Академический Документы
Профессиональный Документы
Культура Документы
IO Alcohol
Загружено:
Dr Churamani Dev Mishra0 оценок0% нашли этот документ полезным (0 голосов)
59 просмотров13 страницThe document discusses bio-alcohol (ethanol) production from various feedstocks like sugarcane, corn, and cellulosic materials. Ethanol production involves fermenting sugars from these feedstocks with yeast to produce ethanol. The process includes milling, cooking, cooling, fermentation, distillation, dehydration, and denaturing to produce anhydrous ethanol suitable for use as a fuel. Ethanol has advantages as a motor fuel including high octane rating and ability to replace lead, but is not suitable for diesel engines due to low cetane rating. Coproducts of ethanol production like distillers grains and carbon dioxide also have market value.
Исходное описание:
Оригинальное название
bio-alcohol-200202030927
Авторское право
© © All Rights Reserved
Доступные форматы
PDF, TXT или читайте онлайн в Scribd
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документThe document discusses bio-alcohol (ethanol) production from various feedstocks like sugarcane, corn, and cellulosic materials. Ethanol production involves fermenting sugars from these feedstocks with yeast to produce ethanol. The process includes milling, cooking, cooling, fermentation, distillation, dehydration, and denaturing to produce anhydrous ethanol suitable for use as a fuel. Ethanol has advantages as a motor fuel including high octane rating and ability to replace lead, but is not suitable for diesel engines due to low cetane rating. Coproducts of ethanol production like distillers grains and carbon dioxide also have market value.
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PDF, TXT или читайте онлайн в Scribd
0 оценок0% нашли этот документ полезным (0 голосов)
59 просмотров13 страницIO Alcohol
Загружено:
Dr Churamani Dev MishraThe document discusses bio-alcohol (ethanol) production from various feedstocks like sugarcane, corn, and cellulosic materials. Ethanol production involves fermenting sugars from these feedstocks with yeast to produce ethanol. The process includes milling, cooking, cooling, fermentation, distillation, dehydration, and denaturing to produce anhydrous ethanol suitable for use as a fuel. Ethanol has advantages as a motor fuel including high octane rating and ability to replace lead, but is not suitable for diesel engines due to low cetane rating. Coproducts of ethanol production like distillers grains and carbon dioxide also have market value.
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PDF, TXT или читайте онлайн в Scribd
Вы находитесь на странице: 1из 13
BIO-ALCOHOL
Dr. Ajay Singh Lodhi
Assistant Professor
College of Agriculture, Balaghat
Jawahar Lal Krishi Vishwa Vidyalaya, Jabalpur (M.P.)
Bio-Alcohol
Ethyl alcohol, known as alcohol and technically termed
ethanol is a colourless and flammable liquid with a
chemical formula C2H5OH.
It is the alcoholic product of fermenting the sugars in
natural raw materials with yeast. The natural raw
materials include vegetable matter, growing crops, farm
waste, waste organic products (straw and saw dust),
molasses, wastes of paper and pulp industries etc.
From this annually renewed store of raw materials,
ethanol can be readily produced in quantities sufficient
to meet the world demand. It thus has attributes of
renewal in nature.
ALCOHOL AS AUTOMOBILE FUEL
Ethanol offers certain advantages if used as an
automobile engine fuel:
It is an antiknock fuel and has the ability to stand very
high compression ratios.
It is a high octane, water-free alcohol. Octane rating is
defined as the fuel’s ability to resist engine knock.
Ethanol is both a fuel and an octane enhancer.
It is an ideal replacement for lead.
It has a high latent heat of vaporisation. This property
can be utilised to achieve lower charge temperature
during induction for higher charge density. Hence,
higher volumetric efficiency is obtained.
It tends to produce less carbon deposits than normal
petrol and the deposits are softer and easier to remove.
Its lower calorific value, higher viscosity, greater surface
tension and hygroscopic nature are some of the
difficulties in its use as a complete fuel in present day
combustion engines.
It can, however, be mixed with other fuel or fuel
mixtures so as to impart to the resulting blend some of
its important properties namely higher compression
operation without knock, cleaner combustion and cooler
engine operation.
