ДокументHCF-Guidelines-Proposal Template-2021загружено:
ДокументHCF-Guidelines-Proposal Template-2021загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMedical Devices Violation - Ver 2.1загружено:
ДокументMedical Devices Violation - Ver 2.1загружено: Mohamed Ezzat
Mohamed Ezzat ДокументResolution No. (24) for 2021загружено:
ДокументResolution No. (24) for 2021загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMedical Devices Authorized Representatives Check Listзагружено:
ДокументMedical Devices Authorized Representatives Check Listзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументInspection Request Form (1)загружено:
ДокументInspection Request Form (1)загружено: Mohamed Ezzat
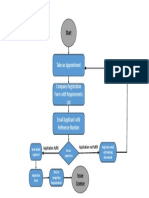
Mohamed Ezzat ДокументProcess of Authorized Representative Registration (1)загружено:
ДокументProcess of Authorized Representative Registration (1)загружено: Mohamed Ezzat
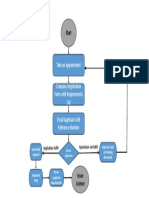
Mohamed Ezzat Документprocess of Authorized Representative registration (1)загружено:
Документprocess of Authorized Representative registration (1)загружено: Mohamed Ezzat
Mohamed Ezzat Документabc flayer_Modзагружено:
Документabc flayer_Modзагружено: Mohamed Ezzat
Mohamed Ezzat Документabc flayerзагружено:
Документabc flayerзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументPricing Proposal NHRAзагружено:
ДокументPricing Proposal NHRAзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументMagedSh - CVзагружено:
ДокументMagedSh - CVзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументMagedSh - CL (1)загружено:
ДокументMagedSh - CL (1)загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMD Companies Registrationзагружено:
ДокументMD Companies Registrationзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументABC prop p1загружено:
ДокументABC prop p1загружено: Mohamed Ezzat
Mohamed Ezzat Документtable 2загружено:
Документtable 2загружено: Mohamed Ezzat
Mohamed Ezzat Документproposal ar and mdrзагружено:
Документproposal ar and mdrзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументTwins and Siblingsзагружено:
ДокументTwins and Siblingsзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDD_11163-2017-CE-KOR-NA-PS Rev. 6.0загружено:
ДокументMDD_11163-2017-CE-KOR-NA-PS Rev. 6.0загружено: Mohamed Ezzat
Mohamed Ezzat ДокументUzinmedicareзагружено:
ДокументUzinmedicareзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументISO13485_10317-2017-AQ-KOR-NA-PS Rev. 2.0.- 20210413загружено:
ДокументISO13485_10317-2017-AQ-KOR-NA-PS Rev. 2.0.- 20210413загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 11_Prohibition of Importation of Transvaginal Mesh_2018загружено:
ДокументMDR_Circular 11_Prohibition of Importation of Transvaginal Mesh_2018загружено: Mohamed Ezzat
Mohamed Ezzat ДокументCircular 12 2018загружено:
ДокументCircular 12 2018загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular_21_2020_Extension of Temporary Holdup of Receiving New Medical Devices and Supplies Registration Requestsзагружено:
ДокументMDR_Circular_21_2020_Extension of Temporary Holdup of Receiving New Medical Devices and Supplies Registration Requestsзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular_21_2020_Extension of temporary holdup of receiving new medical devices and supplies registration requestsзагружено:
ДокументMDR_Circular_21_2020_Extension of temporary holdup of receiving new medical devices and supplies registration requestsзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 8_Classification of criteria of Combined Medical Device_2018загружено:
ДокументMDR_Circular 8_Classification of criteria of Combined Medical Device_2018загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 7 _New Appointment Booking System_2019загружено:
ДокументMDR_Circular 7 _New Appointment Booking System_2019загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 8_2017загружено:
ДокументMDR_Circular 8_2017загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 10_Prohibition of Importation of Powdered Medical Gloves_2018загружено:
ДокументMDR_Circular 10_Prohibition of Importation of Powdered Medical Gloves_2018загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular_17_2020_Guideline of hand sanitizersзагружено:
ДокументMDR_Circular_17_2020_Guideline of hand sanitizersзагружено: Mohamed Ezzat
Mohamed Ezzat Документno 45 for 2020загружено:
Документno 45 for 2020загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 5_Medical Devices Certificate Verification and Validation_2018загружено:
ДокументMDR_Circular 5_Medical Devices Certificate Verification and Validation_2018загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular_7_2020_Rapid test for research purposes only and not for diagnosisзагружено:
ДокументMDR_Circular_7_2020_Rapid test for research purposes only and not for diagnosisзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 14_New Details to clear Medical Devices Shipments_2017загружено:
ДокументMDR_Circular 14_New Details to clear Medical Devices Shipments_2017загружено: Mohamed Ezzat
Mohamed Ezzat Документ2021_Circular No. (4) 2021загружено:
Документ2021_Circular No. (4) 2021загружено: Mohamed Ezzat
Mohamed Ezzat ДокументCircular No. ) 2 ( 2021загружено:
ДокументCircular No. ) 2 ( 2021загружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular_6_2020_Air Doctor Productзагружено:
ДокументMDR_Circular_6_2020_Air Doctor Productзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументMDR_Circular 10_A new Document to clear the shipment of medical device_2017загружено:
ДокументMDR_Circular 10_A new Document to clear the shipment of medical device_2017загружено: Mohamed Ezzat
Mohamed Ezzat ДокументNHRA Closed due to Shifting to New Locationзагружено:
ДокументNHRA Closed due to Shifting to New Locationзагружено: Mohamed Ezzat
Mohamed Ezzat Документ401474910 CGP CGP GSCE Chemistry AQA Revision Guide 2018 PDFзагружено:
Документ401474910 CGP CGP GSCE Chemistry AQA Revision Guide 2018 PDFзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументWPI_Log_2017.03.26_17.33.58загружено:
ДокументWPI_Log_2017.03.26_17.33.58загружено: Mohamed Ezzat
Mohamed Ezzat ДокументWPI_Log_2017.03.26_17.17.44загружено:
ДокументWPI_Log_2017.03.26_17.17.44загружено: Mohamed Ezzat
Mohamed Ezzat Документmktg-1030-swot-presentationзагружено:
Документmktg-1030-swot-presentationзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументIndicesзагружено:
ДокументIndicesзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументAdverse Event Procedureзагружено:
ДокументAdverse Event Procedureзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументSupplementary Exercise 3 - Summary statisticsзагружено:
ДокументSupplementary Exercise 3 - Summary statisticsзагружено: Mohamed Ezzat
Mohamed Ezzat Документreview report for renewзагружено:
Документreview report for renewзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументField Safety Noticeзагружено:
ДокументField Safety Noticeзагружено: Mohamed Ezzat
Mohamed Ezzat ДокументAdverse Event-procedure (2)загружено:
ДокументAdverse Event-procedure (2)загружено: Mohamed Ezzat
Mohamed Ezzat ДокументComplaint Formзагружено:
ДокументComplaint Formзагружено: Mohamed Ezzat
Mohamed Ezzat
Академический Документы
Профессиональный Документы
Культура Документы