Академический Документы
Профессиональный Документы
Культура Документы
The Magazine
Загружено:
AlyssaОригинальное название
Авторское право
Доступные форматы
Поделиться этим документом
Поделиться или встроить документ
Этот документ был вам полезен?
Это неприемлемый материал?
Пожаловаться на этот документАвторское право:
Доступные форматы
The Magazine
Загружено:
AlyssaАвторское право:
Доступные форматы
HOME
Click the cards to proceed
MAGSCIEYA Intermolecular Forces
HOME
In this magazine, the following topics will be
discussed:
1. Intermolecular Forces
Hydrogen bond
Ion-dipole force
Dipole-dipole force
London-dispersion force
The topics listed here seemed too “scientific” but the
knowledge you will gain form them can be used in
different aspects of your life. Moreover, to help you
understand the topics, trace the love story of a couple
and find out what happens then answer the questions in
the activities given. You can answer the activities using
other sheet of paper.
Click the icon to go to the next page
MAGSCIEYA Intermolecular Forces
HOME
Here’s a simple guide for you in going about the LAS:
Follow the instructions.
1. Read and understand the lesson carefully.
2. Take note of the essential parts of the lesson.
3. Conduct the activities. These activities are simple
and they can help you understand the concepts
presented.
4. Don’t cheat.
Learning by yourself takes a lot of self-discipline and
determination. I’m sure you have these traits in you.
“The capacity to learn is a gift; the ability to learn is a
skill; the willingness to learn is a choice.”
Brian Herbert
GOOD LUCK AND HAVE FUN!
Click the icon to go back to the card page
MAGSCIEYA Intermolecular Forces
What to
learn?
Still Trying to Hold On
The life we had was like molecules
oozing with kinetic energy and
continuously moving within a chemical
system. We are still trying to find…
trying to find our significance in this
world… trying to find stability… From
where? From whom? Did you know that
I found that stability? I found it the
moment I met you. When I met you, I
found strength and calmness. Believe it
or not, I felt complete.
We are like the oxygen and hydrogen of
two water molecules (H20) bonded
together by hydrogen bond; a special
type of dipole-dipole. We have different
characteristics… yes very different… but
somehow, my high electronegativity was
pacified by your relative positivity and
made our bond stronger and better than I
could ever imagine.
One good thing about our relationship?
We know how to give not just to receive
despite of our differences because we
always believe that this is what makes us
stronger just like in hydrogen bonding.
Because as they say “Hindi ka lang dapat
tanggap ng tanggap marunong ka rin
magbigay. Para ang relasyon niyo lalong
tumibay.”
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Still Trying to Hold On
Since we are like hydrogen bonds, I
know that it takes a really strong force to
break us apart. But I was wrong… I was
wrong because in instant, everything
was gone.
Hindi ko namalayan na nawawalan na
tayo ng oras sa isa’t isa. Hindi ko
namalayan na nabibitawan na pala kita.
There is still attraction left between us
but… it’s not as strong as what we had
before…
On and off na tayong dalawa. Parang
yung lagnat lang na dala ng dengue diba?
Ano yun naging ion-dipole na?
Ion-dipole force which involves
temporary dipole and permanently
charged ion and the strength depends on
the polarity, electronegativity, and
charge of the ion.
Parang tayo. Tayo na magiging maayos
lang kapag maganda ang ugaling
pinakita. Tayo na nagiging matatag lang
kapag puro saya ang nadarama . At tayo
na laging may batayan para maging tama
para sa isa’t isa. Pero ayos lang sana..
Ayos lang sana kung hindi na lumala.
Ngunit hindi… unti-unti kang lumayo
tangay ang pagmamahal mo… kaya
tuluyan tayong lumabo.
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Still Trying to Hold On
That is the reason why Pinilit kong paniwalain ang
the ion-dipole sarili ko na matibay ang
relationship that you konesyon nating dalawa.
have was gone… Ngunit unti –unti na tayong
Yung ion-dipole naging nasisira. Nagmakaawa ako na
ordinary dipole-dipole maayos pa natin ito dahiil
nalang. hindi ko kayang mawala ka sa
Dipole –dipole which piling ko. Ngunit binalewala
are between two polar mo ako. Ngayon napagtanto ko
molecules. Dipole – ang tagal na ng panahon na
dipole which will work ikaw lagi ang inuuna ko mat sa
if and only if we have paglipas ng panahon sarili ko
the same characteristics. na pala ang nasasaktan ko.
Dipole-dipole na
relationship na maayos
lang dahil parehong
polar. Ganun ba talaga
dapat sa isang relasyon?