Ethanol has the potential as the alternative fuel to solve
the problems of limited source of fossil fuels.
Ethanol is most commonly used as a 10 percent blend
with petrol known as gasohol that can be burned in
unmodified automotive engines.
UNSUITABILITY OF ETHANOL IN DIESEL ENGINES
Diesel engines operate on an entirely different combustion
principle in comparison to spark ignition engines. It relies on
the fuel to self ignite from an increased pressure and
temperature of the compression cycle.
Diesel fuel has a high cetane rating. It also has a low octane
rating. The converse is true for ethanol, which is the major
problem in the use of ethanol in diesel engines.
Ethanol can be burned in diesel engines, but because it
resists self ignition, it can have accompanying knock and
engine stresses that may be unacceptable due to noise and
lower engine life.
Cetane-enhancer additives are available but expensive and
of questionable performance. Also, phase separation of diesel
and ethanol is more pronounced than with petrol blends,
which limits its performance during cold weather. Hence,
ethanol is not suitable to use as a fuel in diesel engine
systems.
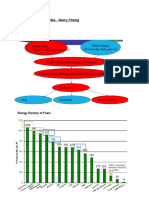
FEEDSTOCKS FOR ETHANOL PRODUCTION
Ethanol can be produced by the fermentation of any
feedstock that contains sugar or starches. It can be
produced from cellulose materials that can be converted
into fermentable sugar.
These three groups of biomass are as follows:
(i) sugars (sugar beets, sugarcane, sweet sorghum, fruits);
(ii) starches (small grains such as corn and wheat,
potatoes, cassava); and
(iii) cellulose (wood, solid waste, agricultural residues).
Sugar crops contain monosaccharide forms of sugar such
as glucose. This can readily be fermented into alcohol by
yeast with no intermediate processing.
Starch crops contain sugar units that are tied together
in long chains. Yeast cannot use these disaccharide
forms of sugar until the starch chains are converted into
individual six-carbon groups such as glucose or fructose.
This conversion process can be done fairly simply by the
use of cooking in a dilute acid solution or reacting the
starch with thermophilic (heat tolerant) enzymes.
Cellulose crops contain chemicals called polysaccharides.
These chains must be broken down to release the sugar.
Breaking the chemical bonds of cellulose is more
complicated than breaking down starch to simple sugar.
The conversion is typically done physically (milling or
heat treatment) or chemically (basic or acid reactions).
ETHANOL PRODUCTION PROCESS
Ethanol production proceeds as follows:
(i) formation of a fermentable sugar solution;
(ii) fermentation of the sugar solution to ethanol; and
(iii) separation of ethanol from other process ingredients.
THE ETHANOL PRODUCTION PROCESS IS
OUTLINED AS FOLLOWS:
1. Milling: Feedstock is first passed through hammer
mills and then it is ground into a fine powder.
2. Sterilisation: If distressed or damaged crop are used,
sterilisation facilities are included to avoid biological
contamination.
3. Cooking: Enzymes (alpha-amylase) and water are
added to form a mash that is cooked for about 30 min.
Heat is applied at this stage to enable liquefaction.
4. Cooling: The mash from the cooker is cooled. The
secondary enzyme (glucoamylase) is added to convert the
liquefied mash to fermentable sugars (dextrose).
5. Fermentation: Yeast is added to the mash to ferment
the sugars to ethanol and carbon dioxide. Mash ferments
for about 2 days, during which the sugar is converted to
alcohol.
6. Distillation:
❑ The fermented mash, now called ‘‘beer’’ contains about
10% alcohol as well as the nonfermentable solids.
❑ Initially the mass is introduced into the first-stage
distillation column that is regulated to maintain a
temperature just a little higher than the boiling point of
ethanol. Alcohol vaporises and ascends out of the
column. Liquid water mixed with residual grain and
yeast (distilliers grain) flows down and out.
❑ The vaporised alcohol (now about 50 percent) is passed
through a second distillation column. It concentrates the
alcohol content to 95 percent (e.g. 190 proof).
❑ Proof is a measure of ethanol content. One percent
ethanol content equals 2 proofs.
