Академический Документы
Профессиональный Документы
Культура Документы
Chemistry Formulas 11-1-2010
Загружено:
karlitos047Исходное описание:
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Chemistry Formulas 11-1-2010
Загружено:
karlitos047Авторское право:
Доступные форматы
ThinkMCAT.
com
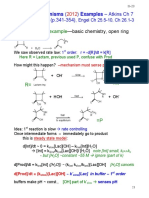
Abridged CHEMISTRY
U = ±Q±W Ka = [H+][A–]
adiabatic [HA]
isothermic
isobaric Kb = [OH-][B+]
[B]
PV = nRT
pKa = -logKa
P1V1 = P2V2 pKb = -logKb
V1 = V2 pH = pKa + log([Conj. Base]/[Acid])
T1 T2
∆Hreaction = ∆Hf(products) - ∆Hf(reactants)
Tkelvin = Tcelsius +273
∆Hreaction = Bonds broken– Bonds formed
M =moles solute/Liter solution
m = moles of solute/kg of solvent
∆Sreaction = ∆Sf(products) - ∆Sf(reactants)
Colligative Properties
Π = MRT ∆Greaction = ∆Gf(products) - ∆Gf(reactants)
∆Tb = Kbm (elevation) ∆G = -nFE; F=105
∆Tf = Kfm (depression) ∆G = -RTlnKeq
Pa = PtotalXsolvent ∆G = ∆H – T∆S
Conductance 1ATM = 760 torr
Avagadro = 6 X 1023
Xa= moles X__ Standard vs. ideal conditions; STP
total moles
HCl, HBr, H2SO4,
%composition= mass X/ total weight HI, HNO3, HClO4 Strong Acids
q = mc∆t NaOH, KOH Strong Bases
q= mL
Hydrogen Bond
E = hf Dipole – Dipole
Van der Waals, Hydrophobic, Intermolecular
v=fƛ
Dispersion Forces
pH = -log [H+]
pOH = -log [OH–] Acidic [H+] > [OH-]
Neutral [H+] = [OH-]
Basic [H+] < [OH-]
[H+][OH–] = 1X10-14
[Ka][Kb] = 1X10-14
ThinkMCAT.com
Abridged CHEMISTRY
1. CNOF Satisfy the octet
a. Except carbocation
b. Except radicals
2. O likes H Lewis
3. Center atom usually least
electronegative Structures
4. Obtain formal charge as close
to zero as possible
Fe3+ → Ferric
(AnOx) Anode → Oxidation Fe2+ → Ferrous Common
(RedCat) Cathode → Reduction Cu2+ → Cupric Names
(OiL) Oxidation is Lost Cu2O → Cuprous Oxide
(RiG) Reduction is Gained
Batteries
∆G E Cathode Anode
Galvanic - + + -
Cell
Electrolytic + - - +
Cell
Move across a Period (left to right) /
Move up a Group
↑ Ionization Energy
↑ Electron Affinity Periodic Trends
↑ Electronegativity
↓ Atomic Radius
Q = Products
Reactants
∆G = (-) Q < Keq → moves to the right Thermodynamics
∆G = 0 Q = Keq → moves both ways
∆G = (+) Q > Keq → moves to the left
Think Education
90 East 3rd Street (between 1st and 2nd Avenue)
New York City, New York, 10003
ThinkMCAT.com
ThinkMCAT@gmail.com
Вам также может понравиться
- BlahДокумент8 страницBlahkwood84100% (1)
- The Brew Your Own Big Book of Clone Recipe - Brew Your OwnДокумент716 страницThe Brew Your Own Big Book of Clone Recipe - Brew Your OwnFrederico Ribeiro67% (3)
- Pescatarian Mediterranean Diet Cookbook 2 - Adele TylerДокумент98 страницPescatarian Mediterranean Diet Cookbook 2 - Adele Tylerrabino_rojoОценок пока нет
- BottomДокумент4 страницыBottomGregor SamsaОценок пока нет
- CASE 721F TIER 4 WHEEL LOADER Operator's Manual PDFДокумент17 страницCASE 721F TIER 4 WHEEL LOADER Operator's Manual PDFfjskedmmsme0% (4)
- Critical Care Nursing Assessment Form: R R R R R R R RДокумент2 страницыCritical Care Nursing Assessment Form: R R R R R R R RPipit Permata100% (1)
- The Aging Brain: Course GuidebookДокумент126 страницThe Aging Brain: Course GuidebookIsabel Cristina Jaramillo100% (2)
- Fortune Falls - Lou VaneДокумент402 страницыFortune Falls - Lou VanetesteОценок пока нет
- Organic Chemistry Crash CourseДокумент4 страницыOrganic Chemistry Crash CourseRobert Velázquez LucianoОценок пока нет
