Академический Документы
Профессиональный Документы
Культура Документы
Checkpoint 9.4
Загружено:
Az MYИсходное описание:
Оригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Checkpoint 9.4
Загружено:
Az MYАвторское право:
Доступные форматы
Checkpoint 9.
1. The root-mean-square of a gas at temperature of 300 K is v. What is the temperature of the gas when
the root-mean-square speed increases to 2v?
2. The root-mean-square speed of an ideal gas is c. If the gas is heated at constant volume so that its
pressure increases from p to 2p, determine the new root-mean-square speed of the gas.
3. Six molecules of gas move with speeds of 450 ms -1 , 400 ms-1, 500 ms-1 , 550 ms-1, 450 ms-1 and
500 ms-1. Calculate:
(a) the mean speed of the molecules
(b) the root-mean-square speed of the molecules.
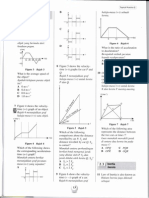
4. (a) Sketch the molecular speed distributions graphs for a sample of gas at (i) 300 K and (ii) 500 K.
Mark the most probable speed at 300 K as v p and at 500 K as Vp’
(b) Use the graphs to compare the number of molecules with speeds greater than the most probable
speed at 300 K and 500 K.
5. If given the root-mean-square speed of gas molecules at a temperature of 300 K is 420 ms -1, what is
the new root-mean-square speed of the gas molecules at a temperature of 450 K?
Вам также может понравиться
- Checkpoint 9.4Документ1 страницаCheckpoint 9.4Az MYОценок пока нет
- Chapter 10b Kinetic Theory For Ideal GasesДокумент3 страницыChapter 10b Kinetic Theory For Ideal GasesPathmanathan NadesonОценок пока нет
- Kinetic Theory of Gas: Concept MapДокумент7 страницKinetic Theory of Gas: Concept MapSoham NagОценок пока нет
- CHM 221 Questions Tutorial 4 07.09.18Документ1 страницаCHM 221 Questions Tutorial 4 07.09.18Manish KОценок пока нет
- Chapter 4 - ThermodynamicsДокумент4 страницыChapter 4 - ThermodynamicsTHIÊN LÊ TRẦN THUẬNОценок пока нет
- Exercises Unit2 1Документ9 страницExercises Unit2 1Clara Carrera0% (1)
- Gas Field Engineering (PAB4013) : May 2016 SemesterДокумент3 страницыGas Field Engineering (PAB4013) : May 2016 SemesterShen YenОценок пока нет
- Cbse Test Paper-03 CLASS - XI CHEMISTRY (States of Matter: Gases and Liquids) Topic: - Ideal Gas EquationДокумент1 страницаCbse Test Paper-03 CLASS - XI CHEMISTRY (States of Matter: Gases and Liquids) Topic: - Ideal Gas Equationsatya176Оценок пока нет
- Thermal Physics QuestionsДокумент2 страницыThermal Physics Questionsapi-3804363100% (1)
- Physical 10Документ2 страницыPhysical 10Rodriguez RommelОценок пока нет
- 11 Physics Kinetic Theory of Gases Test 03Документ1 страница11 Physics Kinetic Theory of Gases Test 03Edukondalu NamepalliОценок пока нет
- Jee MДокумент16 страницJee Mkhushhal2024Оценок пока нет
- Question PART 2 (2023)Документ6 страницQuestion PART 2 (2023)01khanh26Оценок пока нет
- Kinetic Theory of Gases2Документ1 страницаKinetic Theory of Gases2RagsОценок пока нет
- Latihan Soal Energi Kinetik GasДокумент13 страницLatihan Soal Energi Kinetik GasDevita Mira Lestari100% (2)
- Take Home Exam Gases Quantum Theory and Atomic StructureДокумент2 страницыTake Home Exam Gases Quantum Theory and Atomic StructureBeri NaysОценок пока нет
- Quiz 19: Multiple-Choice Questions On Ideal Gas LawsДокумент2 страницыQuiz 19: Multiple-Choice Questions On Ideal Gas LawsRoger Lenin Maya MartinezОценок пока нет
