometrial hyperplasia (overgrowth of the uterus lining) and
(medroxyprogesterone acetate) the risk of cancer of the uterus. Seven (7) days after stopping
PROVERA treatment, you should have withdrawal bleeding. If
This leaflet is part III of a three-part "Product bleeding occurs during the PROVERA treatment, talk to your
Monograph" published when PROVERA was approved doctor, as the dose may need to be modified.
for sale in Canada and is designed specifically for
Consumers. This leaflet is a summary and will not tell you Progesterone therapy regulates bleeding patterns. When given to
everything about PROVERA. Contact your doctor or non-pregnant women to treat menstrual disorders, the
pharmacist if you have any questions about the drug. endometrium (inner lining of the uterus) does not grow as much
and bleeding decreases. This is because PROVERA helps to
balance the effect of estrogen. You will know that PROVERA is
ABOUT THIS MEDICATION
effective if it induces a period (any bleeding more than light
spotting) within 2 weeks after PROVERA is given. This
What the medication is used for: bleeding will usually occur 2-7 days after the PROVERA is
finished.
• Hormonal Replacement Therapy for Menopause –
PROVERA is used for women with intact uteri who are For Oncology:
also receiving estrogen therapy. When used to treat cancer, PROVERA is thought to work two-
• To Treat Menstrual Disorders due to Hormonal Imbalance ways: it decreases hormone release and it prevents the cancer
- In non-pregnant women, PROVERA helps to balance the cells from multiplying by balancing out high levels of estrogen.
effect of estrogen. The response to hormonal therapy for endometrial or breast
cancer may not be evident until 8 to 10 weeks of therapy.
• To Treat Cancer:
- To treat and relieve symptoms associated with When it should not be used:
endometrial cancer.
- To treat and relieve symptoms of breast cancer in You should not take PROVERA if you have any of the
postmenopausal women. following conditions:
PROVERA should be used only under the supervision of a • In the presence of liver disease;
doctor, with regular follow-up at least once a year to identify • If you have had or have cancer of the breast or uterus,
the side effects associated with its use. Your first follow-up unless PROVERA is being used to treat and relieve the
visit should be within 3 to 6 months of starting treatment. symptoms associated with these cancers;
Your visit may include a blood pressure check, a breast exam, • If you have abnormal vaginal bleeding;
a Pap smear and pelvic exam. You should have a • If you are pregnant;
mammogram before starting treatment and at regular intervals • If you have had or have any blood circulation problems,
as recommended by your doctor. Your doctor may such as blood clots, stroke, blindness or migraine
recommend some blood tests. headaches;
• If you have had a stroke or heart attack;
Progestin, when used with estrogen therapy, have important • If you have had an unusual allergic response to
benefits but also some risks. You should carefully discuss the progestin or to any of the ingredients in the tablet.
risks and benefits of hormone replacement therapy (HRT)
with your doctor. If you use progestin with estrogen therapy, Do not take PROVERA for conditions for which it was not
check with your doctor to be sure you are using the lowest prescribed. Do not give PROVERA to other people, even if they
possible dose that works, and that you don’t use them longer have the same symptoms you have. It may harm them.
than necessary. How long you need to use HRT will depend
on the reason for use. You should regularly talk with your What the medicinal ingredient is:
doctor about whether you still need treatment with HRT.
Medroxyprogesterone acetate
If you have any questions about this medication or your
condition, please ask your doctor or pharmacist. What the nonmedicinal ingredients are:
What it does: PROVERA tablets also contain calcium stearate, corn starch,
lactose monohydrate, mineral oil, sucrose, talc. The 2.5 mg
PROVERA (medroxyprogesterone acetate) is a hormone tablet contains FD&C Yellow No. 6 and the 5 mg tablet contains
replacement therapy (HRT) that contains a manufactured FD&C Blue No.2 aluminum lake.
progesterone hormone, progestin, that is similar to the
progesterone produced by a woman’s ovaries.