- Practice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersОт EverandPractice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersОценок пока нет
- Photosynthesis PastPaper QuestionsДокумент24 страницыPhotosynthesis PastPaper QuestionsEva SugarОценок пока нет
- Practice Makes Perfect in Chemistry: Oxidation-ReductionОт EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionРейтинг: 5 из 5 звезд5/5 (1)
- Catalyst MaterialsДокумент64 страницыCatalyst MaterialskbcloveОценок пока нет
- 4-Acid-Base 1Документ32 страницы4-Acid-Base 1José de Jesús Treviño ReséndezОценок пока нет
- Organic ChemistryДокумент7 страницOrganic ChemistryABDULLAH SHAHZADОценок пока нет
- Chemistry 2 Physical Chemistry Lecture NotesДокумент48 страницChemistry 2 Physical Chemistry Lecture Noteskittycat1chauОценок пока нет
- Chapter 10: ELECTROLYTE: 10.1. Equilibrium Properties of ElectrolyteДокумент64 страницыChapter 10: ELECTROLYTE: 10.1. Equilibrium Properties of ElectrolyteNguyễn Hoàng DũngОценок пока нет
- SV Epp Zs 2023 30 10 Intro Imr 1aabДокумент241 страницаSV Epp Zs 2023 30 10 Intro Imr 1aabmekkirofaidanОценок пока нет
- 09Документ12 страниц09ZenPhiОценок пока нет
- 1-Introduction 1Документ27 страниц1-Introduction 1watersoul.nОценок пока нет
- Equilibrium Constant of ReactionДокумент55 страницEquilibrium Constant of ReactionTÚ Cao Ngọc ThiệnОценок пока нет
- C3 ElectrochemistryДокумент35 страницC3 ElectrochemistryaliesyaОценок пока нет
- Environmental Engineering Industrial WaterДокумент25 страницEnvironmental Engineering Industrial WaterTowfiq AhmedОценок пока нет
- Energetics - Summary: Exothermic EndothermicДокумент7 страницEnergetics - Summary: Exothermic EndothermicKiara LimОценок пока нет
- AY 2022 Sem 1 A105 Reference MaterialДокумент4 страницыAY 2022 Sem 1 A105 Reference Material65scgdwmbkОценок пока нет
- Chem 3Документ23 страницыChem 3陳家寶Оценок пока нет
- Name: Mantara, Moh - Fahrky A. Section: BS CRIM 3-CHARLIE: 4 2 3 (S) 3 (G) 2 (G) 2 (G)Документ3 страницыName: Mantara, Moh - Fahrky A. Section: BS CRIM 3-CHARLIE: 4 2 3 (S) 3 (G) 2 (G) 2 (G)Farks MantaraОценок пока нет
- Polyprotic Acids and Bases: Very Important!: + 2 3 A 3 4 + 2 4 - A1 - 3 2 4 - + 4 2-A2 - 8 4 2 - + 4 3 - A3 - 13Документ18 страницPolyprotic Acids and Bases: Very Important!: + 2 3 A 3 4 + 2 4 - A1 - 3 2 4 - + 4 2-A2 - 8 4 2 - + 4 3 - A3 - 13Anas SaadОценок пока нет
- Lecture 4 - Strength of Acids and Bases IIДокумент18 страницLecture 4 - Strength of Acids and Bases IIjanetluhangah0526Оценок пока нет
- Formula Sheet - Ony PDFДокумент5 страницFormula Sheet - Ony PDFSana ImamОценок пока нет
- AlkenesДокумент30 страницAlkenesapi-3734333Оценок пока нет
- Chap 11-New PDFДокумент18 страницChap 11-New PDF임준현Оценок пока нет
- NOTES ElectrochemistryДокумент30 страницNOTES ElectrochemistryAlexander LeeОценок пока нет
- Sri Venkateshwara Vidhyalayaa Higher Secondary School: 1. Define R ValueДокумент3 страницыSri Venkateshwara Vidhyalayaa Higher Secondary School: 1. Define R ValueHari HaranОценок пока нет
- 2018 A Level Solutions (Compiled)Документ38 страниц2018 A Level Solutions (Compiled)Faith SeahОценок пока нет
- PRESENTASI - Salt Hydrolysis and ExercisesДокумент34 страницыPRESENTASI - Salt Hydrolysis and ExercisesSalim Sanjaya100% (1)
- Chapter5-Kesetimbangan Asam BasaДокумент115 страницChapter5-Kesetimbangan Asam BasaAnnisah MardiyyahОценок пока нет
- Chapter21 PDFДокумент12 страницChapter21 PDFMauryaОценок пока нет
