Академический Документы
Профессиональный Документы
Культура Документы
Solvents
Загружено:
Joske DuchatooИсходное описание:
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
Solvents
Загружено:
Joske DuchatooАвторское право:
Доступные форматы
Properties of some common solvents.
solvent
acetone chloroform diethyl ether ethanol (95%) ethyl acetate hexanes isopropanol methanol methylene chloride toluene water
formula
(CH3)2C=O CHCl3 (CH3CH2)20 CH3CH2OH CH3CO2CH2CH3 C6H14 CH3CHOHCH3 CH3OH CH2Cl2 C6H5CH3 H2O 56 61 35 78 77
bp (C)
density (g/mL)
0.8 1.5 0.7 0.8 0.9 0.7 0.8 0.8 1.3 0.9 1.0
hazards
ammable toxic highly ammable ammable ammable ammable ammable ammable, toxic toxic ammable
6769 82 65 41 111 100
Properties of some common acids and bases. compound
acetic acid, conc., 99.8% sulfuric acid, conc., 96% phosphoric acid, conc., 85% nitric acid, conc., 70% hydrochloric acid, conc., 37% ammonium hydroxide, conc., 29%
formula
CH3CO2H H2SO4 H3PO4 HNO3 HCl NH4OH
MW of pure compound
60.05 98.08 98.00 63.02 36.47 35.05
normality
17.4 36 14.7 17 12 14.8
density (g/ mL)
1.05 1.84 1.69 1.42 1.19 0.90
Some properties of salts/salt solutions used in organic chemistry. name
sodium bicarbonate sodium carbonate sodium chloride
formula
NaHCO3 Na2CO3 NaCl
MW
84.00 106.00 58.45
solubility
1 part in 10 parts water at 25 1 part in 3.5 parts water at 25 1 gram dissolves in 2.8 mL of water at 25
miscellaneous
pH of 0.1 M aq. solution = 8.3; density is about 1 Density is about 1 Density of saturated solution = 1.202
Chromatography solvents eluting order. hexanes increasing polarity (more powerful eluters) toluene methylene chloride diethyl ether ethyl acetate acetone methanol, ethanol acetic acid
Usual eluting order of organic compounds. saturated hydrocarbons unsaturated hydrocarbons more polar (stronger bonding) ethers esters halides ketones aldehydes amines alcohols acids and bases
Вам также может понравиться
- Solvent Miscibility and Polarity ChartДокумент1 страницаSolvent Miscibility and Polarity Chartdiptafara100% (3)
- Chemical Resistance of PlasticsДокумент2 страницыChemical Resistance of PlasticsAFLAC ............100% (2)
- Advanced Pharmaceutical analysisОт EverandAdvanced Pharmaceutical analysisРейтинг: 4.5 из 5 звезд4.5/5 (2)
- 7detergent Manufacturing and FormulationДокумент20 страниц7detergent Manufacturing and FormulationFADZAI DUBE100% (2)
- Glycerine Ecogreen SpecДокумент1 страницаGlycerine Ecogreen SpecrusyadОценок пока нет
- The Chemistry of Fertilisers and Manure - Including Information on the Chemical Constituents and Types of Fertilisers and ManuresОт EverandThe Chemistry of Fertilisers and Manure - Including Information on the Chemical Constituents and Types of Fertilisers and ManuresРейтинг: 5 из 5 звезд5/5 (1)
- LC Training Basic HPLC 2001 AДокумент124 страницыLC Training Basic HPLC 2001 Abile86Оценок пока нет
- Experiment 1. Solubility of Organic CompoundsДокумент7 страницExperiment 1. Solubility of Organic CompoundsJeanette Vivien JalalonОценок пока нет
- The Chemistry of Dairy Products - A Chemical Analysis of Milk, Cream and ButterОт EverandThe Chemistry of Dairy Products - A Chemical Analysis of Milk, Cream and ButterОценок пока нет
- Common and Trade Names of ChemicalsДокумент6 страницCommon and Trade Names of ChemicalsCY ChewОценок пока нет
- Perfumery As Science (Steeve Herman)Документ82 страницыPerfumery As Science (Steeve Herman)SpiceandWood87% (15)
- Chapter 1 CHE 124Документ62 страницыChapter 1 CHE 124RobinsОценок пока нет
- Chemical Compatibility Guide: Key To The SymbolsДокумент5 страницChemical Compatibility Guide: Key To The SymbolsmniarunОценок пока нет
- Macrogol Cetostearyl EtherДокумент1 страницаMacrogol Cetostearyl EtherTeodor BoianovОценок пока нет
- Final Mint TableДокумент1 страницаFinal Mint Tablesg005Оценок пока нет
- Common Name Vs Chemical NameДокумент5 страницCommon Name Vs Chemical NameManeesh RanjanОценок пока нет
- 316L Korozif Kimyasallar Için MalzemelerДокумент5 страниц316L Korozif Kimyasallar Için MalzemelerwenigmaОценок пока нет
- Student Copy True Lipids TrueДокумент113 страницStudent Copy True Lipids TrueSree Balaji Srinivas KatakamОценок пока нет
- S.No List of Common Solvents in Pharma Chemical Forumla Boiling PointДокумент9 страницS.No List of Common Solvents in Pharma Chemical Forumla Boiling PointtpchowoОценок пока нет
