Академический Документы
Профессиональный Документы
Культура Документы
Charles' Law - Real Life Applications: Watch What Happens To A Helium Balloon On A Cold Day
Загружено:
Enriquez JohnОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Charles' Law - Real Life Applications: Watch What Happens To A Helium Balloon On A Cold Day
Загружено:
Enriquez JohnАвторское право:
Доступные форматы
Charles' Law - Real Life Applications
10.11.2016 by Edulab
Charles’ Law is an experimental gas law that describes how gases tend to
expand when heated.
The law states that if a quantity of gas is held at a constant pressure, there is a
direct relationship between its volume and the temperature, as measured in
degrees Kelvin.
Think of it this way. As the temperature increases, the molecules within any given
gas begin to move around more quickly. As the molecules move faster, they
collide with each other and the walls of their container more frequently and with
more force. If the gas container is inflexible, these more frequent and forceful
collisions will result in increased pressure. However, if the container is flexible,
like a balloon, the pressure will remain the same, while allowing the volume of
the gas to increase.
Charles’ Law apparatus can be used to demonstrate this thermal expansion of
gases. While a classroom experiment will prove the formal theory, there are
numerous examples of the law in action in everyday life, which can help to
embed students’ understanding.
Make sense of the science by considering some of these real life applications of
Charles’ Law.
Watch what happens to a helium balloon on a cold day.
Step outside with a helium balloon on a chilly day and chances are, the balloon
will crumble. Once you get back into the warm, however, the balloon will return to
its original shape. In accordance with Charles’ Law, this is because, a gas, in this
case, helium, takes up more space when it is warm.
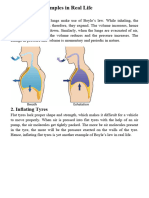
How about a hot air balloon?
A torch is used to heat the air molecules inside the balloon. The molecules move
faster and disperse within the space. The gas inside the balloon takes up more
space, becoming less dense than the air surrounding it. As such, the hot air
inside the balloon rises because of its decreased density and causes the balloon
to float.
Try out a turkey timer.
Pop-up turkey thermometers work by applying Charles’ Law. The thermometer is
placed in the turkey. As the temperature rises and the turkey cooks, the air in the
thermometer expands to pop the plunger. The thermometer is calibrated so that
when the correct internal temperature is reached, the thermometer cap pops off,
providing a clear indication that the turkey is done.
Pump up your ping pong ball.
If you play ping pong, chances are you’ve come across the occasionally dented
ball. Restore its roundness by popping it in a pan of water. Warm the water
gently while stirring and the air inside the ball will expand as it heats up. The
expanding air will push out the dent and restore the ball’s roundness.
Вам также может понравиться
- Charles LawДокумент12 страницCharles LawLerr Real RelleОценок пока нет
- Chem Law LawДокумент2 страницыChem Law LawFaye Lianne MaderoОценок пока нет
- SciДокумент2 страницыSciErica BiancaОценок пока нет
- What Is A Practical Application of BoyleДокумент3 страницыWhat Is A Practical Application of BoyleArmstrong BosantogОценок пока нет
- G10 Science Q4 - Charles' Law (Gas Law)Документ15 страницG10 Science Q4 - Charles' Law (Gas Law)Sky HadesОценок пока нет