- 8.3 Solving Equilibrium Problems For Bases (SCH4U)Документ3 страницы8.3 Solving Equilibrium Problems For Bases (SCH4U)Michelle NgОценок пока нет
- Final Exam Equations, Constants and TablesДокумент4 страницыFinal Exam Equations, Constants and Tablessammy alanОценок пока нет
- Hydrocarbon To Amines Short NotesДокумент31 страницаHydrocarbon To Amines Short NotesMd AmanОценок пока нет
- Biomass Gasification in Afluidized Bed Reactor (ASPEN SimДокумент22 страницыBiomass Gasification in Afluidized Bed Reactor (ASPEN Simapi-3799861Оценок пока нет
- Lesson 1Документ45 страницLesson 1Pepa GomezОценок пока нет
- 0 0 +1 - 1 Oss of Lectron, Xidation, Educing Gent Ain of Lectron, EductionДокумент6 страниц0 0 +1 - 1 Oss of Lectron, Xidation, Educing Gent Ain of Lectron, EductionTheresaОценок пока нет
- Organics: Oxidation: Organic Molecule CO,, (Complete Mineralization)Документ13 страницOrganics: Oxidation: Organic Molecule CO,, (Complete Mineralization)SantJhaОценок пока нет
- Chemical ReactionДокумент23 страницыChemical Reactionedward comboyОценок пока нет
- 2 Acid Base EquilibriumДокумент30 страниц2 Acid Base Equilibriumu3537671Оценок пока нет
- Aqueous Solution and Chemical EquilibriaДокумент27 страницAqueous Solution and Chemical EquilibriaS. MartinezОценок пока нет
- Assignment Ionic Equilibrium JH Sir-2626Документ34 страницыAssignment Ionic Equilibrium JH Sir-2626d anjilappa100% (1)
- Ch-8:-Redox Reactions: (Pottasium Ferricinite) (Pottasium Ferrocinite)Документ21 страницаCh-8:-Redox Reactions: (Pottasium Ferricinite) (Pottasium Ferrocinite)PATEL AUM S.Оценок пока нет
- Dhoom # 9 Haloalkane & Haloarene in One Shot (10.6.2020)Документ156 страницDhoom # 9 Haloalkane & Haloarene in One Shot (10.6.2020)Jeet RathodОценок пока нет
- 05oxidationnum Half ReactionsДокумент5 страниц05oxidationnum Half ReactionsYesh KumarОценок пока нет
- Exercises: Significant Figures and LogarithmsДокумент15 страницExercises: Significant Figures and LogarithmsSarah ChoiОценок пока нет
- Notes 3 (Kinetic Examples) 12Документ12 страницNotes 3 (Kinetic Examples) 12Sharon FonsecaОценок пока нет
- Reagent Chemistry Jeet Sir FinalДокумент217 страницReagent Chemistry Jeet Sir Finalallenclass11workОценок пока нет
- Ionic EquilibriumДокумент34 страницыIonic EquilibriumTanayОценок пока нет
- Organometallics and Catalysis - IVДокумент36 страницOrganometallics and Catalysis - IVankit guptaОценок пока нет
- Electrochemistry and NanomaterialsДокумент76 страницElectrochemistry and NanomaterialsCharles Arthel ReyОценок пока нет
- 100S120 CS16 IoyДокумент34 страницы100S120 CS16 Ioyb101112154Оценок пока нет
- Redox ReviewДокумент20 страницRedox Reviewapi-3706290100% (1)
- Sample Paper-02 Chemistry (Theory) Class - XI Time Allowed: 3 Hours Answers Maximum Marks: 70Документ8 страницSample Paper-02 Chemistry (Theory) Class - XI Time Allowed: 3 Hours Answers Maximum Marks: 70Yt Status WorldОценок пока нет
- LU 5.1 ElectrochemistryДокумент32 страницыLU 5.1 ElectrochemistryNurAkila Mohd YasirОценок пока нет
- Acidity of H X Compounds No SolventДокумент19 страницAcidity of H X Compounds No SolventBRUNO RAMOS DE LIMAОценок пока нет
- ApformdataДокумент2 страницыApformdataspatters123Оценок пока нет
- Formula Sheet For Grade 11 Chemistry CanadaДокумент2 страницыFormula Sheet For Grade 11 Chemistry CanadaS DemelloОценок пока нет
- Temperature Dependence: Change in G - (Change in T) X SДокумент5 страницTemperature Dependence: Change in G - (Change in T) X SShahday BayanОценок пока нет
