Академический Документы
Профессиональный Документы
Культура Документы
Serial Dilution Explained
Загружено:
Likith Kumar Likith0%(1)0% нашли этот документ полезным (1 голос)
1K просмотров6 страницA serial dilution is a dilution method where the concentration decreases by the same factor in each successive step. This is done by taking a portion of the diluted solution from the previous step and adding it to fresh diluent. The overall dilution factor is calculated by multiplying the individual dilution factors at each step. For example, if a 1:20 dilution is performed four times in succession, the final dilution would be 1:160,000.
Исходное описание:
serial dilution
Оригинальное название
Serial Dilution Ppt 2019
Авторское право
© © All Rights Reserved
Доступные форматы
PPTX, PDF, TXT или читайте онлайн в Scribd
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документA serial dilution is a dilution method where the concentration decreases by the same factor in each successive step. This is done by taking a portion of the diluted solution from the previous step and adding it to fresh diluent. The overall dilution factor is calculated by multiplying the individual dilution factors at each step. For example, if a 1:20 dilution is performed four times in succession, the final dilution would be 1:160,000.
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PPTX, PDF, TXT или читайте онлайн в Scribd
0%(1)0% нашли этот документ полезным (1 голос)
1K просмотров6 страницSerial Dilution Explained
Загружено:
Likith Kumar LikithA serial dilution is a dilution method where the concentration decreases by the same factor in each successive step. This is done by taking a portion of the diluted solution from the previous step and adding it to fresh diluent. The overall dilution factor is calculated by multiplying the individual dilution factors at each step. For example, if a 1:20 dilution is performed four times in succession, the final dilution would be 1:160,000.
Авторское право:
© All Rights Reserved
Доступные форматы
Скачайте в формате PPTX, PDF, TXT или читайте онлайн в Scribd
Вы находитесь на странице: 1из 6
Serial Dilution
a serial dilution is any dilution where the concentration
decreases by the same quantity in each successive step.
A serial dilution is any dilution in which the concentration
decreases by the same factor in each successive step.
In serial dilutions, you multiply the dilution factors for each
step.
• If a solution has a 1/10 dilution the number represents
1 part of the patient sample added to 9 parts of
diluent.
• So the volumes used would be 10-1= 9.
• This represents 1 part patient sample added to 9 parts
of diluent.
The dilution factor or the dilution is the initial volume
divided by the final volume.
DF = Vi
Vf
For example, if you add a 1 mL sample to 9 mL of diluent to
get 10 mL of solution, therefore
DF = Vi = 1mL = 1 This is a 1:10 dilution.
Vf 10 mL 10
Example 1
What is the dilution factor if you add 0.2 mL of a stock
solution to 3.8 mL of diluent?
Vf = 0.2 mL + 3.8 mL = 4.0 mL
DF = Vi/Vf = 0.2mL/4.0mL = 1/20
This is a 1:20 dilution or 5-2
Example 2
If you did the above dilution 4 times, what would be the final dilution
factor?
Solution 2
Remember that serial dilutions are always made by taking a set quantity
of the initial dilution and adding it successively to tubes with the same
volume. So you multiply each successive dilution by the dilution factor.
You would transfer 0.2 mL from Tube 1 to 3.8 mL of diluent in Tube 2
and mix. Then transfer 0.2 mL from Tube 2 to 3.8 mL of diluent in Tube
3 and mix. Repeat the process until you have four tubes.
The dilution factor after four dilutions is
DF = 1/20 × 1/20 × 1/20 × 1/20 = 1/160000 = 1:160000 or 6.25 x 10⁻6
If the concentration of the original stock solution was 100 µg/µL, the
concentration in Tube 4 would be
100 µg/µL × 1/160000 = 6.25 × 10⁻⁴ µg/µL
Вам также может понравиться
- Serial DilutionДокумент8 страницSerial DilutionRiyanОценок пока нет
- Aseptic Technique Lab ReportДокумент8 страницAseptic Technique Lab ReportSleepyHead ˋωˊОценок пока нет
- Cell CountДокумент2 страницыCell CountBridget HaleyОценок пока нет
- Serial Dilution MethodДокумент3 страницыSerial Dilution Methoddraneesh75% (4)
- Dna Purification and Extraction Practical ReportДокумент8 страницDna Purification and Extraction Practical ReportAnselmo ManishaОценок пока нет