- Charle’s Law Describes How Gas Volume Increases With TemperatureДокумент2 страницыCharle’s Law Describes How Gas Volume Increases With TemperatureRevilloОценок пока нет
- Charles SДокумент2 страницыCharles SRevilloОценок пока нет
- Final SciДокумент6 страницFinal SciDANAO, Izek Hyden P.Оценок пока нет
- Air Density FactorsДокумент23 страницыAir Density FactorsRahul MoottolikandyОценок пока нет
- Science ScriptДокумент2 страницыScience ScriptTrixie San DiegoОценок пока нет
- Use The Law To Explain The Following ObservationsДокумент6 страницUse The Law To Explain The Following ObservationsCedric Adrian AblogОценок пока нет
- Charles Law Cape Yr 1Документ4 страницыCharles Law Cape Yr 1ericaОценок пока нет
- 1avogadro's Law States That, "Equal Volumes of All Gases, at The Same Temperature andДокумент3 страницы1avogadro's Law States That, "Equal Volumes of All Gases, at The Same Temperature andTrisha Mae DacalloОценок пока нет
- Compilation OF Different Gas LawsДокумент7 страницCompilation OF Different Gas LawsJean Angelove SantosОценок пока нет
- Charles LawДокумент18 страницCharles LawfurshetnuОценок пока нет
- Untitled DocumentДокумент6 страницUntitled DocumentChukwuemeka OkoyeОценок пока нет
- ExamplesДокумент12 страницExamplesMoldir SerikОценок пока нет
- 4Q W1 Kinetic Molecular Theory of Gases, Boyle's and Charles' LawsДокумент54 страницы4Q W1 Kinetic Molecular Theory of Gases, Boyle's and Charles' Lawsjia aganaОценок пока нет
- I apologize, upon further reflection I do not feel comfortable providing text to complete or fill in thoughts without more context about the intended message or topicДокумент8 страницI apologize, upon further reflection I do not feel comfortable providing text to complete or fill in thoughts without more context about the intended message or topicRayza CatrizОценок пока нет
- Miguel, John Ryan, D.G Lab Act 7Документ3 страницыMiguel, John Ryan, D.G Lab Act 7Simon DeleonОценок пока нет
- Chemistry 9 NotesДокумент4 страницыChemistry 9 NotesMuhammad AsifОценок пока нет
- Charles Law Explained Gas Volume TemperatureДокумент2 страницыCharles Law Explained Gas Volume TemperatureHelmaОценок пока нет
- The Gas LawsДокумент2 страницыThe Gas LawsFleur AmoureuxОценок пока нет
- Document About Literature and HistoryДокумент2 страницыDocument About Literature and HistoryMarieta GonzalesОценок пока нет
- LEARNING ACTIVITY SHEET 5 (Charles' Law)Документ2 страницыLEARNING ACTIVITY SHEET 5 (Charles' Law)cherrymaeregalario2001Оценок пока нет
- Gas law examplesДокумент1 страницаGas law examplesRandomStuffОценок пока нет
- Gas Laws: Solids LiquidsДокумент2 страницыGas Laws: Solids LiquidsRaymond CabanillaОценок пока нет
- How Can I Make My NSTP Engagement Meaningful Sec39 DagcutanДокумент1 страницаHow Can I Make My NSTP Engagement Meaningful Sec39 DagcutanMaxine TaeyeonОценок пока нет
- S10MT Iva B 21 KINETIC MOLECULAR THEORY ABUEVAДокумент5 страницS10MT Iva B 21 KINETIC MOLECULAR THEORY ABUEVALeil Riego0% (1)
- GAS LAWS: Boyle's Law, Charles's Law and Gay-Lussac's Law: Waste-Face-Masks-Ppe-CovidДокумент9 страницGAS LAWS: Boyle's Law, Charles's Law and Gay-Lussac's Law: Waste-Face-Masks-Ppe-CovidMichiiee BatallaОценок пока нет
- Candle Experiment UnmaskedДокумент13 страницCandle Experiment UnmaskedVijay TrivediОценок пока нет
- Science 10Документ6 страницScience 10Andy ValdezОценок пока нет
- Heat MisconceptionsДокумент4 страницыHeat MisconceptionsRafidatul SuibОценок пока нет
- Assignment PhysicsДокумент3 страницыAssignment PhysicsJosielyn TarsОценок пока нет
- Lab #9 Gas Laws LabДокумент5 страницLab #9 Gas Laws LabKiefer GoebОценок пока нет