7. Dehydration:
Since the alcohol from the second-stage column contains
5% water, which is unusual for commercial gasohol, it
must be totally dehydrated.
Benzene and petrol are common additives in a further
process to separate the residual water and produce
anhydrous ethanol (100 percent or 200 proof alcohol).
8. Denaturing:
Ethanol that will be used for fuel is then denatured with
a small amount (2–5%) of some product, like petrol, to
make it unfit for human consumption.
9. Fuel:
The denatured anhydrous alcohol is transported to the
gasohol market. It is blended with nonleaded petrol at
the ratio of 10 percent ethanol to 90 percent petrol.
10. Coproducts:
Two primary coproducts of ethanol production are
distillers grain and carbon dioxide. Both of which have
market value.
Distiller grain consists of the protein value of corn,
yeast, fibre and water discharge from the first-stage
distillation column. The distillers grain has a high
protein content of about 27 percent. Therefore, it makes
an excellent animal feedstock supplement. It is
competitive with soybean meal, which is about 44
percent protein.
Carbon dioxide (CO2) that evolves from the fermentation
process can be used in grain storage silos to retard
spoilage.
Thank You
Вам также может понравиться
- Liquid BiofuelДокумент9 страницLiquid BiofueltrishaОценок пока нет
- Production of EthanolДокумент5 страницProduction of EthanolGhazanfer AliОценок пока нет
- Alternative Fuels - Alcohols by VivekДокумент38 страницAlternative Fuels - Alcohols by Vivekprakash11111Оценок пока нет
- Ethyl Alcohol or Ethanol Production From Molasses by FermentationДокумент3 страницыEthyl Alcohol or Ethanol Production From Molasses by FermentationQasim ShahzadОценок пока нет
- Power Alcohol and BiodieselДокумент12 страницPower Alcohol and BiodieselVenkatesh Perumal MОценок пока нет
- 11.6 Further Thermochemical Processes: 374 Biomass and BiofuelsДокумент24 страницы11.6 Further Thermochemical Processes: 374 Biomass and BiofuelsEswahyudiKurniawanОценок пока нет
- Bioethanol Technology: by DR Sammer FatimaДокумент20 страницBioethanol Technology: by DR Sammer FatimaAlina RajputОценок пока нет
- Alternative Fuels: by Jubair, Thohura and MukhtarДокумент11 страницAlternative Fuels: by Jubair, Thohura and MukhtarSammey Jubair AhmedОценок пока нет
- B.SC III Year Vi Sem Microbial TechnologyДокумент119 страницB.SC III Year Vi Sem Microbial TechnologySrilatha LathaОценок пока нет
- Ethanol Tu BapДокумент4 страницыEthanol Tu BapKhanhHoaNPОценок пока нет
- 6-Shang XueyingДокумент45 страниц6-Shang Xueyingrushilraju29Оценок пока нет
- Bio EthanolДокумент19 страницBio EthanolIbrahim DibalОценок пока нет
- RnwbleДокумент8 страницRnwbleYasin OmaryОценок пока нет
- Industrial AlcoholДокумент15 страницIndustrial AlcoholDave Anthony LauroОценок пока нет
- Ethanol and Its HistoryДокумент6 страницEthanol and Its HistoryAlgin Mark DaquioagОценок пока нет
- For Frequently Asked Questions On Ethanol, Click HereДокумент3 страницыFor Frequently Asked Questions On Ethanol, Click HerefidoborderОценок пока нет
- ANIKETДокумент12 страницANIKETyjyk gkcukuОценок пока нет
- Grade 11 Chemistry Week 7 Lesson 2Документ4 страницыGrade 11 Chemistry Week 7 Lesson 2RealGenius (Carl)Оценок пока нет
- Industrial Training 1Документ32 страницыIndustrial Training 1KumarShiveshОценок пока нет
