Документssoar-2003-berthoin_antal_et_al-hoechst_challenges_received_wisdom_onзагружено:
Документssoar-2003-berthoin_antal_et_al-hoechst_challenges_received_wisdom_onзагружено: hicham1963
hicham1963 Документcir_48-DMP-00_fiche_de_declarationзагружено:
Документcir_48-DMP-00_fiche_de_declarationзагружено: hicham1963
hicham1963 Документcirculaire104-DMPзагружено:
Документcirculaire104-DMPзагружено: hicham1963
hicham1963 ДокументLAMBERT_JEREMY_2014загружено:
ДокументLAMBERT_JEREMY_2014загружено: hicham1963
hicham1963 ДокументPharma physicasзагружено:
ДокументPharma physicasзагружено: hicham1963
hicham1963 ДокументCarobhydrate metabolismзагружено:
ДокументCarobhydrate metabolismзагружено: hicham1963
hicham1963 ДокументNIRALI Pharmaceutics Iзагружено:
ДокументNIRALI Pharmaceutics Iзагружено: hicham1963
hicham1963 ДокументAdvanced Instrumentation Techniques (Nirali Prakashan) (1)загружено:
ДокументAdvanced Instrumentation Techniques (Nirali Prakashan) (1)загружено: hicham1963
hicham1963 ДокументLa-Vague-61загружено:
ДокументLa-Vague-61загружено: hicham1963
hicham1963 ДокументLaVague_60загружено:
ДокументLaVague_60загружено: hicham1963
hicham1963![«[Drying Technology ] Gagnon, Francis_ Bouchard, Jocelyn_ Desbiens, André_ Poulin, Éri - Development and validation of a batch fluidized bed dryer model for pharmaceutical particles (2020) [10.1080_07373937.201» завершено на 0%](https://s-f.scribdassets.com/images/progress/bar.png?932fa3dc0) Документ[Drying Technology ] Gagnon, Francis_ Bouchard, Jocelyn_ Desbiens, André_ Poulin, Éri - Development and validation of a batch fluidized bed dryer model for pharmaceutical particles (2020) [10.1080_07373937.201загружено:
Документ[Drying Technology ] Gagnon, Francis_ Bouchard, Jocelyn_ Desbiens, André_ Poulin, Éri - Development and validation of a batch fluidized bed dryer model for pharmaceutical particles (2020) [10.1080_07373937.201загружено: hicham1963
hicham1963 ДокументValidation-dun-procede-defabrication-dune-forme-secheзагружено:
ДокументValidation-dun-procede-defabrication-dune-forme-secheзагружено: hicham1963
hicham1963 Документthese_susanne_muschertзагружено:
Документthese_susanne_muschertзагружено: hicham1963
hicham1963 ДокументScdpha t 2008 El Haimer Sihamзагружено:
ДокументScdpha t 2008 El Haimer Sihamзагружено: hicham1963
hicham1963 ДокументBUPHA_T_2017_PREVOST_HUGUESзагружено:
ДокументBUPHA_T_2017_PREVOST_HUGUESзагружено: hicham1963
hicham1963 Документ2017TOU32032загружено:
Документ2017TOU32032загружено: hicham1963
hicham1963 Документ2018LILUE136загружено:
Документ2018LILUE136загружено: hicham1963
hicham1963 Документ2017LIL2E163загружено:
Документ2017LIL2E163загружено: hicham1963
hicham1963 Документ2016LIL2S025загружено:
Документ2016LIL2S025загружено: hicham1963
hicham1963 ДокументThese Carboxymethyl Amidonзагружено:
ДокументThese Carboxymethyl Amidonзагружено: hicham1963
hicham1963 ДокументScale up of HSM granulatorsзагружено:
ДокументScale up of HSM granulatorsзагружено: hicham1963
hicham1963 ДокументLA DEMARCHE QUALITY BY DESIGNзагружено:
ДокументLA DEMARCHE QUALITY BY DESIGNзагружено: hicham1963
hicham1963 Документhkmbioavailabilityзагружено:
Документhkmbioavailabilityзагружено: hicham1963
hicham1963![«[C30] Technical Specification_EN» завершено на 0%](https://s-f.scribdassets.com/images/progress/bar.png?932fa3dc0) Документ[C30] Technical Specification_ENзагружено:
Документ[C30] Technical Specification_ENзагружено: hicham1963
hicham1963 ДокументVa Gendre Claire 12122011загружено:
ДокументVa Gendre Claire 12122011загружено: hicham1963
hicham1963 ДокументOpérations pharmaceutiques_ Enrobageзагружено:
ДокументOpérations pharmaceutiques_ Enrobageзагружено: hicham1963
hicham1963![«[Thérapie vol. 71 iss. 2] Montastruc, Jean-Louis_ Bondon-Guitton, Emmanuelle_ Abadie, Delp - Pharmacovigilance _ risques et effets indésirables de l’automédication (2016) [10.1016_j.therap.2016.02.011] - libge» завершено на 0%](https://s-f.scribdassets.com/images/progress/bar.png?932fa3dc0) Документ[Thérapie vol. 71 iss. 2] Montastruc, Jean-Louis_ Bondon-Guitton, Emmanuelle_ Abadie, Delp - Pharmacovigilance _ risques et effets indésirables de l’automédication (2016) [10.1016_j.therap.2016.02.011] - libgeзагружено:
Документ[Thérapie vol. 71 iss. 2] Montastruc, Jean-Louis_ Bondon-Guitton, Emmanuelle_ Abadie, Delp - Pharmacovigilance _ risques et effets indésirables de l’automédication (2016) [10.1016_j.therap.2016.02.011] - libgeзагружено: hicham1963
hicham1963 ДокументLA TECHNOLOGIE «LIT FLUIDISE» Séchage - granulation - enrobage. S. VERRIER ed. 2007-1загружено:
ДокументLA TECHNOLOGIE «LIT FLUIDISE» Séchage - granulation - enrobage. S. VERRIER ed. 2007-1загружено: hicham1963