PROVERA (medroxyprogesterone acetate) Product Monograph Page 34 of 38
IMPORTANT: PLEASE READ
What dosage forms it comes in: In addition, women with a family history of breast cancer or
women with a history of breast lumps, breast biopsies or
PROVERA tablets are available as: abnormal mammograms (breast x-rays) should consult with their
doctor before starting HRT.
• 2.5 mg circular, orange tablets embossed with "U 64"
on one side and scored on the other. Women should have a mammogram before starting HRT and at
• 5 mg circular, blue tablets embossed with "U 286" on regular intervals during treatment as recommended by their
both sides of a break score and on the other surface doctor.
with “U”. Regular breast examinations by a doctor and regular breast self-
• 10 mg circular, white tablets embossed with "Upjohn examinations are recommended for all women. You should
50" on one side and scored on the other. review technique for breast self-examination with your doctor.
WARNINGS AND PRECAUTIONS Overgrowth of the lining of the uterus and cancer of the uterus
Serious Warnings and Precautions The use of estrogen-alone therapy by post menopausal women
who still have a uterus increases the risk of developing
The Women’s Health Initiative (WHI) trial is a large clinical endometrial hyperplasia (overgrowth of the lining of the uterus),
study that assessed the benefits and risks of oral combined which increases the risk of endometrial cancer (cancer of the
estrogen plus progestin therapy and oral estrogen-alone lining of the uterus).
therapy compared with placebo (a pill with no active
ingredients) in postmenopausal women. The purpose of adding a progestin medication to estrogen
therapy is to reduce the risk of endometrial hyperplasia.
The WHI trial indicated an increased risk of myocardial
infarction (heart attack), stroke, breast cancer, pulmonary You should discuss progestin therapy and risk factors for
emboli (blood clots in the lungs) and deep vein thrombosis endometrial hyperplasia and endometrial carcinoma with your
(blood clots in the large veins) in postmenopausal women doctor. You should also report any unexpected or unusual
taking oral combined estrogen plus progestin. vaginal bleeding to your doctor.
The WHI trial indicated an increased risk of stroke and deep If you have had your uterus removed, you are not at risk of
vein thrombosis in postmenopausal women with prior developing endometrial hyperplasia or endometrial carcinoma.
hysterectomy (surgical removal of the uterus) taking oral Progestin therapy is therefore not generally required in women
estrogen-alone. who have had a hysterectomy.
Therefore, you should highly consider the following: Ovarian Cancer
• There is an increased risk of developing invasive breast
cancer, heart attack, stroke and blood clots in both lungs In some studies, the use of estrogen-alone and estrogen plus
and large veins with the use of estrogen plus progestin progestin therapies for 5 or more years has been associated with
therapy. an increased risk of ovarian cancer.
• There is an increased risk of stroke and blood clots in the
large veins with the use of estrogen-alone therapy. Heart Disease and Stroke
• Estrogens with or without progestins should not be used
for the prevention of heart disease or stroke. The results of the WHI trial indicated an increased risk of stroke
• Estrogens with or without progestins should be used at and coronary heart disease in post-menopausal women taking
the lowest effective dose and for the shortest period of combined estrogen plus progestin compared to women taking
time possible. Regular medical follow-up is advised. placebo.
Breast Cancer The results of the WHI trial indicated an increased risk of stroke,
but no difference in the risk of coronary heart disease in post-
The results of the WHI trial indicated an increased risk of menopausal women with prior hysterectomy taking estrogen
breast cancer in post-menopausal women taking combined alone compared to women taking placebo.
estrogen plus progestin compared to women taking placebo.
The results of the WHI trial indicated no difference in the risk Abnormal Blood Clotting
of breast cancer in postmenopausal women with prior
hysterectomy taking estrogen-alone compared to women The results of the WHI trial indicated an increased risk of blood
taking placebo. clots in the lungs and large veins in post-menopausal women
taking combined estrogen plus progestin compared to women
Estrogens with or without progestins should not be taken by taking placebo.
women who have a personal history of breast cancer.