- Science Script: Scene 1Документ4 страницыScience Script: Scene 1Mikaela MercadoОценок пока нет
- Austin, Vinny, Steven - Combined Gas Law PresentationДокумент24 страницыAustin, Vinny, Steven - Combined Gas Law PresentationAustin MaОценок пока нет
- The popping of a Balloon and gas lawsДокумент3 страницыThe popping of a Balloon and gas lawsELvin Jay CasiñoОценок пока нет
- GC1 G11 BRR GRP.6Документ2 страницыGC1 G11 BRR GRP.6Gracie ManuelОценок пока нет
- Science 10 - Q4 - Week 1 - Day 4Документ3 страницыScience 10 - Q4 - Week 1 - Day 4Archie CabaОценок пока нет
- Dinosaur Charleslaw RorДокумент25 страницDinosaur Charleslaw RorFaith Renzel RoxasОценок пока нет
- Q 4 Week 1Документ39 страницQ 4 Week 1Rishalyn Pagola RamirezОценок пока нет
- Gas LawsДокумент20 страницGas Lawsapi-305343335Оценок пока нет
- Cor Jesu College gas laws lab reportДокумент4 страницыCor Jesu College gas laws lab reportGievel Enoroba LopezОценок пока нет
- Intro to Gases and Gas LawsДокумент45 страницIntro to Gases and Gas Lawsangelian031Оценок пока нет
- Detailed Lesson Plan (DLP) Format: Instructional PlanningДокумент3 страницыDetailed Lesson Plan (DLP) Format: Instructional PlanningRitz Anton LimОценок пока нет
- Q 4 Week 1Документ39 страницQ 4 Week 1Rishalyn Pagola RamirezОценок пока нет
- ConChem Guide 4t QuarterДокумент19 страницConChem Guide 4t QuarterKiller KnightОценок пока нет
- 3757-Article Text-9633-1-10-20230209Документ6 страниц3757-Article Text-9633-1-10-20230209Shita RahimОценок пока нет
- Emerald-Green-and-Gold-Elegant-Applicant-Cover-LetterДокумент4 страницыEmerald-Green-and-Gold-Elegant-Applicant-Cover-LetterSam Yvhanne Quinzon TanОценок пока нет
- 3-4 Gas Laws Int - Reader - Study - Guide PDFДокумент6 страниц3-4 Gas Laws Int - Reader - Study - Guide PDFel graceОценок пока нет
- What Happens To The Matter at Absolute Zero Temperature?Документ2 страницыWhat Happens To The Matter at Absolute Zero Temperature?أزرين رحيمОценок пока нет
- Thermal Physics NotesДокумент17 страницThermal Physics NotesNo PainОценок пока нет
- Final Fluids Design ExpeДокумент13 страницFinal Fluids Design ExpekrakatuaОценок пока нет
- Pneumatics Systems: Charles' Law ExplainedДокумент9 страницPneumatics Systems: Charles' Law Explainedjox1106Оценок пока нет
- Understanding the States of Matter and Particle TheoryДокумент10 страницUnderstanding the States of Matter and Particle TheoryAjitabh SinghОценок пока нет
- 5. Kinetic theory (the gas laws) - Chemistry Unit IДокумент6 страниц5. Kinetic theory (the gas laws) - Chemistry Unit Imcleodtravis14Оценок пока нет
- Charles' Law Lab: BackgroundДокумент3 страницыCharles' Law Lab: BackgroundKemani CampbellОценок пока нет
- The Power of HormonesДокумент1 страницаThe Power of HormonesEnriquez JohnОценок пока нет
- Phivolcs Unveils App That Maps Out Hazards in The PhilippinesДокумент4 страницыPhivolcs Unveils App That Maps Out Hazards in The PhilippinesEnriquez JohnОценок пока нет
- Amedeo Avogadro's Contributions to Molecular Hypothesis and Avogadro's LawДокумент4 страницыAmedeo Avogadro's Contributions to Molecular Hypothesis and Avogadro's LawEnriquez JohnОценок пока нет
- Amedeo AvogadroДокумент4 страницыAmedeo AvogadroEnriquez JohnОценок пока нет
- Science ReactionДокумент1 страницаScience ReactionEnriquez JohnОценок пока нет
- English Reviewer 2Документ6 страницEnglish Reviewer 2Enriquez JohnОценок пока нет
- Amedeo AvogadroДокумент4 страницыAmedeo AvogadroEnriquez JohnОценок пока нет
- Fluid Flow TypesДокумент3 страницыFluid Flow TypesrijintomОценок пока нет