- Salman Khan (Trek)Документ41 страницаSalman Khan (Trek)slmnkhn1988Оценок пока нет
- Modified Ehanol As Aternate FuelДокумент13 страницModified Ehanol As Aternate Fuelਸਰਦਾਰ Supreet SinghОценок пока нет
- Che 243Документ47 страницChe 243Carlyn HarfordОценок пока нет
- Form 5 Chemistry PBLДокумент6 страницForm 5 Chemistry PBLKronix GamingОценок пока нет
- 3 EthanolДокумент14 страниц3 EthanolVISHAL PATELОценок пока нет
- BioethanolДокумент12 страницBioethanolRuvimbo ShokoОценок пока нет
- Biochemical FuelsДокумент13 страницBiochemical FuelsAnonymous na314kKjOAОценок пока нет
- Ethanol Fermentation, Also Called Alcoholic Fermentation, Is AДокумент2 страницыEthanol Fermentation, Also Called Alcoholic Fermentation, Is AtapiwaОценок пока нет
- BT (H) - VI-Industrial and Environmetal Microbiology-1Документ15 страницBT (H) - VI-Industrial and Environmetal Microbiology-1Antonio LopezОценок пока нет
- HW0021 Manufacture of EthanolДокумент5 страницHW0021 Manufacture of Ethanollily augustОценок пока нет
- Presentation 1Документ27 страницPresentation 1Gabriel AyauОценок пока нет
- Production of Bioethanol From Empty Fruit Bunch (Efb) of Oil PalmДокумент26 страницProduction of Bioethanol From Empty Fruit Bunch (Efb) of Oil PalmcelestavionaОценок пока нет
- Complet ProДокумент64 страницыComplet ProGabriel AyauОценок пока нет
- BABS 1202 Study NotesДокумент82 страницыBABS 1202 Study NotesyishadabeeОценок пока нет
- Liquid Fuels For Transportation: Biofuels Are A Wide Range of Fuels Which Are in Some Way Derived FromДокумент11 страницLiquid Fuels For Transportation: Biofuels Are A Wide Range of Fuels Which Are in Some Way Derived FromsadhanasvceОценок пока нет
- Ethanol From MolassesДокумент6 страницEthanol From MolassesSparrowGospleGilbertОценок пока нет
- Can Be Used in SI-engines, To Increase The Resistance To Knock Not Suitable For CI-engines DueДокумент21 страницаCan Be Used in SI-engines, To Increase The Resistance To Knock Not Suitable For CI-engines Duesagar shinkarОценок пока нет
- The Company and The Market 1.1. Summary of The Proceedings of The Business PlanДокумент16 страницThe Company and The Market 1.1. Summary of The Proceedings of The Business PlanPatricia YennyОценок пока нет
- Ethanol DistillationДокумент28 страницEthanol DistillationHoongОценок пока нет
- Ethanol PlantДокумент17 страницEthanol Plantnalindwivedi41Оценок пока нет
- BioethanolДокумент13 страницBioethanolvkchintu53Оценок пока нет
- Production of BioethanolДокумент22 страницыProduction of BioethanolAniket Chaki100% (1)
- PERP Program - Ethanol New Report AlertДокумент8 страницPERP Program - Ethanol New Report AlertGraciaVelitarioОценок пока нет
- Mepe14-Bio Fuels: Unit - IiiДокумент22 страницыMepe14-Bio Fuels: Unit - IiiKrishna GummadiОценок пока нет
- BioethanolДокумент29 страницBioethanolVikas JanuОценок пока нет
- Bioethanol: Roselen, Wanxi, EugeniaДокумент36 страницBioethanol: Roselen, Wanxi, EugeniaGhulam RabbaniОценок пока нет
- Bioprocessing EngineeringДокумент9 страницBioprocessing EngineeringKuo SarongОценок пока нет
- SEMINAR (Autosaved)Документ13 страницSEMINAR (Autosaved)Abhishek PuriОценок пока нет
- Chemistry: Single Bonds PhotosynthesisДокумент17 страницChemistry: Single Bonds PhotosynthesisJerusha JemmottОценок пока нет
- Biofuel: Biofuels Are A Wide Range of Fuels Which Are in Some Way Derived FromДокумент16 страницBiofuel: Biofuels Are A Wide Range of Fuels Which Are in Some Way Derived Fromsuniyoti100% (1)