The results of the WHI trial indicated an increased risk of blood
clots in the large veins, but no difference in the risk of blood
PROVERA (medroxyprogesterone acetate) Product Monograph Page 35 of 38
IMPORTANT: PLEASE READ
clots in the lungs in post-menopausal women with prior
INTERACTIONS WITH THIS MEDICATION
hysterectomy taking estrogen-alone compared to women
taking placebo.
Tell your doctor or pharmacist if you are taking any other
The risk of blood clots also increases with age, if you or a medications, including prescription medications, over-the-
family member has had blood clots, if you smoke or if you are counter medications, vitamins or herbal products.
severely overweight. The risk of blood clots is also
temporarily increased if you are immobilized for long periods Drugs that may interact with PROVERA include:
of time and following major surgery. You should discuss risk • Preparations inducing liver enzymes (eg. barbiturates,
factors for blood clots with your doctor since blood clots can hydantoins, carbamazepine, meprobamates,
be life threatening or cause serious disability. phenylbutazone or rifampin);
• Aminoglutethimide;
Gallbladder Disease • Some herbal (eg. St. John’s wort) and natural products
which are bought without a prescription.
The use of estrogens by postmenopausal women has been
associated with an increased risk of gallbladder disease
requiring surgery. PROPER USE OF THIS MEDICATION
Dementia
PROVERA should be taken by mouth.
The Women’s Health Initiative Memory Study (WHIMS) was
a substudy of the WHI trial and indicated an increased risk of PROVERA can be taken with or without food.
dementia (loss of memory and intellectual function) in
postmenopausal women age 65 and over taking oral combined Usual dose:
estrogen plus progestin compared to women taking placebo.
The WHIMS indicated no difference in the risk of dementia in 1. Hormonal Replacement Therapy for Menopause
post-menopausal women age 65 and over with prior The recommended dose for women is 5 to 10 mg daily for
hysterectomy taking oral estrogen-alone compared to women 12 to 14 days.
taking placebo.
2. Functional Menstrual Disorders due to Hormonal
BEFORE you use PROVERA talk to your doctor or Imbalance
pharmacist if: (a) Secondary amenorrhea (absence of menstrual period):
After ruling out pregnancy, PROVERA, may be
• have a history of allergy or intolerance to any medications administered in doses ranging from 5 to 10 mg daily for
or other substances 12 to 14 days every month.
• have a personal history of breast disease (including breast (b) Dysfunctional uterine bleeding:
lumps) and/or breast biopsies, or a family history of breast PROVERA may be given in doses ranging from 5 to 10
cancer mg daily for 10 to 14 days, beginning on the 12th to 16th
• have experienced any unusual or undiagnosed vaginal day of the cycle. This regimen should be repeated for 2
bleeding subsequent cycles or longer if necessary.
• have a history of uterine fibroids or endometriosis
• have a history of liver disease, jaundice (yellowing of the 3. Endometrial Cancer
eyes and/or skin) or itching related to estrogen use or during The recommended dose is 200 to 400 mg daily.
pregnancy
• have a history of migraine headache 4. Breast Cancer
• have a history of high blood pressure The recommended dose is 400 mg daily, given in divided
• have a personal or family history of blood clots, or a doses.
personal history of heart disease or stroke
• have a history of kidney disease, asthma or epilepsy It is important that you take PROVERA regularly at the same
(seizures) time each day.
• have a history of bone disease (this includes certain
metabolic conditions or cancers that can affect blood levels Overdose:
of calcium and phosphorus)
• have been diagnosed with diabetes Symptoms: Overdosage may result in a period of amenorrhea
• have been diagnosed with porphyria (a disease of blood (absence or cessation of menstrual period) and may be followed
pigment) by irregular menses for several cycles.