Kailangan pareho kayo
ng characteristics para
magkaroon ng konesyon?
Ngunit sige pumapayag
na ako huwag mo lang
akong bitawan…
pipilitin kong kumapit
dahil sayang
pinagsamahan.
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Still Trying to
Hold On INTERMOLECULAR
FORCES
Kaya hindi pala tayo
hydrogen bond o dipole-
dipole… London tayo…
London bridge na unti-
unting gumuguho …
London dispersion force
na pinakamahina sa
lahat ng intermolecular
forces.
DIPOLE– DIPOLE ION– DIPOLE
Now, you can tell me
FORCES FORCES
whatever you want. You
can get mad, you can
shout at me. I’m hoping
that one day you can
forgive me I know that
you are strong enough to
handle this .
I just want you to
remember that I’m LONDON
giving you up not to hurt DISPERSION HYDROGEN BOND
you… I’m giving you up FORCES
to save you… I’m giving
you up to preserve you
because I don’t want you
to get hurt as we
continue this holding on
to this. Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Water can change its phase from liquid to gas when exposed to extreme
heat. Water is known to have a high boiling point. This characteristic is
attributed to the attraction between the molecules of water.
The force of attraction between neighboring molecules is called
intermolecular force. There are different kinds of intermolecular forces
– dipole – dipole, ion- dipole, hydrogen bond, and London dispersion.
Another collective term for intermolecular forces is van der Waals
forces named after Johannes van der Waal, a Dutch chemist.
The different intermolecular forces are the
following:
1. London Dispersion Forces
2. Dipole – Dipole Forces
3. Ion – Dipole Forces
4. Hydrogen bond
Intermolecular
Forces
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Hydrogen
Hydrogen bond is a special type of dipole – dipole interaction that exists
Bond
only in molecules that contain a hydrogen atom bonded to a small, highly
electronegative atom such as N, O, or F. The more electronegative atom
pulls the electron toward itself and gains a relatively large partial negative
charge.
In turn, the
hydrogen acquires
a similarly large
partial positive
charge that
strongly attracts
the partial negative
charge of a
neighboring
electronegative
atom. Typically, a
hydrogen bond is
about five to ten
times stronger than
other dipole –
dipole attractions.
Hydrogen bonds
keep the DNA bases
paired together,
helping DNA
maintain its unique
structure.
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Hydrogen
Hydrogen bond donor
Bond
A hydrogen atom attached to a relatively electronegative atom is usually
fluorine, oxygen, or nitrogen. In the molecule on ethanol, there is one
hydrogen atom bonded to an oxygen atom, which is very electronegative.
This hydrogen atom is a hydrogen donor.
Hydrogen bond
acceptor
Hydrogen bond
results when this
strong partial
positive charge
attracts a lone
pair of electrons
on another atom,
which becomes
the hydrogen
bond acceptor.
An
electronegative
atom such as
fluorine, oxygen,
or nitrogen is a
hydrogen bond
acceptor.
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
ION – DIPOLE FORCES
Ion – dipole force acts between an
ion (either positive or negative) and
a polar molecule. Such is the case of
the aqueous solution of sodium
chloride, where Na+ and Cl- ions are
Ion- dispersed amidst polar water
molecule. The degree of ion-dipole
Dipole
interaction depends on the size and
charge of the ion and on the size and
dipole moment of the polar
Force molecule. Cations (positively
charged ions) interact more strongly
with dipoles than anions (negatively
charged ions) of the same magnitude
of charge since they are smaller. This
means that between Na+ and Cl-
(with +1 and -1 charges,
respectively), Na+ will have greater
interactions with water than Cl-. Ion-
dipole attraction becomes stronger
either as the charge of the ion
increases, or as the magnitude of the
dipole moment of the polar molecule
increases.
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Dipole-Dipole
Force Each polar molecule has
unequal electron
DIPOLE – DIPOLE FORCES densities, resulting in a
Dipole –dipole forces exist dipole – a partial positive
between polar molecules. Recall end and a partial negative
that polar substances, such as end. Thus, when two
hydrogen chloride (HCl), are polar molecules are
those that possess a permanent brought close together,
dipole moment attributed to the the partial positive charge
different electronegativities of of one molecule will be
their component atoms and how attracted to the partial
these atoms are arranged in negative charge of the
space. neighboring molecule.
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to
learn?