- Science Assignment 2Документ2 страницыScience Assignment 2christian sanchezОценок пока нет
- Alcohol Fermentation: Prepared By:Prachi Patel Sem:Iii Saurashtra University RajkotДокумент22 страницыAlcohol Fermentation: Prepared By:Prachi Patel Sem:Iii Saurashtra University RajkotBerlian SitorusОценок пока нет
- Ethanol: Done By: Shalenne Bahadur Subject: Chemistry Class: U6SДокумент10 страницEthanol: Done By: Shalenne Bahadur Subject: Chemistry Class: U6SVarune kingОценок пока нет
- Fermentation IndustriesДокумент17 страницFermentation IndustriesVenkata RathnamОценок пока нет
- Production of Ethanol - MicrobiologyДокумент5 страницProduction of Ethanol - Microbiologysubbaraau heghdeОценок пока нет
- Other Renewable Energy SourcesДокумент54 страницыOther Renewable Energy SourcesFrancis UmbaoОценок пока нет
- Process Design Considerations For The Production of Ethanol From Corn Stover-FinalДокумент19 страницProcess Design Considerations For The Production of Ethanol From Corn Stover-FinalNikhil wadicharОценок пока нет
- Gasohol ProductionДокумент16 страницGasohol ProductionPrashant Kumar82% (11)
- Cellulosic Ethanol As A Viable Essay)Документ3 страницыCellulosic Ethanol As A Viable Essay)Petrell VereenОценок пока нет
- Biodiesel Production101: Homebrew Edition: A Do It Yourself Guide to Produce Biodiesel on Your BackyardОт EverandBiodiesel Production101: Homebrew Edition: A Do It Yourself Guide to Produce Biodiesel on Your BackyardОценок пока нет
- Ethanol Fuel Learn to Make and Use Ethanol to Power Your VehiclesОт EverandEthanol Fuel Learn to Make and Use Ethanol to Power Your VehiclesОценок пока нет
- IO FuelsДокумент25 страницIO FuelsDr Churamani Dev MishraОценок пока нет
- Principles of Wind ErosionДокумент58 страницPrinciples of Wind ErosionDr Churamani Dev MishraОценок пока нет
- Iodiesel Roduction RocessesДокумент22 страницыIodiesel Roduction RocessesDr Churamani Dev MishraОценок пока нет
- Centrifugal Pump: Hardik Goswami Mechanical Engg. Department, SOT PDPU, GandhinagarДокумент40 страницCentrifugal Pump: Hardik Goswami Mechanical Engg. Department, SOT PDPU, GandhinagarDr Churamani Dev MishraОценок пока нет
- Lecture 9Документ7 страницLecture 9Dr Churamani Dev MishraОценок пока нет
- Protected Cultivation and Secondary AgricultureДокумент24 страницыProtected Cultivation and Secondary AgricultureDr Churamani Dev MishraОценок пока нет
- Separation: ScalpingДокумент30 страницSeparation: ScalpingDr Churamani Dev MishraОценок пока нет
- 1grassedwaterways 200120062308Документ60 страниц1grassedwaterways 200120062308Dr Churamani Dev MishraОценок пока нет
- Protected Cultivation and Secondary AgricultureДокумент24 страницыProtected Cultivation and Secondary AgricultureDr Churamani Dev MishraОценок пока нет
- Seminar Presentation Dual FuelДокумент20 страницSeminar Presentation Dual FuelRohan Patil100% (8)
- Diesel COLD FLOW IMPROVER Fuel AdditiveДокумент2 страницыDiesel COLD FLOW IMPROVER Fuel AdditiveBruce WappmanОценок пока нет
- It Report On Desiel and Coolant in TrainsДокумент39 страницIt Report On Desiel and Coolant in Trainsslash graphicsОценок пока нет
- Elimination of Carbon Particles From Exhaust GasДокумент4 страницыElimination of Carbon Particles From Exhaust GasParamesh WaranОценок пока нет
- Pollution Control Law Series 2021 7th EditionДокумент1 270 страницPollution Control Law Series 2021 7th EditionViju K G100% (1)