• have a history of high cholesterol or high triglycerides
• are pregnant or may be pregnant For management of a suspected drug overdose, contact your
• have had a hysterectomy (surgical removal of the uterus) regional Poison Control Centre
• smoke
PROVERA (medroxyprogesterone acetate) Product Monograph Page 36 of 38
IMPORTANT: PLEASE READ
Missed Dose:
SERIOUS SIDE EFFECTS, HOW OFTEN THEY
Do not take a double dose to make up for a forgotten tablet. If
HAPPEN AND WHAT TO DO ABOUT THEM
you miss a dose, take it as soon as you can if it is within 12
hours of the missed dose. If it is over 12 hours since your
missed dose, skip the missed dose and go back to your usual Frequency Symptom / Talk with Stop taking
possible side your doctor or drug and
dosing times
effect pharmacist call your
doctor or
Only In all pharmacist
SIDE EFFECTS AND WHAT TO DO ABOUT THEM
if cases
severe
The following side effects have been reported with the use of Not Persistent sad √
PROVERA: known mood
• Breast tenderness; Abdominal pain,
• Breast milk secretion; nausea or
• Breakthrough bleeding; vomiting
√
• Spotting (minor vaginal bleeding); Pain or swelling √
• Irregular menstrual periods; in the leg
• Amenorrhea (absence of menstrual periods); /inflamed vein
• Vaginal secretions; Breast lump
• Headaches;
• Nervousness; √
• Dizziness; Sharp pain in √
• Insomnia, sleepiness, fatigue; the chest,
• Premenstrual syndrome-like symptoms; coughing blood
• Itching, hives, skin rash; or sudden
• Acne; shortness of
breath
• Hair loss, hair growth;
Sudden severe
• Abdominal discomfort; nausea; bloating; √
headache or
• Fever; worsening of
• Increase in weight; headache,
• Swelling; vomiting,
• Moon shaped face. dizziness,
fainting,
If you experience any of these side effects, contact your disturbance of
vision or speech
doctor or pharmacist.
or weakness or
numbness in an
The side effects listed in the table below have been observed arm or leg
with estrogen/progestin combination in general, but not Sudden partial
necessarily with PROVERA. √
or complete loss
of vision
Unexpected
√
vaginal bleeding
Yellowing of the
√
skin or eyes
(jaundice)
Crushing chest √
pain or chest
heaviness
This is not a complete list of side effects. For any unexpected
effects while taking PROVERA, contact your doctor or
pharmacist.
HOW TO STORE IT
PROVERA should be stored at controlled room temperature,
15-30°C. Keep out of the reach of children.
PROVERA (medroxyprogesterone acetate) Product Monograph Page 37 of 38
IMPORTANT: PLEASE READ
REPORTING SUSPECTED SIDE EFFECTS
You can report any suspected adverse reactions
associated with the use of health products to the
Canada Vigilance Program by one of the
following 3 ways:
• Report online at
www.healthcanada.gc.ca/medeffect
• Call toll-free at 1-866-234-2345
• Complete a Canada Vigilance Reporting
Form and:
- Fax toll-free to 1-866-678-6789, or
- Mail to: Canada Vigilance Program
Health Canada
Postal Locator 0701C
Ottawa, ON K1A 0K9
Postage paid labels, Canada Vigilance Reporting
Form and the adverse reaction reporting
guidelines are available on the MedEffectTM
Canada Web site at
www.healthcanada.gc.ca/medeffect.
NOTE: Should you require information related
to the management of side effects, contact your
health professional. The Canada Vigilance
Program does not provide medical advice.
MORE INFORMATION
This document plus the full product monograph,
prepared for health professionals, may be obtained at:
http://www.pfizer.ca or by contacting the sponsor,
Pfizer Canada Inc. at: 1-800-463-6001.
This leaflet was prepared by Pfizer Canada Inc.
