Академический Документы
Профессиональный Документы
Культура Документы
Acids, Bases and Salts
Загружено:
Fouzia0 оценок0% нашли этот документ полезным (0 голосов)
116 просмотров6 страницAcids and bases play an important role in our lives. Acids have a sour taste, turn litmus paper red, and conduct electricity in water, while bases have a bitter taste, turn litmus paper blue, and feel soapy. Common acids include hydrochloric acid, nitric acid, and sulfuric acid. Common bases include sodium hydroxide, potassium hydroxide, and calcium hydroxide. While the Arrhenius concept of acids and bases in terms of hydrogen and hydroxide ions in water helped explain their properties, it has limitations and cannot account for all acids and bases.
Исходное описание:
An overview of acid, bases and Arrhenius concept of acids and bases
Авторское право
© © All Rights Reserved
Доступные форматы
PPTX, PDF, TXT или читайте онлайн в Scribd
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документAcids and bases play an important role in our lives. Acids have a sour taste, turn litmus paper red, and conduct electricity in water, while bases have a bitter taste, turn litmus paper blue, and feel soapy. Common acids include hydrochloric acid, nitric acid, and sulfuric acid. Common bases include sodium hydroxide, potassium hydroxide, and calcium hydroxide. While the Arrhenius concept of acids and bases in terms of hydrogen and hydroxide ions in water helped explain their properties, it has limitations and cannot account for all acids and bases.
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PPTX, PDF, TXT или читайте онлайн в Scribd
0 оценок0% нашли этот документ полезным (0 голосов)
116 просмотров6 страницAcids, Bases and Salts
Загружено:
FouziaAcids and bases play an important role in our lives. Acids have a sour taste, turn litmus paper red, and conduct electricity in water, while bases have a bitter taste, turn litmus paper blue, and feel soapy. Common acids include hydrochloric acid, nitric acid, and sulfuric acid. Common bases include sodium hydroxide, potassium hydroxide, and calcium hydroxide. While the Arrhenius concept of acids and bases in terms of hydrogen and hydroxide ions in water helped explain their properties, it has limitations and cannot account for all acids and bases.
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PPTX, PDF, TXT или читайте онлайн в Scribd
Вы находитесь на странице: 1из 6
ACIDS, BASES AND SALTS
We are frequently using acids and
bases in our every aspects of life.
ACIDS AND BASES
Acids Bases
• `The word acid is derived from • A base is any metal oxide or
Latin words “acidus”. hydroxide that react with an
acid to produce salt and water.
• Acids have sour taste.
• Bases have bitter taste and
• It turn blue litmus paper red. feel soapy.
• It changes the colour of acid- • It turns red litmus paper blue.
base indicators. • It changes the colour of acid
• Aq. solution of acids conduct bases indicators.
electric current and are very • Strong bases are corrosive and
corrosive (destroy fabric and conduct electricity when
cause burns on the skin) dissolve in water.
Arrhenius Concept of Acids and Bases
• A Swedish chemist in 1884 explained the
aqueous solutions of acids and bases can
conduct electric current by producing ions in
aqueous solution.
• An acids is a chemical substance that dissociates in
aqueous solution to give (H+) hydrogen ions.
• HCl H+ + Cl-
• A base is a chemical substance that dissociates in
aqueous solution to give hyroxyl ions (OH-)
• NaOH Na+ + OH-
Some common Acids and their uses
Name Formula Common use
Hydrochloric Acid HCl Cleaning of metals , ricks and removing
scales from boilers
Nitric Acid HNO3 Manufacturing of fertilizers, explosives
Sulphuric acid H2SO4 Manufacturing of many chemicals, drugs,
dyes, paints and explosives.
Phosphoric acid H3PO4 Manufacturing of fertilizers, acidulant for
food.