hicham1963![«[Drug Development and Industrial Pharmacy vol. 8 iss. 1] Rudnic, E. M._ Rhodes, C. T._ Welch, S._ Bernardo, P. - Evaluations of the Mechanism of Disintegrant Action (1982) [10.3109_03639048209052562] - libgen.» завершено на 0%](https://s-f.scribdassets.com/images/progress/bar.png?932fa3dc0) Документ[Drug Development and Industrial Pharmacy vol. 8 iss. 1] Rudnic, E. M._ Rhodes, C. T._ Welch, S._ Bernardo, P. - Evaluations of the Mechanism of Disintegrant Action (1982) [10.3109_03639048209052562] - libgen.загружено:
Документ[Drug Development and Industrial Pharmacy vol. 8 iss. 1] Rudnic, E. M._ Rhodes, C. T._ Welch, S._ Bernardo, P. - Evaluations of the Mechanism of Disintegrant Action (1982) [10.3109_03639048209052562] - libgen.загружено: hicham1963
hicham1963 ДокументManuel des bonnes pratiques de PVзагружено:
ДокументManuel des bonnes pratiques de PVзагружено: hicham1963
hicham1963 ДокументFundamentalofcGMPforPharmaceuticalHVACSystemзагружено:
ДокументFundamentalofcGMPforPharmaceuticalHVACSystemзагружено: hicham1963
hicham1963 Документdraft-international-conference-harmonisation-technical-requirements-registration-pharmaceuticals_en-1загружено:
Документdraft-international-conference-harmonisation-technical-requirements-registration-pharmaceuticals_en-1загружено: hicham1963
hicham1963![«[Powder Technology vol. 302] Gopireddy, Srikanth R._ Hildebrandt, Claudia_ Urbanetz, Nora A. - Numerical simulation of powder flow in a pharmaceutical tablet press lab-scale gravity feeder (2016) [10.1016_j.po» завершено на 0%](https://s-f.scribdassets.com/images/progress/bar.png?932fa3dc0) Документ[Powder Technology vol. 302] Gopireddy, Srikanth R._ Hildebrandt, Claudia_ Urbanetz, Nora A. - Numerical simulation of powder flow in a pharmaceutical tablet press lab-scale gravity feeder (2016) [10.1016_j.poзагружено:
Документ[Powder Technology vol. 302] Gopireddy, Srikanth R._ Hildebrandt, Claudia_ Urbanetz, Nora A. - Numerical simulation of powder flow in a pharmaceutical tablet press lab-scale gravity feeder (2016) [10.1016_j.poзагружено: hicham1963
hicham1963![«[Pharmaceutical Research vol. 08 iss. 3] Dorothy R. Heidemann_ Paul J. Jarosz - Preformulation Studies Involving Moisture Uptake in Solid Dosage Forms (1991) [10.1023_a_1015877011807] - libgen.li» завершено на 0%](https://s-f.scribdassets.com/images/progress/bar.png?932fa3dc0) Документ[Pharmaceutical Research vol. 08 iss. 3] Dorothy R. Heidemann_ Paul J. Jarosz - Preformulation Studies Involving Moisture Uptake in Solid Dosage Forms (1991) [10.1023_a_1015877011807] - libgen.liзагружено:
Документ[Pharmaceutical Research vol. 08 iss. 3] Dorothy R. Heidemann_ Paul J. Jarosz - Preformulation Studies Involving Moisture Uptake in Solid Dosage Forms (1991) [10.1023_a_1015877011807] - libgen.liзагружено: hicham1963
hicham1963 ДокументGood Pharmaceutical Manufacturing Practice_ Rationale and Complianceзагружено:
ДокументGood Pharmaceutical Manufacturing Practice_ Rationale and Complianceзагружено: hicham1963
hicham1963![«[PharmacoEconomics vol. 20 iss. Supplement 3] DiMasi, Joseph A. - The Value of Improving the Productivity of the Drug Development Process (2002) [10.2165_00019053-200220003-00001] - libgen.li» завершено на 0%](https://s-f.scribdassets.com/images/progress/bar.png?932fa3dc0) Документ[PharmacoEconomics vol. 20 iss. Supplement 3] DiMasi, Joseph A. - The Value of Improving the Productivity of the Drug Development Process (2002) [10.2165_00019053-200220003-00001] - libgen.liзагружено:
Документ[PharmacoEconomics vol. 20 iss. Supplement 3] DiMasi, Joseph A. - The Value of Improving the Productivity of the Drug Development Process (2002) [10.2165_00019053-200220003-00001] - libgen.liзагружено: hicham1963
hicham1963 ДокументR_and_D_management_in_the_pharma_industrзагружено:
ДокументR_and_D_management_in_the_pharma_industrзагружено: hicham1963
hicham1963 Документ06 - Processus de Recherche, Développement Et Production Du Médicamentзагружено:
Документ06 - Processus de Recherche, Développement Et Production Du Médicamentзагружено: hicham1963
hicham1963 ДокументCours de Psychologieзагружено:
ДокументCours de Psychologieзагружено: hicham1963
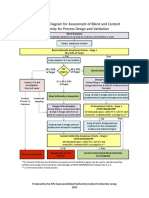
hicham1963 ДокументBlend Content Uniformity Process Flow Diagram Design Validationзагружено:
ДокументBlend Content Uniformity Process Flow Diagram Design Validationзагружено: hicham1963
hicham1963 Документ208135002-Norme-afnor-x-35-109-fr-pdf.pdfзагружено:
Документ208135002-Norme-afnor-x-35-109-fr-pdf.pdfзагружено: hicham1963
hicham1963 ДокументSample Article Frзагружено:
ДокументSample Article Frзагружено: hicham1963
hicham1963 Документ6 Sigma Mesureзагружено:
Документ6 Sigma Mesureзагружено: hicham1963
hicham1963 ДокументSix Sigmaзагружено:
ДокументSix Sigmaзагружено: hicham1963
hicham1963
Академический Документы
Профессиональный Документы
Культура Документы