Last revised: 29 September 2014
PROVERA (medroxyprogesterone acetate) Product Monograph Page 38 of 38
Вам также может понравиться
- Narcissistic MothersДокумент11 страницNarcissistic MothersSeraph100% (7)
- HRTДокумент62 страницыHRTArpita ArpitaОценок пока нет
- Guide For Brachytherapy QA PDFДокумент270 страницGuide For Brachytherapy QA PDFtutorhowОценок пока нет
- Maternal Health: Ekta Modi 2 MPT in RehabДокумент368 страницMaternal Health: Ekta Modi 2 MPT in RehabMihir_Mehta_5497100% (1)
- Contraception and FertilityДокумент11 страницContraception and FertilityWil LesterОценок пока нет
- Uwise Gynecology and BreastДокумент24 страницыUwise Gynecology and Breastlizzy596Оценок пока нет
- Ozone Unsung Hero by Navy Captain - Ajit VadakayilДокумент153 страницыOzone Unsung Hero by Navy Captain - Ajit Vadakayilsudhir shahОценок пока нет
- ABCDs During A Code Blue Response in An Adult PatientДокумент23 страницыABCDs During A Code Blue Response in An Adult PatientChakra PuspitaОценок пока нет
- From Outrage To Courage: The Unjust and Unhealthy Situation of Women in Poorer Countries and What They Are Doing About It PDFДокумент97 страницFrom Outrage To Courage: The Unjust and Unhealthy Situation of Women in Poorer Countries and What They Are Doing About It PDFNadine Avila100% (2)
- Drugs 2 (Gyneac)Документ103 страницыDrugs 2 (Gyneac)Aman Shaikh100% (2)
- OBG DrugsДокумент30 страницOBG DrugsSANAОценок пока нет
- Hormonal ContraceptivesДокумент25 страницHormonal Contraceptivesmd easarur rahmanОценок пока нет
- Metabolic and Endocrine Pharmacology: Gonadol DrugsДокумент38 страницMetabolic and Endocrine Pharmacology: Gonadol Drugstheintrov100% (1)
- Bazaar DrugsДокумент104 страницыBazaar DrugsKartik Vashishta100% (1)
- Oral Contraceptives PDFДокумент24 страницыOral Contraceptives PDFSheema TabassumОценок пока нет
- Progesterona La Hormona Definitiva del Bienestar FemeninoОт EverandProgesterona La Hormona Definitiva del Bienestar FemeninoРейтинг: 1 из 5 звезд1/5 (1)
- Medical TermsДокумент4 страницыMedical TermsSamОценок пока нет
- Progynova Drugs PDFДокумент5 страницProgynova Drugs PDFYudha fedrian AKОценок пока нет
- Estradiol 10mcg (Vagifen)Документ13 страницEstradiol 10mcg (Vagifen)asdwasdОценок пока нет
- Angeliq PM enДокумент45 страницAngeliq PM enTomacho HitachiОценок пока нет
- Gyne Case Contraception ReportДокумент16 страницGyne Case Contraception ReportKristia Anne PomadaОценок пока нет
- Progestin Only ContraceptionДокумент4 страницыProgestin Only ContraceptionTareq SawanОценок пока нет
- Progestin-Only Injectables Fact Sheet Final - 102510Документ2 страницыProgestin-Only Injectables Fact Sheet Final - 102510Chan Myae AungОценок пока нет
- Premarin Drug StudyДокумент8 страницPremarin Drug StudyjanОценок пока нет
- Artificial Methods of Family PlanningДокумент17 страницArtificial Methods of Family PlanningEduard Anthony AjeroОценок пока нет
- ContraceptionДокумент33 страницыContraceptionShams AtrashОценок пока нет
- Oripro®: What Is in This LeafletДокумент3 страницыOripro®: What Is in This LeafletKirubakaranОценок пока нет