- Thermal Physics A2 Matter (Ideal Gas) WS FinalДокумент4 страницыThermal Physics A2 Matter (Ideal Gas) WS FinalMahbub KhanОценок пока нет
- Answers To Questions 1 - 5 Should Be Handed in by Friday 4 November, 2005. Question 6 Is For Tutorial DiscussionДокумент1 страницаAnswers To Questions 1 - 5 Should Be Handed in by Friday 4 November, 2005. Question 6 Is For Tutorial DiscussionShootingStarPhotonsОценок пока нет
- Assignment CДокумент3 страницыAssignment CWilson JonesОценок пока нет
- Volume, and Process CB Is Adiabatic. The Temperatures of The Gas States A, C, and B AreДокумент4 страницыVolume, and Process CB Is Adiabatic. The Temperatures of The Gas States A, C, and B AreJay KayeОценок пока нет
- Latihan Soal GasДокумент1 страницаLatihan Soal GasnajmahsОценок пока нет
- 3r PC ActivitiesUD1Документ3 страницы3r PC ActivitiesUD1danidomingo2009Оценок пока нет
- Assignment Physics2Документ2 страницыAssignment Physics2Phạm Thiên LongОценок пока нет
- Xii Iit RPTM - 16 Q.paper (17.12.22)Документ13 страницXii Iit RPTM - 16 Q.paper (17.12.22)Dinesh BabuОценок пока нет
- Midterm & Final ExamДокумент2 страницыMidterm & Final ExambcanturkyilmazОценок пока нет
- ExtraProbPhys2Ch4 KineticsGasДокумент2 страницыExtraProbPhys2Ch4 KineticsGasDuy AnhОценок пока нет
- Assignment 1, Autumn 2023Документ2 страницыAssignment 1, Autumn 2023cocodarshi2022Оценок пока нет
- B.Tech. Degree Examination Me & Ie: (Nov-16) (EME-101)Документ3 страницыB.Tech. Degree Examination Me & Ie: (Nov-16) (EME-101)kohli kingОценок пока нет
- Quiz 07Документ3 страницыQuiz 07Niomi ButtermilkОценок пока нет
- Chap 17Документ60 страницChap 17noscribdyoucantОценок пока нет
- Addis Ababa Science & Technology University, College of Biological & Chemical Engineering, Department of Chemical EngineeringДокумент2 страницыAddis Ababa Science & Technology University, College of Biological & Chemical Engineering, Department of Chemical EngineeringElias LibayОценок пока нет
- KTG UpdatedДокумент5 страницKTG Updateddeejam123Оценок пока нет
- Problems Chapter 1 Sec BДокумент7 страницProblems Chapter 1 Sec BSana AshfaqОценок пока нет
- Worksheet - 5 - ( (KMT, Effusion. Diffusion, Van Der Waals Equation)Документ3 страницыWorksheet - 5 - ( (KMT, Effusion. Diffusion, Van Der Waals Equation)Jose Ruben SortoОценок пока нет
- Assignment 2Документ5 страницAssignment 2SouravОценок пока нет
- Physics ALMCДокумент185 страницPhysics ALMClavina rachelОценок пока нет
- Physical Chemistry TutorialДокумент1 страницаPhysical Chemistry TutorialfarrahnajihahОценок пока нет
- Behaviour of Perfect Gas Class XIДокумент12 страницBehaviour of Perfect Gas Class XIKAMAL KANT KUSHWAHAОценок пока нет
- SOAL Modul 9 Fidas IA - 2022 2023 enДокумент2 страницыSOAL Modul 9 Fidas IA - 2022 2023 enMario Hermawan SetiadiОценок пока нет
- Thermo 3 Ideal Gas 2022 OnlineДокумент4 страницыThermo 3 Ideal Gas 2022 OnlineRonnieОценок пока нет
- Tses QPSДокумент15 страницTses QPSDilleswararao ChintalaОценок пока нет
- University of LondonДокумент6 страницUniversity of LondonShootingStarPhotonsОценок пока нет
- PExam MBДокумент1 страницаPExam MBzedrickОценок пока нет
- WS28 Gas Law (III)Документ4 страницыWS28 Gas Law (III)hiu nok kwanОценок пока нет
- Practice Problems 4 (KMT, Effusion. Diffusion, Van Der Waals Equation)Документ3 страницыPractice Problems 4 (KMT, Effusion. Diffusion, Van Der Waals Equation)Jose Ruben SortoОценок пока нет