- Chemical Formula of Different Reagents Used in HistopathologyДокумент5 страницChemical Formula of Different Reagents Used in HistopathologyViralyn Mores EscalanteОценок пока нет
- R F (Solvent)Документ8 страницR F (Solvent)MedОценок пока нет
- Substante Organice Pentru Admitere MedicinaДокумент21 страницаSubstante Organice Pentru Admitere MedicinaDragotaОценок пока нет
- Denumire Substanta Formula Chimica Clorura de Vinil CH2 CH-CLДокумент21 страницаDenumire Substanta Formula Chimica Clorura de Vinil CH2 CH-CLLayla NicoОценок пока нет
- C Aromatics: Hazchem Code EPA CodeДокумент3 страницыC Aromatics: Hazchem Code EPA CodeHector Flores MarcosОценок пока нет
- ml7b00356 Si 001 PDFДокумент75 страницml7b00356 Si 001 PDFPULIDO PEÑA JOHN SEBASTIANОценок пока нет
- Op034094j Si 001Документ18 страницOp034094j Si 001dhavalesh1Оценок пока нет
- Epoxy CoatingsДокумент1 страницаEpoxy Coatingsgreyhound726415Оценок пока нет
- Smart Polymers & HydrogelsДокумент8 страницSmart Polymers & HydrogelsSabiruddin Mirza DipuОценок пока нет
- LipidsДокумент75 страницLipidsanaОценок пока нет
- Protein, Dan LemakДокумент70 страницProtein, Dan LemakAlief MoulanaОценок пока нет
- MXEДокумент2 страницыMXEEn DaОценок пока нет
- Ol9011772 Si 001Документ8 страницOl9011772 Si 001James PerianayagamОценок пока нет
- CHEM 180: Christian MANAHAN Anna Esperanza LEGASPIДокумент19 страницCHEM 180: Christian MANAHAN Anna Esperanza LEGASPIAnna LegaspiОценок пока нет
- Alcano 9Документ4 страницыAlcano 9Antônio Neto MachadoОценок пока нет
- Eat of Solution Data For Aqueous SolutionsДокумент2 страницыEat of Solution Data For Aqueous SolutionsJúlio Gabriel Queiroz dos SantosОценок пока нет
- Heat of Solution Data PDFДокумент2 страницыHeat of Solution Data PDFdodofan2000Оценок пока нет
- Materiales Empaques IДокумент1 страницаMateriales Empaques IAnonymous uMusOEjaCОценок пока нет
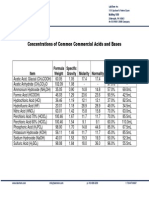
- Concentrations of Common Commercial Acids and BasesДокумент1 страницаConcentrations of Common Commercial Acids and BasesNazimah MaqboolОценок пока нет
- Mau So 1 - Du Tru Hoa Chat 09dhltДокумент87 страницMau So 1 - Du Tru Hoa Chat 09dhltNghĩa LêОценок пока нет
- jm901265h Si 001Документ37 страницjm901265h Si 001Quoc AnhОценок пока нет
- jm9b01579 Si 001Документ36 страницjm9b01579 Si 001Jagjeet GujralОценок пока нет
- Appendix 1 Thermodynamic Data For Selected Chemical CompoundsДокумент1 страницаAppendix 1 Thermodynamic Data For Selected Chemical CompoundsDelroy Washington BrownОценок пока нет
- Donohoe Lab Reagent ListДокумент9 страницDonohoe Lab Reagent Listmcclain_matthewОценок пока нет
- 07.Mmc ExperimentalДокумент14 страниц07.Mmc ExperimentalNurul Putry SyahrulОценок пока нет
- MMC 1Документ83 страницыMMC 1James PerianayagamОценок пока нет
- The Dried Aerial PartsДокумент2 страницыThe Dried Aerial PartsJhon Sebastian Baracaldo HernandezОценок пока нет
- SolventДокумент1 страницаSolventlycheatnstealОценок пока нет
- Acrylic Technical DataДокумент1 страницаAcrylic Technical DataimamudeenОценок пока нет
- Fluid Reference TableДокумент3 страницыFluid Reference TableSB216Оценок пока нет
- AspirinДокумент15 страницAspirinJuan PerezОценок пока нет
- Water Quality Gaza MapsДокумент10 страницWater Quality Gaza MapsReemALMousawiОценок пока нет
- Dissociation Constants of Organic Acids and BasesДокумент20 страницDissociation Constants of Organic Acids and BasesMarcel Chevalier100% (1)
- Mono Chloro Acetic AcidДокумент2 страницыMono Chloro Acetic AcidSantosh Sharma VaranasiОценок пока нет
- List of Saturated Fatty Acids - WikipediaДокумент4 страницыList of Saturated Fatty Acids - WikipediaKurt RussellОценок пока нет
- Carboxylic AcidДокумент6 страницCarboxylic Acidbalweg mackyОценок пока нет
- 1-15 Tutorial Questions: HG HGДокумент4 страницы1-15 Tutorial Questions: HG HGAyez SassinОценок пока нет
- BetamethasonaДокумент10 страницBetamethasonaJuan PerezОценок пока нет
- Process Reactor Phase Temperature Pressure Catalyst Metal Conversion Per Pass (%) Yield By-ProductsДокумент2 страницыProcess Reactor Phase Temperature Pressure Catalyst Metal Conversion Per Pass (%) Yield By-ProductsBhuneshwar ChelakОценок пока нет
- Carbohydrate Analysis by HPLC II ApplicationsДокумент1 страницаCarbohydrate Analysis by HPLC II ApplicationsYan XunОценок пока нет
- Application of IC-MS and IC-ICP-MS in Environmental ResearchОт EverandApplication of IC-MS and IC-ICP-MS in Environmental ResearchRajmund MichalskiОценок пока нет