- Combustion: Flame Theory and Heat Produced: Arthur Anconetani Oscar Castillo Everett HendersonДокумент29 страницCombustion: Flame Theory and Heat Produced: Arthur Anconetani Oscar Castillo Everett HendersonTommy Cha Yee WenОценок пока нет
- Critical Evaluation of Some Equilibrium Constants Involving Organophosphorus ExtractantsОт EverandCritical Evaluation of Some Equilibrium Constants Involving Organophosphorus ExtractantsОценок пока нет
- Critical Evaluation of Equilibrium Constants Involving 8-Hydroxyquinoline and Its Metal Chelates: Critical Evaluation of Equilibrium Constants in Solution: Part B: Equilibrium Constants of Liquid-Liquid Distribution SystemsОт EverandCritical Evaluation of Equilibrium Constants Involving 8-Hydroxyquinoline and Its Metal Chelates: Critical Evaluation of Equilibrium Constants in Solution: Part B: Equilibrium Constants of Liquid-Liquid Distribution SystemsОценок пока нет
- Cross Rate and Merchant RateДокумент26 страницCross Rate and Merchant RateDivya NadarajanОценок пока нет
- EE2401 Power System Operation and ControlДокумент93 страницыEE2401 Power System Operation and ControlPrasanth GovindarajОценок пока нет
- Market Pulse Q4 Report - Nielsen Viet Nam: Prepared by Nielsen Vietnam February 2017Документ8 страницMarket Pulse Q4 Report - Nielsen Viet Nam: Prepared by Nielsen Vietnam February 2017K57.CTTT BUI NGUYEN HUONG LYОценок пока нет
- Biology Project Cronary Heart Diseas (CHD)Документ7 страницBiology Project Cronary Heart Diseas (CHD)احمد المغربي50% (2)
- IS 4049-Part-IДокумент5 страницIS 4049-Part-ISrinivas KadivetiОценок пока нет
- Case Study LenovoДокумент10 страницCase Study LenovoGOHAR GHAFFARОценок пока нет
- Soft Tissue SarcomaДокумент19 страницSoft Tissue SarcomaEkvanDanangОценок пока нет
- TFU-Risk Assessment RA-11 - Use of Grooving & Threading MachinesДокумент1 страницаTFU-Risk Assessment RA-11 - Use of Grooving & Threading Machinesarshin wildanОценок пока нет
- Top 6 Beginner Work Out MistakesДокумент4 страницыTop 6 Beginner Work Out MistakesMARYAM GULОценок пока нет
- Cat 880620 R11Документ60 страницCat 880620 R11pawelprusОценок пока нет
- The Coca-Cola Company - Wikipedia, The Free EncyclopediaДокумент11 страницThe Coca-Cola Company - Wikipedia, The Free EncyclopediaAbhishek ThakurОценок пока нет
- DDEV SPICES PVT LTD (Product List)Документ1 страницаDDEV SPICES PVT LTD (Product List)jaymin zalaОценок пока нет
- Evidence Based DesignДокумент4 страницыEvidence Based Designmartinadam82Оценок пока нет
- Minerals and Resources of IndiaДокумент11 страницMinerals and Resources of Indiapartha100% (1)
- 5000mah Mi Power Bank 2 - PDFДокумент6 страниц5000mah Mi Power Bank 2 - PDFManuel Jesús Fernández lavadoОценок пока нет
- Additional Activity 3 InsciДокумент3 страницыAdditional Activity 3 InsciZophia Bianca BaguioОценок пока нет
- Ancamine Teta UsДокумент2 страницыAncamine Teta UssimphiweОценок пока нет
- Do Statins Lower TestosteroneДокумент3 страницыDo Statins Lower TestosteroneNandia SeptiyoriniОценок пока нет
- Withania DSC PDFДокумент6 страницWithania DSC PDFEfsha KhanОценок пока нет
- Baxshin LABORATORY: Diagnostic Test and AnalysisДокумент1 страницаBaxshin LABORATORY: Diagnostic Test and AnalysisJabary HassanОценок пока нет
- The Preparation of Culture MediaДокумент7 страницThe Preparation of Culture MediaNakyanzi AngellaОценок пока нет