Dispersion
London dispersion
forces, or simply Force
freedom to move
about the molecule;
instantaneous dipole
and an induced
dipole. The size of
dispersion forces, therefore, at any
are intermolecular instance, the molecule can affect
forces of attraction molecule the London
that exist between momentarily dispersion force
all atoms and acquires a non- between two
molecules. They uniform electron molecules. The
arise from the density, resulting in bigger the molecule
continuous a temporary dipole (which means that
movement of (also called the molecule has
electrons in instantaneous greater surface area),
particles. None dipole). This dipole the strong er the
polar molecules can then induce attraction between
have zero dipole dipoles in two molecules. Ex.
movement because neighboring between helium and
their electron molecules. argon, 2 argon atoms
density is uniform Therefore, London will have greater
and symmetrical. dispersion forces LDF because they
Nevertheless, the are attractions are bigger than the
electrons have some between an helium atoms.
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to do?
Fill in the blanks with appropriate concepts based from what
you have learned from the discussion.
1. The weakest type of intermolecular force is called
_________________________.
(London Dispersion Forces)
(Hydrogen Bond)
(Dipole-Dipole Forces)
(Ion-Dipole Forces)
2. Dipole-dipole intermolecular force can be formed between
two _________________ molecules.
(Ionic)
(Polar)
(Non-Polar)
(Atomic)
3. The intermolecular forces holds _________ together while
intramolecular forces holds ________ together.
(molecules, atoms)
(atoms, molecules)
4. Ion-dipole force is a force which can hold __________ and
_________ together.
(Non-polar, polar)
(Atom, non-polar)
(ion, polar)
5. The strongest type of intermolecular force is called
__________________.
(London Dispersion Forces)
(Hydrogen Bond)
(Dipole-Dipole Forces)
(Ion-Dipole Forces)
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
What to do?
Activity #2
What is the strongest intermolecular force
present for each of the following molecules?
1. Hydrogen - ______________________________
2. Carbon Monoxide - ______________________
3. Silicon Tetrafluoride –
____________________
4. Nitrogen Tribromide - ___________________
5. Water - _________________________________
6. Acetone – _______________________________
7. Methane –
_______________________________
8. Benzene – _______________________________
9. Ammonia –
______________________________
10.Methanol - ______________________________
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
Who should be
asked?
Katewin Mae H. Acdal
21 years old
Calaocan, Santiago City
Click the icons to proceed
MAGSCIEYA Intermolecular Forces
Вам также может понравиться
- The Cell Structure & Fxns FOR OBSERVATIONДокумент48 страницThe Cell Structure & Fxns FOR OBSERVATIONAlyssaОценок пока нет
- Plant Reproduction: Click To EnterДокумент41 страницаPlant Reproduction: Click To EnterAlyssaОценок пока нет
- Name: - Section: - Date: - Module/ ActivityДокумент8 страницName: - Section: - Date: - Module/ ActivityAlyssaОценок пока нет
- What Happens When You Mix Substances Together?Документ19 страницWhat Happens When You Mix Substances Together?AlyssaОценок пока нет
- Section 32 Acids and BasesДокумент55 страницSection 32 Acids and BasesAlyssaОценок пока нет
- KinematicsДокумент52 страницыKinematicsAlyssaОценок пока нет
- Webform: Pproach Ubsection Equired For All HesesДокумент6 страницWebform: Pproach Ubsection Equired For All HesesAlyssaОценок пока нет
- Final ThesisДокумент105 страницFinal ThesisAlyssaОценок пока нет
- The Yellow House: A Memoir (2019 National Book Award Winner)От EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Рейтинг: 4 из 5 звезд4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeОт EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeРейтинг: 4 из 5 звезд4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingОт EverandThe Little Book of Hygge: Danish Secrets to Happy LivingРейтинг: 3.5 из 5 звезд3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureОт EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureРейтинг: 4.5 из 5 звезд4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryОт EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryРейтинг: 3.5 из 5 звезд3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceОт EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceРейтинг: 4 из 5 звезд4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnОт EverandTeam of Rivals: The Political Genius of Abraham LincolnРейтинг: 4.5 из 5 звезд4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItОт EverandNever Split the Difference: Negotiating As If Your Life Depended On ItРейтинг: 4.5 из 5 звезд4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerОт EverandThe Emperor of All Maladies: A Biography of CancerРейтинг: 4.5 из 5 звезд4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaОт EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaРейтинг: 4.5 из 5 звезд4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaОт EverandThe Unwinding: An Inner History of the New AmericaРейтинг: 4 из 5 звезд4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersОт EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersРейтинг: 4.5 из 5 звезд4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyОт EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyРейтинг: 3.5 из 5 звезд3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreОт EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreРейтинг: 4 из 5 звезд4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)От EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Рейтинг: 4.5 из 5 звезд4.5/5 (121)
- Chem Chapter 4 NotesДокумент11 страницChem Chapter 4 NotesTara HebbarОценок пока нет
- Imp Questions ChemistryДокумент6 страницImp Questions ChemistryAnuradha UppalapatiОценок пока нет
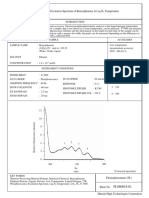
- Spectrophotometric Analysis of KMnO4 SolutionsДокумент5 страницSpectrophotometric Analysis of KMnO4 SolutionsHassan Ali Samoo100% (5)
- NMR SpectrosДокумент45 страницNMR SpectrosfatemaОценок пока нет
- Benzoic Acid - SpectrosДокумент12 страницBenzoic Acid - SpectrosY.P.Singh100% (1)
- 1454924410CHE P8 M24 E-TextДокумент8 страниц1454924410CHE P8 M24 E-TextVimalОценок пока нет
- 150 and More NMR ExperimentsДокумент129 страниц150 and More NMR Experimentsustorzer100% (1)
- C - Done at School - To Move Home Strong and Weak Acids and Bases Phet LabДокумент2 страницыC - Done at School - To Move Home Strong and Weak Acids and Bases Phet Labapi-262304087Оценок пока нет
- AtomicДокумент3 страницыAtomicRashidОценок пока нет
- Introduction To Intermolecular ForcesДокумент8 страницIntroduction To Intermolecular ForcesLouie DelavegaОценок пока нет
- Ap ch9 SQДокумент18 страницAp ch9 SQHuấnĐìnhNguyễnОценок пока нет
- Accepted Manuscript: PolyhedronДокумент41 страницаAccepted Manuscript: PolyhedronkawtherahmedОценок пока нет
- Phosphorescence Excitation Spectrum of Benzophenone at Liq.N TemperatureДокумент5 страницPhosphorescence Excitation Spectrum of Benzophenone at Liq.N TemperatureNisar Ali Mphil-Chem AОценок пока нет
- Nuclear Magnetic ResonanceДокумент34 страницыNuclear Magnetic ResonanceD.N.S DE ZOYSAОценок пока нет
- Chemical Bonding PDFДокумент80 страницChemical Bonding PDFQuỳnh PhạmОценок пока нет
- Lab Report Scientific Communiation PDFДокумент9 страницLab Report Scientific Communiation PDFMuhammad QasimОценок пока нет
- Student Exploration: Covalent BondsДокумент4 страницыStudent Exploration: Covalent BondsDjeneba DembeleОценок пока нет
- Chemical Bonding (Basic) : Inorganic ChemistryДокумент37 страницChemical Bonding (Basic) : Inorganic ChemistrySATYAM GAMINGОценок пока нет
- Lewis Structures Molecular Geometry and Polarity 1A KEYДокумент3 страницыLewis Structures Molecular Geometry and Polarity 1A KEYrsleoОценок пока нет
- Molecule Shapes ActivityДокумент7 страницMolecule Shapes ActivitySilverio, Mark Andre L.Оценок пока нет
- Quantum Numbers WorksheetДокумент2 страницыQuantum Numbers WorksheetSepehr Masoumi-AlamoutiОценок пока нет
- Gaussian TipsДокумент70 страницGaussian Tips顧浩Оценок пока нет
- (Career Endeavour) MOLECULAR COMPOUNDSДокумент15 страниц(Career Endeavour) MOLECULAR COMPOUNDSVaibhav NikharОценок пока нет
- Tut 4 UV-Vis With SolutionsДокумент2 страницыTut 4 UV-Vis With SolutionsLondeka Buthelezi100% (1)
- Slater'S Rules and Periodicity (Worksheet) : Skills To DevelopДокумент3 страницыSlater'S Rules and Periodicity (Worksheet) : Skills To DevelopRituraj TripathyОценок пока нет
- Class 8 - Structure of Atom ppt2Документ18 страницClass 8 - Structure of Atom ppt2mau_chak5449Оценок пока нет
- Band TheoryДокумент19 страницBand TheoryTwishaОценок пока нет
- FTIR, Raman y SERS Eosin YДокумент8 страницFTIR, Raman y SERS Eosin YMarceloОценок пока нет
- Chemistry Unit-1Документ32 страницыChemistry Unit-1RajeshОценок пока нет
- Chemistry Chapter 6 ReviewДокумент3 страницыChemistry Chapter 6 Reviewjuan carlos quispe mamaniОценок пока нет