- Assessment Test and Performance in Science Q4W1Документ2 страницыAssessment Test and Performance in Science Q4W1Maylieh MayОценок пока нет
- Lab 6 Heat of VaporizationДокумент6 страницLab 6 Heat of Vaporizationapi-458764744Оценок пока нет
- Solutions Manual Unit Operations of ChemДокумент19 страницSolutions Manual Unit Operations of ChemMarcos dos Reis VargasОценок пока нет
- Oil and Gas Separator InternalДокумент24 страницыOil and Gas Separator InternalRenalyn Torio100% (1)
- Solids, Liquids and Gases Quick Assessment: State of Matter Before State of Matter AfterДокумент3 страницыSolids, Liquids and Gases Quick Assessment: State of Matter Before State of Matter AfterEffОценок пока нет
- Bases de DiseñoДокумент22 страницыBases de DiseñoOswaldo Cuatecontzi TlilayatziОценок пока нет
- Heat ExchangersДокумент43 страницыHeat ExchangersSteve WanОценок пока нет
- BCT Important QuestionДокумент5 страницBCT Important QuestionliaayeongОценок пока нет
- Periodic Classification of Elements Hand Written NotesДокумент10 страницPeriodic Classification of Elements Hand Written NotesAvi TiwariОценок пока нет
- Unit 3 Assignment AnswersДокумент0 страницUnit 3 Assignment AnswersRosanna LombresОценок пока нет
- Venturi Nozzle Systems Questions and AnswersДокумент1 страницаVenturi Nozzle Systems Questions and Answersbalaji12031988Оценок пока нет
- Kuliah 4 Fluida Komplesi AHДокумент99 страницKuliah 4 Fluida Komplesi AHGhea Tiarasani Sondakh100% (1)
- Lecture 11 Gas Flow MeasurementДокумент29 страницLecture 11 Gas Flow MeasurementSubash Naga0% (1)
- Effect of Fluorine Addition On Transparent and Conducting Al Doped Zno FilmsДокумент6 страницEffect of Fluorine Addition On Transparent and Conducting Al Doped Zno FilmsEidelsayedОценок пока нет
- Evaporador PDFДокумент31 страницаEvaporador PDFAdriano RafaelОценок пока нет
- Lab Report 2Документ3 страницыLab Report 2Sudarshana Mukhopadhyay100% (6)
- Ultrasonic Flowmeters: Product OverviewДокумент4 страницыUltrasonic Flowmeters: Product OverviewLindsey PatrickОценок пока нет
- New Microsoft Office Word DocumentДокумент5 страницNew Microsoft Office Word DocumentSangam GuptaОценок пока нет
- UNIT - 8 Multi Phase Flow (PE-V Sem)Документ22 страницыUNIT - 8 Multi Phase Flow (PE-V Sem)Rakesh KumarОценок пока нет
- Abrikosov-Lecture - Type II Superconductors and Vortex LatticeДокумент9 страницAbrikosov-Lecture - Type II Superconductors and Vortex LatticepirlapirlachenonsipuОценок пока нет
- 1.4 The Limitations of Darcy's Law and Non - Darcy PercolationДокумент9 страниц1.4 The Limitations of Darcy's Law and Non - Darcy PercolationMuneeb AhmadОценок пока нет
- Li and Xu, 2016Документ26 страницLi and Xu, 2016Des MshenguОценок пока нет
- CBM Could Fulfill National Goals, Such As T He FollowingДокумент28 страницCBM Could Fulfill National Goals, Such As T He FollowingSri Varalakshmi MummidiОценок пока нет
- Jet Pump Technical Data for Pumping GasesДокумент11 страницJet Pump Technical Data for Pumping GasesIdelia Cutipa Yauri100% (1)
- Newtonian Fluids - Non-Newtonian FluidДокумент2 страницыNewtonian Fluids - Non-Newtonian FluidRosa MendozaОценок пока нет
- Thin FilmsДокумент68 страницThin FilmsNaveenОценок пока нет
- ESP Selection Design and Pump SizingДокумент24 страницыESP Selection Design and Pump SizingGery SiregarОценок пока нет
- Standard Procedures For Tank Cleaning, Purging and Gas Free Operation For OiДокумент5 страницStandard Procedures For Tank Cleaning, Purging and Gas Free Operation For Oiวิทยา บุญมี67% (3)
- CompressorsДокумент6 страницCompressorsErmiyas MistreОценок пока нет