- SerialDilutions PDFДокумент5 страницSerialDilutions PDFAmelia_KharismayantiОценок пока нет
- Lab # 7 - Serial DilutionДокумент2 страницыLab # 7 - Serial DilutionKESHAWN TYRONE SUTHERLANDОценок пока нет
- 9th Biology Practicals 2Документ1 страница9th Biology Practicals 2Muhammad Hassan Tahir100% (1)
- Minimal Inhibitory Concentration TestДокумент26 страницMinimal Inhibitory Concentration TestZandhika Alfi PratamaОценок пока нет
- Lab 17 Serial DilutionДокумент15 страницLab 17 Serial DilutionCedricDiehlОценок пока нет
- Renal Anatomy & Physiology OverviewДокумент5 страницRenal Anatomy & Physiology OverviewChristopher BucuОценок пока нет
- Lab Report 1 SERIAL DILUTION OF MICROBIAL SAMPLEДокумент6 страницLab Report 1 SERIAL DILUTION OF MICROBIAL SAMPLERidma madusanka weerasinghaОценок пока нет
- Isolation of Bacteria To Pure CultureДокумент7 страницIsolation of Bacteria To Pure CultureMike RelucioОценок пока нет
- Osmotic Fragility of Red Blood CellsДокумент3 страницыOsmotic Fragility of Red Blood Cellschaudhry umar farooqОценок пока нет
- Clinical Lab DilutionsДокумент19 страницClinical Lab DilutionsAlex Joshua Maglasang100% (1)
- Lab 7 Blood Smear and Morphology AnalysisДокумент8 страницLab 7 Blood Smear and Morphology Analysisapi-309917909100% (1)
- Polymerase Chain Reaction (PCR)Документ21 страницаPolymerase Chain Reaction (PCR)greateinsteinОценок пока нет
- Acid Fast StainingДокумент4 страницыAcid Fast Stainingchaudhary TahiraliОценок пока нет
- Lab Report DNA ExtractionДокумент1 страницаLab Report DNA ExtractionEzio Auditore100% (1)
- Bacterial Enumeration 2013Документ27 страницBacterial Enumeration 2013ririsrk100% (1)
- 099 - VaccinesДокумент4 страницы099 - VaccinesArjan LallОценок пока нет
- Restriction Enzyme DigestionДокумент3 страницыRestriction Enzyme DigestionMahathir Mohmed100% (1)
- Antibodies Structure and FunctionДокумент16 страницAntibodies Structure and Functionياسين احمد علي الشيخОценок пока нет
- Protoplast Culture TechniquesДокумент76 страницProtoplast Culture Techniquesrajiv pathakОценок пока нет
- Equipments and Materials Used in MCB LabДокумент14 страницEquipments and Materials Used in MCB LabBarry Allen100% (1)
- Pure Culture TechniqueДокумент6 страницPure Culture Techniquebetu8137Оценок пока нет
- SDS-PAGE of Protein: BT 510 Analytical Biotechnology LabДокумент6 страницSDS-PAGE of Protein: BT 510 Analytical Biotechnology LabBarish RoyОценок пока нет
- Cell Counting Neubauer ChamberДокумент6 страницCell Counting Neubauer ChamberHerma ZulaikhaОценок пока нет
- Penicillin FermentationДокумент30 страницPenicillin FermentationUlfia Al RahmaОценок пока нет
- Cell Biology Lab Manual for B.Tech BiotechnologyДокумент31 страницаCell Biology Lab Manual for B.Tech BiotechnologyVenus Divinagracia0% (2)
- Bacterial, GeneticsДокумент21 страницаBacterial, GeneticsMoona WahabОценок пока нет
- Differential CountДокумент7 страницDifferential CountMohsen HaleemОценок пока нет
- Full Report ELISA LatestДокумент8 страницFull Report ELISA LatestBilal TahirОценок пока нет
- Ultracentrifugation AssignmentДокумент3 страницыUltracentrifugation AssignmentStuti SrivastavОценок пока нет
- Inheritance Biology EDUNCLEДокумент54 страницыInheritance Biology EDUNCLEEvita Almeida100% (1)
- 11.1 Antibody Production and VaccinationДокумент28 страниц11.1 Antibody Production and VaccinationFRENCHONLYОценок пока нет
- Polymerase Chain Reaction (PCR)Документ42 страницыPolymerase Chain Reaction (PCR)zamakhsyarifattah100% (3)
- Classification of MicroorganismsДокумент13 страницClassification of MicroorganismsIngrid Amadea100% (2)
- Techniques For Collection, Isolation and Preservation of MicroorganismsДокумент42 страницыTechniques For Collection, Isolation and Preservation of Microorganismsaziskf100% (2)
- DNA Quantification ProtocolДокумент4 страницыDNA Quantification Protocolme_dayakar100% (1)
- Microbiology PDFДокумент71 страницаMicrobiology PDFDanny Alexander TullumeОценок пока нет
- Isolation of Bacterial Plasmid DNA (Compatibility Mode)Документ18 страницIsolation of Bacterial Plasmid DNA (Compatibility Mode)Khandoker Faisal100% (1)
- Blood Smear Examination 1معدل Документ74 страницыBlood Smear Examination 1معدل Kenesa100% (1)
- Restriction Enzymes PDFДокумент7 страницRestriction Enzymes PDFmanoj_rkl_07Оценок пока нет
- Chicken Embryo Lab ReportДокумент10 страницChicken Embryo Lab Reportgmk5031Оценок пока нет