Common uses of bases
Name Formula Common name
Sodium hydroxide NaOH Soap making, drain cleaners
Potassium hydroxide KOH Making liquid soap, shaving cream
Calcium Hydroxide Ca(OH)2 Making mortar, plaster, cement
Magnesium hydoxide Mg(OH)2 Anacids, laxative
Limitations Arrhenius Concept of Acids
and Bases
The limitations of this concept are:
i. It is limited to aqueous medium only.
ii. It is limited to hydrogen ion (H+) only and
bases are limited to hydroxide ions.
iii. It cannot explain the acidic nature of CO2
and SO2 and also the basic nature of NH3
Вам также может понравиться
- MCQ Drill QuestionsДокумент15 страницMCQ Drill QuestionsLab Rat0% (2)
- A Level Yr 2 Module 5: Communication, Homeostasis and EnergyДокумент52 страницыA Level Yr 2 Module 5: Communication, Homeostasis and EnergyDanielОценок пока нет
- Exercise 4c ChemistryДокумент11 страницExercise 4c Chemistryapi-533545229Оценок пока нет
- Chemistry For WBCS: Atomic Structure-MCQДокумент16 страницChemistry For WBCS: Atomic Structure-MCQMadhab Ch. PoulikОценок пока нет
- The Biology Project - Meiosis TutorialДокумент8 страницThe Biology Project - Meiosis TutorialJimmie MaddoxОценок пока нет
- Cell Cycle Video AssignmentДокумент2 страницыCell Cycle Video Assignmentapi-661317466Оценок пока нет
- CH 13 Online Study GuideДокумент6 страницCH 13 Online Study Guideюрий локтионовОценок пока нет
- Ap Psychology Group Project MotivationДокумент1 страницаAp Psychology Group Project Motivationapi-333631531Оценок пока нет
- Academic Achievement and Cognitive Development of Students: by Rabia Umer MS Clinical PsychologyДокумент26 страницAcademic Achievement and Cognitive Development of Students: by Rabia Umer MS Clinical PsychologyZaryab WaheedОценок пока нет
- Chapter 5 The Periodic TableДокумент45 страницChapter 5 The Periodic Tableapi-30718309Оценок пока нет
- Lab No 2 Mitosis (Final Draft)Документ3 страницыLab No 2 Mitosis (Final Draft)zdkwhwjwfgОценок пока нет
- Chem7-Structure, Bonding and PropertiesДокумент6 страницChem7-Structure, Bonding and PropertiesNor Ashikin IsmailОценок пока нет
- Chemistry MCQДокумент3 страницыChemistry MCQSharkОценок пока нет
- Mole LabДокумент4 страницыMole Labapi-239310645Оценок пока нет
- The Cell Cycle: For Campbell Biology, Ninth EditionДокумент51 страницаThe Cell Cycle: For Campbell Biology, Ninth EditionBelenОценок пока нет
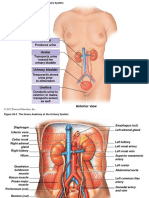
- Anatomy of The Urinary SystemДокумент6 страницAnatomy of The Urinary SystemJeoffrey GabongОценок пока нет
- Chapter 19 Acids, Bases, and SaltsДокумент92 страницыChapter 19 Acids, Bases, and SaltsHo Hsiao JiunОценок пока нет
- Protein Synthesis: How Do We Get Proteins From A Bunch of A's, T'S, C's and G's in DNA??Документ33 страницыProtein Synthesis: How Do We Get Proteins From A Bunch of A's, T'S, C's and G's in DNA??Poy AlisОценок пока нет
- HW 7 Answer KeyДокумент2 страницыHW 7 Answer Keyangelyn martinezОценок пока нет
- Cell Cycle and Its Control: Bi1113 Spring 2021 Mayurika LahiriДокумент42 страницыCell Cycle and Its Control: Bi1113 Spring 2021 Mayurika LahiriSatyam SinhaОценок пока нет
- Lab 2 Moles and Avogadro's Number Vernier H2Документ30 страницLab 2 Moles and Avogadro's Number Vernier H2Helena Metzker0% (1)
- Innovation Management Notes Study MaterialsДокумент101 страницаInnovation Management Notes Study MaterialsSAM's little worlddОценок пока нет
- Buffer KSP ProbДокумент4 страницыBuffer KSP ProbFitria Rahma DewiОценок пока нет
- Unit 8 - Acids, Bases, and Salts Powerpoint (Updated1)Документ46 страницUnit 8 - Acids, Bases, and Salts Powerpoint (Updated1)Rithik Visu100% (1)