![[Drying Technology ] Gagnon, Francis_ Bouchard, Jocelyn_ Desbiens, André_ Poulin, Éri - Development and validation of a batch fluidized bed dryer model for pharmaceutical particles (2020) [10.1080_07373937.201](https://imgv2-1-f.scribdassets.com/img/document/563220522/149x198/1107a16e0d/1710562391?v=1)












![[C30] Technical Specification_EN](https://imgv2-1-f.scribdassets.com/img/document/563165078/149x198/650b80ec14/1710532952?v=1)


![[Thérapie vol. 71 iss. 2] Montastruc, Jean-Louis_ Bondon-Guitton, Emmanuelle_ Abadie, Delp - Pharmacovigilance _ risques et effets indésirables de l’automédication (2016) [10.1016_j.therap.2016.02.011] - libge](https://imgv2-1-f.scribdassets.com/img/document/563164469/149x198/4a5c12dcf9/1710532951?v=1)

![[Drug Development and Industrial Pharmacy vol. 8 iss. 1] Rudnic, E. M._ Rhodes, C. T._ Welch, S._ Bernardo, P. - Evaluations of the Mechanism of Disintegrant Action (1982) [10.3109_03639048209052562] - libgen.](https://imgv2-2-f.scribdassets.com/img/document/563164261/149x198/b97efc938f/1710542382?v=1)



![[Powder Technology vol. 302] Gopireddy, Srikanth R._ Hildebrandt, Claudia_ Urbanetz, Nora A. - Numerical simulation of powder flow in a pharmaceutical tablet press lab-scale gravity feeder (2016) [10.1016_j.po](https://imgv2-2-f.scribdassets.com/img/document/563151414/149x198/49d0de6006/1710585250?v=1)
![[Pharmaceutical Research vol. 08 iss. 3] Dorothy R. Heidemann_ Paul J. Jarosz - Preformulation Studies Involving Moisture Uptake in Solid Dosage Forms (1991) [10.1023_a_1015877011807] - libgen.li](https://imgv2-2-f.scribdassets.com/img/document/563151169/149x198/1a531367bd/1710532963?v=1)

![[PharmacoEconomics vol. 20 iss. Supplement 3] DiMasi, Joseph A. - The Value of Improving the Productivity of the Drug Development Process (2002) [10.2165_00019053-200220003-00001] - libgen.li](https://imgv2-2-f.scribdassets.com/img/document/563121703/149x198/e229894b6d/1710562392?v=1)