- Calculate The Colony Forming UnitsДокумент6 страницCalculate The Colony Forming UnitsKrishna Raj100% (1)
- 8 Culture MethodsДокумент14 страниц8 Culture MethodsPrincess AnnОценок пока нет
- Lab 01 - Streaking For Isolation and Interpreting Primary Culture ResultsДокумент18 страницLab 01 - Streaking For Isolation and Interpreting Primary Culture ResultsVincent ReyesОценок пока нет
- NCCLS Manual of Antimicrobial Susceptibility TestingДокумент241 страницаNCCLS Manual of Antimicrobial Susceptibility TestingManu SantosОценок пока нет
- Semen Analysis (Seminal Cytology, Sperm CountДокумент30 страницSemen Analysis (Seminal Cytology, Sperm Countabdurae100% (1)
- DNA Isolation TechniquesДокумент111 страницDNA Isolation TechniquesPamela MendozaОценок пока нет
- MEDIA Animal Cell CultureДокумент28 страницMEDIA Animal Cell Culturejithinnx100% (10)
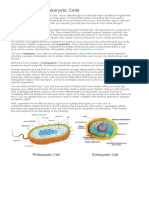
- Differences Between Prokaryotic and Eukaryotic CellsДокумент2 страницыDifferences Between Prokaryotic and Eukaryotic CellsCharisse Viste100% (1)
- Detect Antibodies and Proteins with ELISAДокумент66 страницDetect Antibodies and Proteins with ELISAUsman WaheedОценок пока нет
- Lab Report. Serial DilutionДокумент5 страницLab Report. Serial Dilutionanon_747148947100% (2)
- Senior Medical Laboratory Technician: Passbooks Study GuideОт EverandSenior Medical Laboratory Technician: Passbooks Study GuideОценок пока нет
- Dilutions Concept, Types, Calculations & UsesДокумент26 страницDilutions Concept, Types, Calculations & UsesSasa AbassОценок пока нет
- Dilution SДокумент2 страницыDilution SAndreaОценок пока нет
- Dilutions AVДокумент36 страницDilutions AVtashanplusОценок пока нет
- Laboratory Dilutions GuideДокумент33 страницыLaboratory Dilutions Guide13bellsОценок пока нет
- Chapter 7 UtilityДокумент40 страницChapter 7 UtilityLikith Kumar LikithОценок пока нет
- Elisa PPT 2019Документ24 страницыElisa PPT 2019Likith Kumar LikithОценок пока нет
- Chapter 5 ReliabilityДокумент33 страницыChapter 5 ReliabilityLikith Kumar LikithОценок пока нет
- Chapter 6 ValidityДокумент39 страницChapter 6 ValidityLikith Kumar LikithОценок пока нет
- Ancient Civilizations that Shaped Science and TechnologyДокумент51 страницаAncient Civilizations that Shaped Science and TechnologyLikith Kumar LikithОценок пока нет
- Chapter 4 Physical Development in InfancyДокумент41 страницаChapter 4 Physical Development in InfancyLikith Kumar LikithОценок пока нет
- EAC Student Attitudes Towards Teaching StylesДокумент67 страницEAC Student Attitudes Towards Teaching StylesLikith Kumar LikithОценок пока нет
- Philippine HistoryДокумент5 страницPhilippine HistoryLikith Kumar LikithОценок пока нет
- The Philippines DancesДокумент2 страницыThe Philippines DancesLikith Kumar LikithОценок пока нет
- Concentrations of Solutions 2018Документ31 страницаConcentrations of Solutions 2018Likith Kumar LikithОценок пока нет
- EAC Student Attitudes Towards Teaching StylesДокумент67 страницEAC Student Attitudes Towards Teaching StylesLikith Kumar LikithОценок пока нет
- Frog Reproductive SystemДокумент3 страницыFrog Reproductive SystemLikith Kumar LikithОценок пока нет
- 3 - Meiosis 2019-1Документ62 страницы3 - Meiosis 2019-1Likith Kumar LikithОценок пока нет
- Ethics 1 - BДокумент55 страницEthics 1 - BLikith Kumar LikithОценок пока нет
- Frog Nervous System 2019Документ32 страницыFrog Nervous System 2019Likith Kumar LikithОценок пока нет
- Circulatory System 2019Документ42 страницыCirculatory System 2019Likith Kumar LikithОценок пока нет
- SAT Math Formula eBook: The Ultimate GuideДокумент43 страницыSAT Math Formula eBook: The Ultimate Guidejjilebi100% (3)
- Contemporary World: Mr. Sam Barbo, JRДокумент50 страницContemporary World: Mr. Sam Barbo, JRLikith Kumar LikithОценок пока нет
- BiologyДокумент2 страницыBiologyLikith Kumar LikithОценок пока нет
- Gene Therapy ProcedureДокумент10 страницGene Therapy ProcedureLikith Kumar LikithОценок пока нет
- The Globalisation Effects On ReligionДокумент1 страницаThe Globalisation Effects On ReligionLikith Kumar LikithОценок пока нет
- FUNCTIONSДокумент31 страницаFUNCTIONSLikith Kumar LikithОценок пока нет
- SAT Math Formula eBook: The Ultimate GuideДокумент43 страницыSAT Math Formula eBook: The Ultimate Guidejjilebi100% (3)
- Swara SawantДокумент6 страницSwara SawantLikith Kumar LikithОценок пока нет