- Stoichiometry and Mole Concept Prelim Questions and AnswersДокумент15 страницStoichiometry and Mole Concept Prelim Questions and AnswersSanto Nyuol DengОценок пока нет
- Protein Synthesis Lecture PowerpointДокумент26 страницProtein Synthesis Lecture PowerpointJames Dauray100% (1)
- CHM 100 140 - Empirical and Molecular FormulasДокумент2 страницыCHM 100 140 - Empirical and Molecular FormulasRahul GemnaniОценок пока нет
- Integumentary System Practice Qs 2Документ3 страницыIntegumentary System Practice Qs 2dinsaqiОценок пока нет
- TranscriptionДокумент34 страницыTranscriptiondrhydrogenОценок пока нет
- The Cell Cycle: For Campbell Biology, Ninth EditionДокумент98 страницThe Cell Cycle: For Campbell Biology, Ninth Edition46bwilsonОценок пока нет
- Types and Process of Innovation AДокумент53 страницыTypes and Process of Innovation AMohd ElabedОценок пока нет
- Protein Synthesis and Genetic Code: Departemen BiokimiaДокумент25 страницProtein Synthesis and Genetic Code: Departemen Biokimiaemmm mantabbОценок пока нет
- The Periodic Table QuestionsДокумент1 страницаThe Periodic Table QuestionsRigen AlamОценок пока нет
- Self Concept: by Rabia Umer MS Clinical PsychologyДокумент74 страницыSelf Concept: by Rabia Umer MS Clinical PsychologyRimshaОценок пока нет
- Acids, Bases and SaltsДокумент53 страницыAcids, Bases and SaltsSoumya SarkarОценок пока нет
- Questions Periodic TableДокумент25 страницQuestions Periodic TablejuandesabiОценок пока нет
- CH 15 Acids Bases and SaltsДокумент104 страницыCH 15 Acids Bases and SaltsDaevy Toribio AmdosОценок пока нет
- Ch-2 Acid Base and SaltДокумент40 страницCh-2 Acid Base and SaltRushikKaretiyaОценок пока нет
- Topic3 Periodic TableДокумент66 страницTopic3 Periodic TableNana SazanaОценок пока нет
- Moles Stoichiometry Olevels Only MCQs Complete 2014 Protected PDFДокумент24 страницыMoles Stoichiometry Olevels Only MCQs Complete 2014 Protected PDFAli AshrafОценок пока нет
- Periodic Table WebquestДокумент5 страницPeriodic Table Webquestapi-232022364Оценок пока нет
- The Periodic Table Webquest AnswersДокумент4 страницыThe Periodic Table Webquest AnswersshaneearlОценок пока нет
- Individual Human BehaviourДокумент31 страницаIndividual Human Behavioursaah007Оценок пока нет
- Periodic Table Scavenger HuntДокумент7 страницPeriodic Table Scavenger Huntapi-31851400014% (7)
- 6.2 DNA Replication Full NoteДокумент44 страницы6.2 DNA Replication Full NotejiaenОценок пока нет
- Chapter 6 The Periodic Table and Periodic LawДокумент3 страницыChapter 6 The Periodic Table and Periodic Lawapi-280128396Оценок пока нет
- Urinary System Slides Lab 8Документ17 страницUrinary System Slides Lab 8Asad Javed Mehmood100% (1)
- Small Angle X-Ray ScatteringДокумент5 страницSmall Angle X-Ray ScatteringArvind RenganathanОценок пока нет
- 6.3 MeiosisДокумент38 страниц6.3 Meiosiswickedbiology101Оценок пока нет
- Worksheet 1 - Chemical BondingДокумент4 страницыWorksheet 1 - Chemical BondingFahd KhanОценок пока нет
- Chem 2423 - Chap 5 (Notes) 2017Документ102 страницыChem 2423 - Chap 5 (Notes) 2017FalguniОценок пока нет
- Periodic Table MCQДокумент3 страницыPeriodic Table MCQAbhay Vishwakarma100% (1)
- Atoms & MoleculesДокумент2 страницыAtoms & MoleculesNavas KappilОценок пока нет
- F325 Transition ElementsДокумент18 страницF325 Transition ElementsDoc_CrocОценок пока нет
- Dna Review Packet UpdatedДокумент4 страницыDna Review Packet Updatedapi-259727482Оценок пока нет
- CHAPTER 2 Elements, Compounds, Chem Equations and CalculationsДокумент80 страницCHAPTER 2 Elements, Compounds, Chem Equations and CalculationsNurhayati HasanahОценок пока нет