- Blind or Partially Sighted? Is This Leaflet Hard To See or Read? Phone Lexon (UK) Limited, Tel: 01527 505414 To Obtain The Leaflet in A Format Suitable For YouДокумент4 страницыBlind or Partially Sighted? Is This Leaflet Hard To See or Read? Phone Lexon (UK) Limited, Tel: 01527 505414 To Obtain The Leaflet in A Format Suitable For YouMahmudul Hasan TusharОценок пока нет
- Endometrial Hyperplasia 2019Документ4 страницыEndometrial Hyperplasia 2019indragazaliОценок пока нет
- Contraceptive Methods and Issues Around The MenopauseДокумент18 страницContraceptive Methods and Issues Around The MenopauseLakshmi DheviОценок пока нет
- DmpaДокумент2 страницыDmpaiszahbelleОценок пока нет
- Department of Pharmacy Practice Vels UniversityДокумент29 страницDepartment of Pharmacy Practice Vels UniversityUdtjeVanDerJeykОценок пока нет
- Contraceptives NewДокумент23 страницыContraceptives NewOdiit StephenОценок пока нет
- RH021 Part 3 V 2Документ7 страницRH021 Part 3 V 2Huey VillanuevaОценок пока нет
- Duphaston DR 1410193172635 PDFДокумент8 страницDuphaston DR 1410193172635 PDFjoyce ramirezОценок пока нет
- Menses Manipulation CC JMДокумент9 страницMenses Manipulation CC JMSundar RamanathanОценок пока нет
- Administration of Hormonal Contraceptive Drugs: A Quick Reference Guide For CliniciansДокумент28 страницAdministration of Hormonal Contraceptive Drugs: A Quick Reference Guide For Clinicianshesham00100Оценок пока нет
- Hormonal ContraceptionДокумент3 страницыHormonal ContraceptionSyaZana ShahromОценок пока нет
- Reproductive Pharmacology Lecturio ReflectionДокумент7 страницReproductive Pharmacology Lecturio ReflectionNathaniel SolisОценок пока нет
- Fertility Control: (Contraception)Документ68 страницFertility Control: (Contraception)SaraMohammedОценок пока нет
- Arimidex Drug CardДокумент1 страницаArimidex Drug CardSheri490Оценок пока нет
- PrimolutntabДокумент13 страницPrimolutntabRizki NovitaОценок пока нет
- Family Planning Methods Contraceptive Implants: Who Can Use Implantable Contraception?Документ5 страницFamily Planning Methods Contraceptive Implants: Who Can Use Implantable Contraception?Cindy MaslagОценок пока нет
- Progyluton-26 1DДокумент16 страницProgyluton-26 1DUsma aliОценок пока нет
- Barrier Methods Hormonal Methods Oral Contraceptives Vaginal Rings Implants, Injectables Withdrawal and AbstinenceДокумент37 страницBarrier Methods Hormonal Methods Oral Contraceptives Vaginal Rings Implants, Injectables Withdrawal and AbstinenceEm WalkerОценок пока нет
- d057347e-a773-4d8a-b0d4-5262e798f4b9Документ5 страницd057347e-a773-4d8a-b0d4-5262e798f4b9KirubakaranОценок пока нет
- Pharmacology Reproductive Maternity NewbornДокумент24 страницыPharmacology Reproductive Maternity Newbornamasoud96 amasoud96Оценок пока нет
- Inject Able ContraceptivesДокумент67 страницInject Able Contraceptivessalah subbahОценок пока нет
- Menopause and Hormone Replacement Therapy: Sandra E. Brooks, MD, FACS, FACOG Associate ProfessorДокумент18 страницMenopause and Hormone Replacement Therapy: Sandra E. Brooks, MD, FACS, FACOG Associate ProfessorAris MarutoОценок пока нет
- Warning of use analgesic in dentistry دادعا يرمعلا رمع يرهزلا قراط هلمعش سنا:روكدلا فرشا تحتДокумент8 страницWarning of use analgesic in dentistry دادعا يرمعلا رمع يرهزلا قراط هلمعش سنا:روكدلا فرشا تحتالعمري العمريОценок пока нет