- Problem Set 3 2004Документ2 страницыProblem Set 3 2004jose tellezОценок пока нет
- CHE 220-FALL23-Homework VДокумент2 страницыCHE 220-FALL23-Homework Vsemikonino35Оценок пока нет
- JH EcampusUpload SubjectNote STUDY of GAS LAWSДокумент2 страницыJH EcampusUpload SubjectNote STUDY of GAS LAWSdiamehta1410Оценок пока нет
- TD Assignment 01 2022 23 IsemДокумент1 страницаTD Assignment 01 2022 23 IsemMurali KrishnaОценок пока нет
- Kinetic Theory of Gases WorksheetДокумент5 страницKinetic Theory of Gases WorksheetSajjan BalasubramanyanОценок пока нет
- MSRLДокумент11 страницMSRLAhmad ChuzgapaОценок пока нет
- 11 Physics Unsolved 02 NewДокумент4 страницы11 Physics Unsolved 02 NewANKUR ACHARJEEОценок пока нет
- Physical ChemДокумент8 страницPhysical ChemjahmanОценок пока нет
- PHY607 - Tutorial 4Документ1 страницаPHY607 - Tutorial 4Nahi SakeОценок пока нет
- Lecture - 5 ExamplesДокумент26 страницLecture - 5 ExamplesDimas Angga100% (7)
- AlıştırmaДокумент3 страницыAlıştırmaJanet Baker100% (1)
- Problem SetsДокумент12 страницProblem SetsZAKARIA SUNBOLОценок пока нет
- 08r059210304 ThermodynamicsДокумент8 страниц08r059210304 ThermodynamicsandhracollegesОценок пока нет
- Physics STPMДокумент9 страницPhysics STPMAz MYОценок пока нет
- Read and Understand The Notes. Answer The Examples Questions BelowДокумент1 страницаRead and Understand The Notes. Answer The Examples Questions BelowAz MYОценок пока нет
- 10.1 Heat CapacitiesДокумент2 страницы10.1 Heat CapacitiesAz MYОценок пока нет
- Alternating Current CircuitsДокумент2 страницыAlternating Current CircuitsAz MYОценок пока нет
- Checkpoint 11.1Документ2 страницыCheckpoint 11.1Az MY100% (1)
- 10.1 Heat CapacitiesДокумент2 страницы10.1 Heat CapacitiesAz MYОценок пока нет
- Checkpoint 11.1Документ2 страницыCheckpoint 11.1Az MYОценок пока нет
- Checkpoint 10.1Документ1 страницаCheckpoint 10.1Az MYОценок пока нет
- Checkpoint 10.1Документ1 страницаCheckpoint 10.1Az MYОценок пока нет
- Peta 1Документ1 страницаPeta 1Az MYОценок пока нет
- InertiaДокумент21 страницаInertiaAz MY100% (1)
- Read and Understand The Notes. Answer The Examples Questions BelowДокумент1 страницаRead and Understand The Notes. Answer The Examples Questions BelowAz MYОценок пока нет
- 18.4 RC and RL Circuits in Series PDFДокумент5 страниц18.4 RC and RL Circuits in Series PDFAz MYОценок пока нет
- Paper 3Документ16 страницPaper 3Az MYОценок пока нет
- 18.2 Alternating Currect Through An Inductor PDFДокумент9 страниц18.2 Alternating Currect Through An Inductor PDFAz MYОценок пока нет
- 18.3 Alternating Current Through A Capacitor PDFДокумент6 страниц18.3 Alternating Current Through A Capacitor PDFAz MY100% (1)
- Peperiksaan Percubaan Sijil Pelajaran Malaysia 2008: FizikДокумент7 страницPeperiksaan Percubaan Sijil Pelajaran Malaysia 2008: FizikAz MYОценок пока нет
- L!Tl1 Nldlaq Eqljo Eql ( (L//: ElelsДокумент1 страницаL!Tl1 Nldlaq Eqljo Eql ( (L//: ElelsAz MYОценок пока нет
- Img 20140511 0007Документ1 страницаImg 20140511 0007Az MYОценок пока нет
- Img 20140413 0001Документ1 страницаImg 20140413 0001Az MYОценок пока нет
- Img 20140306 0004Документ1 страницаImg 20140306 0004Az MYОценок пока нет
- Chapter 3 P2 IntensiveДокумент23 страницыChapter 3 P2 IntensiveAz MYОценок пока нет
- Img 20140310 0005Документ1 страницаImg 20140310 0005Az MYОценок пока нет