- 6.1 Role of Water in Showing Chemical Properties of Acid and AlkaliДокумент29 страниц6.1 Role of Water in Showing Chemical Properties of Acid and AlkaliNur Shahirah100% (1)
- Acid, Base and SaltsДокумент10 страницAcid, Base and SaltsLeh Siong LuОценок пока нет
- Acids, Bases and SaltsДокумент28 страницAcids, Bases and SaltsJinsuОценок пока нет
- Istambul TurciaДокумент2 страницыIstambul Turciaantoneacsabin06Оценок пока нет
- Odisha PWD Registration in CDMSДокумент2 страницыOdisha PWD Registration in CDMSSabyasachi Naik (Zico)Оценок пока нет
- 968 BMW X5 (G05) Brochure 20x30cm en v18 Low-Min - 0Документ25 страниц968 BMW X5 (G05) Brochure 20x30cm en v18 Low-Min - 0tarakeshОценок пока нет
- Module Virus Viroids PrionsДокумент17 страницModule Virus Viroids PrionsJennifer MartinОценок пока нет
- UNITRONICS V200-18-E1Документ10 страницUNITRONICS V200-18-E1eddieipenzaОценок пока нет
- Lecture-1: Course Plan / OutlineДокумент3 страницыLecture-1: Course Plan / OutlineNaiem IslamОценок пока нет
- Exercises Problem 2 Lesson 4Документ2 страницыExercises Problem 2 Lesson 4Marianne DevillenaОценок пока нет
- Souvenir As Tourism ProductДокумент13 страницSouvenir As Tourism ProductThree Dimensional Product DesignОценок пока нет
- Spacex PDFДокумент69 страницSpacex PDFEmerovsky ReyesОценок пока нет
- Graphics View FrameworkДокумент7 страницGraphics View FrameworkTxkti BabelОценок пока нет
- Hitachi ZX75USДокумент9 страницHitachi ZX75US3man 9394Оценок пока нет
- Architecture Building Services: Assignment-1Документ36 страницArchitecture Building Services: Assignment-1santhu majiОценок пока нет
- Company Profile Bureau Veritas ThailandДокумент13 страницCompany Profile Bureau Veritas ThailandPitichai PakornrersiriОценок пока нет
- Byzantium Notes CultureДокумент6 страницByzantium Notes Culturepatrick cairoliОценок пока нет
- Quiz ErrorsДокумент5 страницQuiz Errorsanon_843459121Оценок пока нет
- HR-Security Profile OptionДокумент1 страницаHR-Security Profile Optionnil77Оценок пока нет
- Tutorial Week 10 Topic 9Документ14 страницTutorial Week 10 Topic 9patricia93limОценок пока нет
- 2021 Moon Calendar-Cosmic RevolutionДокумент47 страниц2021 Moon Calendar-Cosmic RevolutionYahira NoeliОценок пока нет
- Clock Divider and Pulse CounterДокумент4 страницыClock Divider and Pulse CounterRohit PanwarОценок пока нет
- Return To Sport After Surgical Management of Posterior Shoulder InstabilityДокумент13 страницReturn To Sport After Surgical Management of Posterior Shoulder InstabilityAziz CupinОценок пока нет
- TLE 7 - 8 - Carpentry - Module 1Документ24 страницыTLE 7 - 8 - Carpentry - Module 1Marycon Nacito92% (13)
- Medica EasyLyte BrochureДокумент6 страницMedica EasyLyte BrochureAnderson José Carrillo NárvaezОценок пока нет
- Nts Tevta Test 22-03-15 Paper BДокумент2 страницыNts Tevta Test 22-03-15 Paper BBeTechieОценок пока нет
- À Bout de Souffle (Breathless) : Treatment by François TruffautДокумент10 страницÀ Bout de Souffle (Breathless) : Treatment by François TruffautAlex KahnОценок пока нет
- English Convents in ExileДокумент6 страницEnglish Convents in ExilePickering and ChattoОценок пока нет
- Stoeger Airguns Product Catalog 2022Документ10 страницStoeger Airguns Product Catalog 2022Jack JackОценок пока нет
- Internet BillДокумент2 страницыInternet Billshiva ramОценок пока нет
- "Some of Us May Never Die," by Kathleen SteinДокумент5 страниц"Some of Us May Never Die," by Kathleen SteinMark PlusОценок пока нет
- International Business Trade ReviewerДокумент8 страницInternational Business Trade ReviewerCoffee VanillaОценок пока нет
- Systematic Review Dissertation ExamplesДокумент4 страницыSystematic Review Dissertation ExamplesCheapestPaperWritingServiceBaltimore100% (1)