- Environmental Management System Questionnaire: I.S. EN ISO 14001:2015Документ23 страницыEnvironmental Management System Questionnaire: I.S. EN ISO 14001:2015HUMA TARIQОценок пока нет
- Thermodynamic Analysis of Diesel Hydrotreating ReactionsДокумент8 страницThermodynamic Analysis of Diesel Hydrotreating ReactionsDana MateiОценок пока нет
- PPD AssignmentДокумент8 страницPPD AssignmentMuhammad Mubashir SiddiquiОценок пока нет
- Terms of Reference For FuelДокумент4 страницыTerms of Reference For FuelMaria Cecille Jamito-MaderaОценок пока нет
- Doba Blend Crude Oil Assay: Whole Crude HEAVY NAPHTHA (300-400 °F)Документ2 страницыDoba Blend Crude Oil Assay: Whole Crude HEAVY NAPHTHA (300-400 °F)Wira DicoОценок пока нет
- Performance Analysis of Ci Engine Fueled With Diesel and Used Vegetable Oil BlendДокумент8 страницPerformance Analysis of Ci Engine Fueled With Diesel and Used Vegetable Oil BlendYohannes AlemuОценок пока нет
- New Holland t7030 t7040 t7050 t7060 Tractor PDF Service ManualДокумент3 061 страницаNew Holland t7030 t7040 t7050 t7060 Tractor PDF Service ManualJose Vinicio50% (4)
- PIUSI CatalogДокумент248 страницPIUSI CatalogRafael JasperОценок пока нет
- Diesel Oil Pump 1Документ26 страницDiesel Oil Pump 1Vraja KisoriОценок пока нет
- DДокумент45 страницDLeon PeterОценок пока нет
- Bio-Fuels: A Blessing in Disguise: O. Surriya, Syeda Sarah Saleem, K. Waqar, A. Gul Kazi and M. ÖztürkДокумент21 страницаBio-Fuels: A Blessing in Disguise: O. Surriya, Syeda Sarah Saleem, K. Waqar, A. Gul Kazi and M. ÖztürkcmuruganОценок пока нет
- 655, 755, 855, 955, 756, and 856 Co-TM1360 Tech. SmallДокумент564 страницы655, 755, 855, 955, 756, and 856 Co-TM1360 Tech. SmallBruceHoward100% (4)
- MRT - Belitung Oil TerminalДокумент26 страницMRT - Belitung Oil TerminalMohammadОценок пока нет
- 11.6 Further Thermochemical Processes: 374 Biomass and BiofuelsДокумент24 страницы11.6 Further Thermochemical Processes: 374 Biomass and BiofuelsEswahyudiKurniawanОценок пока нет
- SOT Super Brochure 10.21Документ2 страницыSOT Super Brochure 10.21dennyОценок пока нет
- Alpaslan Atmanli: Highlights GraphicalabstractДокумент7 страницAlpaslan Atmanli: Highlights GraphicalabstractHiwetОценок пока нет
- NON-EDIBLE OIL (Karanja) IndiaДокумент22 страницыNON-EDIBLE OIL (Karanja) IndiaVaibhav MoonОценок пока нет
- Man 48 60crДокумент456 страницMan 48 60crTran Duy Nam100% (5)
- Kerosene and Gas Oil Burner: One Stage OperationДокумент28 страницKerosene and Gas Oil Burner: One Stage Operationtally3tallyОценок пока нет
- AE - 2marksДокумент20 страницAE - 2marksThiruvasagamoorthy KaОценок пока нет
- Biofuels: Certified Reference StandardsДокумент4 страницыBiofuels: Certified Reference StandardsAlexSОценок пока нет
- Environmental Quality Act 1974 (Act 127) : Regulations, Rules & OrdersДокумент70 страницEnvironmental Quality Act 1974 (Act 127) : Regulations, Rules & OrdersFilzah HidayatiОценок пока нет
- Coconut (Cocos Nucifera L. Arecaceae) in Health Promotion and Disease PDFДокумент7 страницCoconut (Cocos Nucifera L. Arecaceae) in Health Promotion and Disease PDFOctavio Niño OrtegaОценок пока нет
- Gasoil PropertiesДокумент1 страницаGasoil PropertiesQaisar Zahoor AwaanОценок пока нет
- 1998 Zhang Biodegradability ofДокумент8 страниц1998 Zhang Biodegradability ofAlejandro Castro YaruroОценок пока нет