- Warning of use analgesic in dentistry دادعا يرمعلا رمع يرهزلا قراط هلمعش سنا:روكدلا فرشا تحتДокумент8 страницWarning of use analgesic in dentistry دادعا يرمعلا رمع يرهزلا قراط هلمعش سنا:روكدلا فرشا تحتالعمري العمريОценок пока нет
- Estradiol Valerate 2mg + Nongestrel 0.5mg (PROGYLUTON)Документ12 страницEstradiol Valerate 2mg + Nongestrel 0.5mg (PROGYLUTON)ddandan_2Оценок пока нет
- Injectable ContraceptionДокумент9 страницInjectable ContraceptionCarlosCorderoLópezОценок пока нет
- Estradiol Hemihydrate 0.5mg + Norethisterone Acetate 0.1mg (Eviana)Документ12 страницEstradiol Hemihydrate 0.5mg + Norethisterone Acetate 0.1mg (Eviana)asdwasdОценок пока нет
- Committee Opinion: Aromatase Inhibitors in Gynecologic PracticeДокумент5 страницCommittee Opinion: Aromatase Inhibitors in Gynecologic Practicew yОценок пока нет
- Patient Information PROPECIA (Pro-Pee-Sha) (Finasteride)Документ3 страницыPatient Information PROPECIA (Pro-Pee-Sha) (Finasteride)babre2020Оценок пока нет
- WOMENâ S HEALTHnewДокумент52 страницыWOMENâ S HEALTHnewHossen AliОценок пока нет
- ContraceptionДокумент38 страницContraceptionMalk OmryОценок пока нет
- Aco y AcogДокумент2 страницыAco y AcogleslyОценок пока нет
- Depo-Provera Contraceptive Injection: What Is It?Документ2 страницыDepo-Provera Contraceptive Injection: What Is It?Marlene FooteОценок пока нет
- Drug Study Rasmien S. JimmyДокумент6 страницDrug Study Rasmien S. JimmyCHRISTINE JOY. MOLINAОценок пока нет
- Depo ProveraДокумент3 страницыDepo ProveraDivika ShilvanaОценок пока нет
- KB HormonalДокумент8 страницKB Hormonalf4rh4t1Оценок пока нет
- Contraception Lec 1Документ38 страницContraception Lec 1fh2785519Оценок пока нет
- Overview Second Tri Presentation 10.30.17Документ31 страницаOverview Second Tri Presentation 10.30.17Lutfa LipiОценок пока нет
- Contraceptive Assignment AdihailuДокумент46 страницContraceptive Assignment AdihailuAbas AhmedОценок пока нет
- My Mifespristone and Misoprostol Story: How I used mifepristone and misoprostol for a successful medical abortion and all you must know about these abortion pillsОт EverandMy Mifespristone and Misoprostol Story: How I used mifepristone and misoprostol for a successful medical abortion and all you must know about these abortion pillsОценок пока нет
- n55f Children Brochure PDFДокумент2 страницыn55f Children Brochure PDFJustin DiazОценок пока нет
- B. Water Exists On The Surface of The Earth in Three Physical StatesДокумент7 страницB. Water Exists On The Surface of The Earth in Three Physical StatesJustin DiazОценок пока нет
- 16 4 Reasons To Marry: Research SynthesisДокумент21 страница16 4 Reasons To Marry: Research SynthesisJustin DiazОценок пока нет
- 16 4 Reasons To Marry: Research SynthesisДокумент21 страница16 4 Reasons To Marry: Research SynthesisJustin DiazОценок пока нет
- Love and Marriage: April 2015Документ9 страницLove and Marriage: April 2015Justin DiazОценок пока нет
- Gerdy V. Abu: Work ExperienceДокумент3 страницыGerdy V. Abu: Work ExperienceJustin DiazОценок пока нет
- 3 Pre Board General EducationДокумент10 страниц3 Pre Board General EducationJustin DiazОценок пока нет
- Case Study 10Документ2 страницыCase Study 10Justin DiazОценок пока нет
- Case Study 7Документ2 страницыCase Study 7Justin DiazОценок пока нет
- Case Study 9Документ1 страницаCase Study 9Justin DiazОценок пока нет
- Case Study 10Документ2 страницыCase Study 10Justin DiazОценок пока нет
- Case Study 5Документ1 страницаCase Study 5Justin DiazОценок пока нет
- Case Study 8Документ2 страницыCase Study 8Justin DiazОценок пока нет
- Case Study 6Документ2 страницыCase Study 6Justin DiazОценок пока нет
- Splice PDFДокумент5 страницSplice PDFpedroОценок пока нет
- At Merienda - Individual ActivityДокумент6 страницAt Merienda - Individual ActivityMoreno, John Gil F.Оценок пока нет
- ST Peter, 2014, BI-Annual DR-TB Performance Report To AAHB, 15 March 2022. JohnДокумент43 страницыST Peter, 2014, BI-Annual DR-TB Performance Report To AAHB, 15 March 2022. Johnሀይደር ዶ.ርОценок пока нет
- Maui Medical Dispensary Application Redacted Sec3Документ541 страницаMaui Medical Dispensary Application Redacted Sec3melipasaОценок пока нет
- Acute Cardiac EKG Interpretation - Basics Part 1 - ScriptДокумент2 страницыAcute Cardiac EKG Interpretation - Basics Part 1 - ScriptDarby GravinoОценок пока нет
- 109 513 1 PB PDFДокумент11 страниц109 513 1 PB PDFKolu koyОценок пока нет
- AyyДокумент187 страницAyyZoé WolfesОценок пока нет
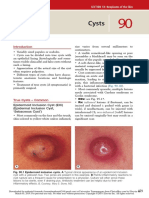
- CystsДокумент5 страницCystsranindita maulyaОценок пока нет
- Euthanasia ThesisДокумент11 страницEuthanasia ThesisAyush MathurОценок пока нет
- Second Hand Smoke Fact SheetДокумент9 страницSecond Hand Smoke Fact Sheetwesa77Оценок пока нет
- Mental Disorder Due To AlcoholДокумент18 страницMental Disorder Due To AlcoholAnonymous Oj5JCpO5xОценок пока нет
- MTL CHAPTER 9 2020 ClickUpДокумент11 страницMTL CHAPTER 9 2020 ClickUpcharlieОценок пока нет
- Covid-19 Expose (Unmask)Документ2 страницыCovid-19 Expose (Unmask)Eduardo CasesОценок пока нет
- Pros and Cons EuthanasiaДокумент3 страницыPros and Cons EuthanasiaMirantika Audina100% (1)
- USMLE Step 1 First Aid 2021-101-230Документ130 страницUSMLE Step 1 First Aid 2021-101-230mariana yllanesОценок пока нет
- Antibiotic Resistance Lesson Plan Final ProjectДокумент10 страницAntibiotic Resistance Lesson Plan Final Projectapi-436796283Оценок пока нет
- RN Physiological Integrity GIДокумент14 страницRN Physiological Integrity GIAaLona RobinsonОценок пока нет
- Ob Osce.04 CTG ReadingДокумент6 страницOb Osce.04 CTG ReadingDasha VeeОценок пока нет
- An Introduction To Volunteering at Camphill DevonДокумент3 страницыAn Introduction To Volunteering at Camphill DevonjohnyОценок пока нет
- Covid-19 Detector Using The Power of Python and Arduino, This Detector Measures and Records User TemperatureДокумент5 страницCovid-19 Detector Using The Power of Python and Arduino, This Detector Measures and Records User TemperatureAshu SОценок пока нет
- Osteoporosis - PathophysiologyДокумент2 страницыOsteoporosis - PathophysiologyMary April MendezОценок пока нет
- Gold Awards: Organization Name Title of InnovationДокумент10 страницGold Awards: Organization Name Title of Innovationchek86351Оценок пока нет
- Maret 18 PDFДокумент178 страницMaret 18 PDFAnonymous vLgCd0U7Оценок пока нет